��Ŀ����
����Ҫ��������и�С��ʵ��Ŀ�ģ���a��b Ϊ���ɼУ��̶�װ�á���β������װ�����ԣ�
��1��Ϊ��֤̼����ǽ����Ե����ǿ������a�ر� b��Ȼ�������������е���Ũ���ᣬװ��A�е��Լ��� ����˵��̼�ķǽ����Աȹ�ǿ��ʵ�������ǣ� ��
��2����֤ SO2�������ԡ���ԭ�Ժ���������������ʣ���b���ر�a��
������֤SO2���������ԵĻ�ѧ����ʽ�ǣ� ��
������֤SO2���л�ԭ�Ե����ӷ���ʽ�ǣ� _��
����˵��SO2����SO2������д�������������������ʵ����ӷ���ʽ�ǣ� ��
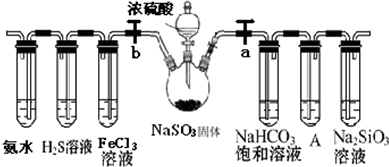
��1��Ϊ��֤̼����ǽ����Ե����ǿ������a�ر� b��Ȼ�������������е���Ũ���ᣬװ��A�е��Լ���
��2����֤ SO2�������ԡ���ԭ�Ժ���������������ʣ���b���ر�a��
������֤SO2���������ԵĻ�ѧ����ʽ�ǣ�
������֤SO2���л�ԭ�Ե����ӷ���ʽ�ǣ�
����˵��SO2����SO2������д�������������������ʵ����ӷ���ʽ�ǣ�

���㣺ʵ��װ���ۺ�
ר�⣺ʵ�������
��������1�����������ǿ�����ԣ���������������A��KMnO4��Һû����ȫ��ɫ��˵�����������Ѿ���ȫ������ʢ��Na2SiO3��Һ���Թ��г��ְ�ɫ������˵�����ɹ��
��2���ٶ�����������Ԫ�صĻ��ϼ���+4�ۣ���������-2�۵���ԭ
�ڶ������������Ȼ�����Ӧ�������
�۶��������백ˮ��Ӧ�����������Σ�
��2���ٶ�����������Ԫ�صĻ��ϼ���+4�ۣ���������-2�۵���ԭ
�ڶ������������Ȼ�����Ӧ�������
�۶��������백ˮ��Ӧ�����������Σ�
���
�⣺��1�����Ը��������Һ��ǿ�����ԣ������������������Կ��������Ը��������Һ�����ն�������A��KMnO4��Һû����ȫ��ɫ��˵�����������Ѿ���ȫ������ʢ��Na2SiO3��Һ���Թ��г��ְ�ɫ������˵��������̼��ˮ������Ʒ�Ӧ���ɹ��ᣬ������̼����ڹ��ᣬ����֤���ǽ�����C����Si��
�ʴ�Ϊ�����Ը��������Һ�����Ը��������Һ����ȫ��ɫ����������Һ���а�ɫ������
��2���ٶ�����������Ԫ�صĻ��ϼ���+4�ۣ��������ԣ���������������H2S��Һ�����·�Ӧ�����ɻ�ɫ�������ʣ���ˮ��
�ʴ�Ϊ��2H2S+SO2=3S��+2H2O��
�ڶ�������ͨ�����Ȼ�����Һ������������������������������Ӻ��������ӣ��䷴Ӧ�����ӷ���ʽΪ��SO2+2Fe3++2H2O�TSO42-+2Fe2++4H+��
�ʴ�Ϊ��SO2+2Fe3++2H2O�TSO42-+2Fe2++4H+��
��������������Ӧ�����κ�ˮ�����������백ˮ��Ӧ�����������Σ��䷴Ӧ�����ӷ���ʽΪ��SO2+2NH3?H20�T2NH4++SO32-+H2O��
�ʴ�Ϊ��SO2+2NH3?H20�T2NH4++SO32-+H2O��
�ʴ�Ϊ�����Ը��������Һ�����Ը��������Һ����ȫ��ɫ����������Һ���а�ɫ������
��2���ٶ�����������Ԫ�صĻ��ϼ���+4�ۣ��������ԣ���������������H2S��Һ�����·�Ӧ�����ɻ�ɫ�������ʣ���ˮ��
�ʴ�Ϊ��2H2S+SO2=3S��+2H2O��
�ڶ�������ͨ�����Ȼ�����Һ������������������������������Ӻ��������ӣ��䷴Ӧ�����ӷ���ʽΪ��SO2+2Fe3++2H2O�TSO42-+2Fe2++4H+��
�ʴ�Ϊ��SO2+2Fe3++2H2O�TSO42-+2Fe2++4H+��
��������������Ӧ�����κ�ˮ�����������백ˮ��Ӧ�����������Σ��䷴Ӧ�����ӷ���ʽΪ��SO2+2NH3?H20�T2NH4++SO32-+H2O��
�ʴ�Ϊ��SO2+2NH3?H20�T2NH4++SO32-+H2O��
���������⿼���˷ǽ����ԱȽϵ�̽��ʵ�鷽����ƣ���ȷ��Ӧԭ���ǽ��Ĺؼ���ע�����֪ʶ�����գ���Ŀ�Ѷ��еȣ������ڿ��������������ʣ�

��ϰ��ϵ�д�
�����Ŀ
��CH4��CO��ɵĻ�����壬�ڱ�״���µ��ܶ�Ϊ1g?L-1������������CH4��CO��������Ϊ��������
| A��1��1 | B��1��2 |
| C��2��3 | D��7��8 |
�����йع�ҵ������������ȷ���ǣ�������
| A���ϳɰ����������н�NH3Һ�����룬�ɼӿ�����Ӧ���ʣ����N2?H2��ת���� |
| B����ҵ�����õ�����ڵ���������ȡ����������ڵ�����þ��ȡþ |
| C����⾫��ͭʱ��ͬһʱ���������ܽ�ͭ����������������ͭ������С |
| D���Ӻ�������ȡ�ⵥ�ʵ�ʵ���У�Ҫ�������ա��ܽ⡢���˺���ȡ�Ȳ��� |
�¶�ΪT��ʱ����2.0L�����ܱ������г���1.0mol PCl5����ӦPCl5��g��?PCl3��g��+Cl2��g����һ��ʱ���ﵽƽ�⣮��Ӧ�����вⶨ�IJ������ݼ��±�������˵����ȷ���ǣ�������
| t/s | 0 | 50 | 150 | 250 | 350 |
| n��PCl3��/mol | 0 | 0.16 | 0.19 | 0.20 | 0.20 |
| A����Ӧ��ǰ50 s��ƽ������Ϊv��PCl3��=0.0032 mol?L-1?s-1 |
| B��T��ʱ���÷�Ӧ��ƽ�ⳣ��Ϊ0.025 |
| C�����������������䣬�����¶ȣ�ƽ��ʱc��PCl3��=0.11 mol?L-1����Ӧ�ġ�H��0 |
| D����ͬ�¶��£����������ٳ���1.0 mol PCl5���ﵽƽ��ʱ��PCl5��ת���ʴ���20% |
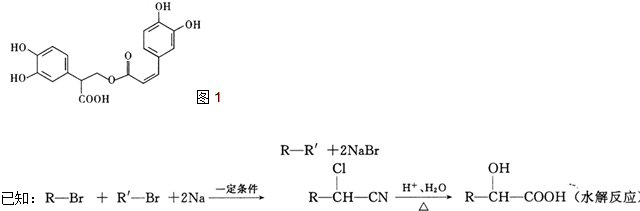
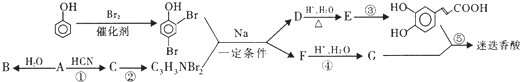
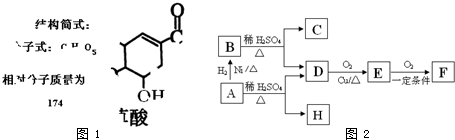
 ������A�����ϣ��������������֪����Է�������Ϊ136������ʽC8H8O2����˴Ź�����������������ͼ���˴Ź����������֮��Ϊ3��2��2��1��
������A�����ϣ��������������֪����Է�������Ϊ136������ʽC8H8O2����˴Ź�����������������ͼ���˴Ź����������֮��Ϊ3��2��2��1��