��Ŀ����
3����ͼ��s�ܼ���p�ܼ���ԭ�ӹ��ͼ���Իش��������⣺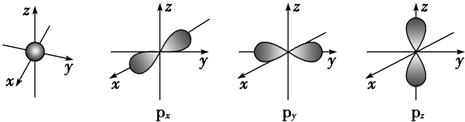
��1��s���ӵ�ԭ�ӹ�������Σ�ÿ��s�ܼ���1��ԭ�ӹ����p���ӵ�ԭ�ӹ���ʷĴ��Σ�ÿ��p�ܼ���3��ԭ�ӹ����
��2��Ԫ��X��ԭ�����������Ų�ʽΪnsnnpn+1��Ԫ��X�������ǵ�Ԫ�أ���ԭ����������ߵ���2p���ӣ������⻯��ĵ���ʽ��
 ��
����3����Ԫ��X��ԭ�����������Ų�ʽΪnsn-1npn+1����ôX��Ԫ�ط���ΪS��
���� ��1������ͼƬ�й������״���з������ע��p�ܼ���3�������
��2��Ԫ��X��ԭ�����������Ų�ʽΪnsnnpn+1��s�ܼ������2�����ӣ���Ԫ��������p�ܼ���˵��s�ܼ��Ѿ�����������n=2�����Ԫ�����������Ų�ʽΪ2s22p3�����Ԫ����NԪ�أ�
��3����Ԫ��X��ԭ�����������Ų�ʽΪnsn-1npn+1ʱ����n-1=2����n=3��Ϊ��Ԫ�أ�
��� �⣺��1������ͼƬ��s���ӵ�ԭ�ӹ�������Σ�����1��ԭ�ӹ����p���ӵ�ԭ�ӹ���ʷĴ��Σ�ÿ��p�ܼ���3��ԭ�ӹ������������������ֱ��
�ʴ�Ϊ����1���Ĵ���3��
��2����ΪԪ��X��ԭ�����������Ų�ʽΪnsnnn+1��np��������ϵ��ӣ�˵��ns������������ӣ���n=2����Ԫ��X��ԭ�Ӻ�������Ų�ʽΪ1s22s22p3���ǵ�Ԫ�أ�ԭ����������ߵ���2p���ӣ���������ڿռ����������ഹֱ����չ�����⻯��ĵ���ʽ�� ��
��
�ʴ�Ϊ����Ԫ�أ�2p�� ��
��
��3����Ԫ��X��ԭ�����������Ų�ʽΪnsn-1npn+1ʱ����n-1=2����n=3����XԪ�ص�ԭ�Ӻ�������Ų�ʽΪ1s22s22p63s23p4������Ԫ�أ�Ԫ�ط���ΪS��
�ʴ�Ϊ��S��
���� ���⿼����ԭ�Ӻ�������Ų��������չ�����֪ʶ�㣬��ȷ�ƶ�Ԫ���ǽⱾ��ؼ�����Ŀ�ѶȲ���

 �п������п��Ծ����ϵ�д�
�п������п��Ծ����ϵ�д� ��������״Ԫ��ϵ�д�
��������״Ԫ��ϵ�д� �ƸԿ�����ҵ��ϵ�д�
�ƸԿ�����ҵ��ϵ�д� ��Ԫ����ĩ��ϰ�ȷ��ϵ�д�
��Ԫ����ĩ��ϰ�ȷ��ϵ�д�| A�� | ���Ӽ�����������֮��ľ������� | |
| B�� | ֻ��ԭ�Ӳ���������������ͺ����������� | |
| C�� | ���ӻ�������һ���н���Ԫ�� | |
| D�� | 뭡�뮡�밻�Ϊ��Ԫ����ͬλ�أ�H2O��D2O��T2O�Dz�ͬ�ķ��� |
| ֥�齴ÿ100 g���� �����ʣ�19.2 g ֬����52.7 g ̼ˮ������16.8 g ά���� A��17 ��g ����50.3 mg ����4 ��g ���ƣ��൱��300 g���� �������൱��800 g���� �����ѣ�֥�齴������Ӥ��������Ƥ�ף� |
����ˮ�����ɰ�����������ǵ����ʣ�
��������ȱ��ά����A����ҹä֢��
��2����֥�齴˵������ж���ȷ����C ������ţ���
A��������Ԫ�� B�������������֬
C��Ӥ�����ʳ�� D���ƺ������ڶ����ͼ�����
| A�� | ��BaCl2��Һ����SO42- �� SO32- | |
| B�� | ��ŨHNO3��Cu ��Ӧ�Ʊ�NO2 | |
| C�� | �����ⷽ���ѹ������ʼӹ���������1nm-100nm�� �ij�ϸ��ĩ���ӣ��Ƶ����ײ��ϣ����ײ���Ҳ��һ�ֽ��� | |
| D�� | ������HCl��Cl2ͨ�뱥��NaCl��Һ�г�ȥHCl |
| A�� | CH2=CH2+HOCl��HOCH2-CH2Cl | |
| B�� |  +HNO3$��_{��}^{Ũ����}$ +HNO3$��_{��}^{Ũ����}$ +H2O +H2O | |
| C�� | 2CH3CH2OH+O2 $��_{��}^{Cu}$2CH3CHO+2H2O | |
| D�� | C12H22O11+H2O $��_{��}^{ϡH_{2}SO_{4}}$ C6H12O6+C6H12O6 ���� ������ ���� |
 ��ѧʵ���ǽ��п�ѧ̽������Ҫ��ʽ�������Ļ�ѧʵ�鼼����ѧϰ��ѧ�ͽ���̽����Ļ����ͱ�֤������Ҫ��ش��������⣮
��ѧʵ���ǽ��п�ѧ̽������Ҫ��ʽ�������Ļ�ѧʵ�鼼����ѧϰ��ѧ�ͽ���̽����Ļ����ͱ�֤������Ҫ��ش��������⣮
 ����������Ԫ��A��B��C��D��E��ԭ��������������AԪ�صĵ��ʳ��³���������������壬BԪ�����γɻ�����������࣬C������������Ӧˮ����������̬�⻯�����ܹ������γ��α���DԪ�ص����Ӱ뾶��ͬ����Ԫ���γɵļ���������С�ģ�
����������Ԫ��A��B��C��D��E��ԭ��������������AԪ�صĵ��ʳ��³���������������壬BԪ�����γɻ�����������࣬C������������Ӧˮ����������̬�⻯�����ܹ������γ��α���DԪ�ص����Ӱ뾶��ͬ����Ԫ���γɵļ���������С�ģ�