��Ŀ����
8����ҵ�����к����ˮ���ŷŻ���Ⱦ��������Ҫ�Ժ����ˮ���д��������ã���1��ij�Ƹﳧ�����ˮ����Ҫ��������Na2S��
�ٳ����£���ø÷�ˮ��ҺpH=12�������ӷ���ʽ������Һ�ʼ��Ե�ԭ��S2-+H2O?HS-+OH-��
�ں����ˮ�Ĵ��������п��Բ��ô�����Na2Sת��ΪNa2SO4����Ӧ�л�ԭ���������������ʵ���֮��Ϊ1��2����֪1000��ʱ���������������������з�Ӧ��Na2SO4��s��+4H2��g��?Na2S��s��+4H2O��g������֪�÷�Ӧ��ƽ�ⳣ��K1000�棼K1400�棬��÷�Ӧ�ġ�H��0���������=����������
��2������ȼ���м����ˮ����Ҫ������ΪNa2SO3�����շ�ˮ�е�����Լ�����Դ���˷ѣ��ɲ������·�����
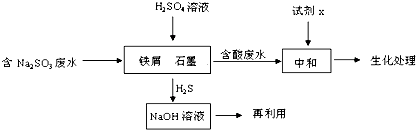
��д��H2S����������NaOH��Һ��Ӧ�Ļ�ѧ����ʽH2S+2NaOH=Na2S+2H2O
����м��ʯī���γ���ԭ��أ�SO${\;}_{3}^{2-}$�����������·ŵ�����H2S��������Ӷ��ﵽ�ӷ�ˮ�г�ȥNa2SO3��Ŀ�ģ�д��SO${\;}_{3}^{2-}$�����������·ŵ�����H2S�ĵ缫��Ӧʽ��SO32-+8H++6e-=H2S��+3H2O��
���� ��1����Na2SΪǿ�������Σ�S2-ˮ��ʼ��ԣ�
�ڸ���������ԭ��Ӧ���������ͻ�ԭ����ʧ������Ŀ��ȼ��㣻�¶�����ƽ�ⳣ������˵�������¶�ƽ��������Ӧ�ƶ���������Ӧ���ȣ�
��2����H2S����������NaOH��Һ��Ӧ��Ӧ����Na2S��ˮ��
��SO32-�����������·ŵ�����H2S�Ĺ���Ϊ��ԭ��Ӧ�����ݷ�Ӧ�����������д�缫��Ӧʽ��
��� �⣺��1����Na2SΪǿ�������Σ�S2-ˮ��ʼ��ԣ�ˮ�ⷽ��ʽΪS2-+H2O?HS-+OH-���ʴ�Ϊ��S2-+H2O?HS-+OH-��
��1molNa2Sת��Ϊ1molNa2SO4��ʧȥ8mol���ӣ���1molO2����ԭ���õ�4mol���ӣ����Ի�ԭ���������������ʵ���֮��Ϊ1��2���¶�����ƽ�ⳣ������˵�������¶�ƽ��������Ӧ�ƶ���������Ӧ���ȣ�����H��O��
�ʴ�Ϊ��1��2������
��2����H2S����������NaOH��Һ��Ӧ��Ӧ����Na2S��ˮ����Ӧ�Ļ�ѧ����ʽΪH2S+2NaOH=Na2S+2H2O���ʴ�Ϊ��H2S+2NaOH=Na2S+2H2O��
��SO32-�����������·ŵ�����H2S�Ĺ���Ϊ��ԭ��Ӧ���缫��ӦʽΪSO32-+8H++6e-=H2S��+3H2O���ʴ�Ϊ��SO32-+8H++6e-=H2S��+3H2O��
���� ���⿼���Ϊ�ۺϣ��漰�����ˮ�⡢������ԭ��Ӧ�����⣬��Ŀ�Ѷ��еȣ�����ע�����ˮ���ԭ����������ԭ��Ӧ�ļ����Լ��缫��Ӧʽ����д�ȷ����Ե����⣮

| A�� | ���ͼױ� | B�� | ����ϩ�뱽 | ||
| C�� | 1-��ϩ����-���ױ� | D�� | ����NaI��Һ |
�ں��º��ݵ��ܱ���ϵ�ڽ����������淴Ӧ�������йر�����ȷ���ǣ�������

| A�� | ��ͼ�������������Ϊ����Ӧ���ʣ���tʱ�̸ı����������Ϊ���»��ѹ | |
| B�� | ��ͼ�������������Ϊ�ص�ת���� | |
| C�� | ��ͼ�������������Ϊ��ѧ��Ӧ���� | |
| D�� | ��ͼ�������������Ϊ��ϵ�ڻ������ƽ����Է������� |
| A�� | �����������ľ�����˳����LiF��NaF��KF��RbF��CsF�����Ƶ�±����ľ�����˳����NaF��NaCl��NaBr��NaI | |
| B�� | ��A��Ԫ���⻯��е�˳����GeH4��SiH4��CH4�����A��Ԫ���⻯��е�˳����AsH3��PH3��NH3 | |
| C�� | �������������ӣ����������ӣ��������������ӣ����������� | |
| D�� | �������Ũ���Ǵ���Ũ�ȵ�2������������H+Ũ��Ҳ�Ǵ�����H+Ũ�ȵ�2�� |
| ���ܵ���� | Mg��OH��2 | Cu��OH��2 | Fe��OH��2 | Fe��OH��3 |
| �ܽ��/g | 9��10-4 | 1.7��10-6 | 1.5��10-4 | 3.0��10-9 |
| ������ȫʱ��pH | 11.1 | 6.7 | 9.6 | 3.7 |
��Ϊ�˳�ȥ�Ȼ���е�����Fe3+���Ƚ����������ˮ���ټ���һ�������Լ���Ӧ�����˽ᾧ���ɣ���Ϊ�˳�ȥ�Ȼ�þ�����е�����Fe3+���Ƚ����������ˮ������������������þ����ַ�Ӧ�����˽ᾧ���ɣ���Ϊ�˳�ȥ����ͭ�����е�����Fe2+���Ƚ����������ˮ������һ������H2O2����Fe2+������Fe3+��������Һ��pH=4�����˽ᾧ���ɣ���ش��������⣺
��1�������������ӷ������ܹ��ﵽ�ܺ�Ч����Fe2+��Fe3+����ת��ΪFe��OH��3���ѧʽ������ȥ��
��2�����м�����Լ�Ӧ��ѡ��ˮ��
��3�����г�ȥFe3+�����������ӷ���ʽΪ2Fe3++3Mg��OH��2=3Mg2++2Fe��OH��3��
��4�������뷽������ص������У���ȷ����ACDE������ĸ����
A��H2O2����ɫ�������������������в��������ʣ���������Ⱦ
B����Fe2+����ΪFe3+����Ҫԭ����Fe��OH��2������Fe��OH��3�����ѹ���
C��������ҺpH=4��ѡ����Լ���������ͭ���ʽ̼��ͭ
D��Cu2+���Դ���������pH=4����Һ��
E����pH��4����Һ��Fe3+һ�����ܴ������ڣ�
| A�� | 3��2 | B�� | 1��6 | C�� | 2��3 | D�� | 1��1 |
 ���и��������У�����֮��ͨ��һ����Ӧ����ʵ����ͼ��ʾת�����ǣ�������
���и��������У�����֮��ͨ��һ����Ӧ����ʵ����ͼ��ʾת�����ǣ�������| a | b | c | |
| A | N2 | NO | NO2 |
| B | S | SO2 | SO3 |
| C | Si | SiO2 | H2SiO3 |
| D | Fe | FeCl2 | FeCl3 |
| A�� | A | B�� | B | C�� | C | D�� | D |