��Ŀ����
 ��ͼװ����X��Y��Ϊʯī�缫�����ҺΪ500mLij��ɫ��Һ�����һ��ʱ�䣬�۲쵽X�缫�����к�ɫ�Ĺ�̬�������ɣ�Y�缫����ɫ�������ɣ���Һ��ԭ��������ȫ����ֹͣ��⣬ȡ��X�缫��ϴ�ӡ�����������缫����1.6g�������й�˵���в���ȷ���ǣ�������
��ͼװ����X��Y��Ϊʯī�缫�����ҺΪ500mLij��ɫ��Һ�����һ��ʱ�䣬�۲쵽X�缫�����к�ɫ�Ĺ�̬�������ɣ�Y�缫����ɫ�������ɣ���Һ��ԭ��������ȫ����ֹͣ��⣬ȡ��X�缫��ϴ�ӡ�����������缫����1.6g�������й�˵���в���ȷ���ǣ�������| A��X�缫������ |
| B��Y�缫������������Ϊ0.224L |
| C��������Һ��pH=1 |
| D��Ҫʹ������Һ�ָ������ǰ��״̬�������һ������CuO��CuCO3 |
���㣺���ԭ��,ԭ��غ͵��صĹ���ԭ��
ר�⣺�绯ѧר��
������A��������Һ��������������ɫ����֪��������ͭ����Һ��������Y�Ϸų�������������X����������ͭ��
B�����ݵ缫��Ӧʽ��ϵ����غ�����������������
C�����ݵ������õ����������ȷ�������ӵ�����������Ũ�ȵõ���ҺpH��
D�����ݵ���ʸ�ԭ��ԭ����ʲô��ʲô�����ش�
B�����ݵ缫��Ӧʽ��ϵ����غ�����������������
C�����ݵ������õ����������ȷ�������ӵ�����������Ũ�ȵõ���ҺpH��
D�����ݵ���ʸ�ԭ��ԭ����ʲô��ʲô�����ش�
���
�⣺������Һ��������������ɫ����֪��������ͭ����Һ��������Y�Ϸų�������������X����������ͭ��
A���õ����У�X�缫�����к�ɫ�Ĺ�̬�������ɣ���X������������A��ȷ��
B���缫X���������ĵ缫��Ӧ����ʽ��Cu2++2e-�TCu������Y��4OH--4e-�TO2��+2H2O�������ɽ���ͭΪ1.6gʱ�������ɽ���ͭ�����ʵ�����0.025mol��ת�Ƶ�����0.05mol����ʱ�������������ʵ�����0.0125 mol�������0.025mol��22.4L/mol=0.56L����B����
C���������ͭ��Һ���缫X���������ĵ缫��Ӧ����ʽ��Cu2++2e-�TCu������Y��4OH--4e-�TO2��+2H2O�������ɽ���ͭΪ1.6gʱ�������ɽ���ͭ�����ʵ�����0.025mol����ʱ������������ʵ�����0.025mol��������Ũ����
=0.1mol/L����ҺpH=1����C��ȷ��
D�������������ݳ�ʲô��ʲôԭ������������ͭ������������Ҫʹ������Һ�ָ������ǰ��״̬��������ͭ��̼��ͭ���Իָ���ҺŨ�ȣ���D��ȷ��
��ѡB��
A���õ����У�X�缫�����к�ɫ�Ĺ�̬�������ɣ���X������������A��ȷ��
B���缫X���������ĵ缫��Ӧ����ʽ��Cu2++2e-�TCu������Y��4OH--4e-�TO2��+2H2O�������ɽ���ͭΪ1.6gʱ�������ɽ���ͭ�����ʵ�����0.025mol��ת�Ƶ�����0.05mol����ʱ�������������ʵ�����0.0125 mol�������0.025mol��22.4L/mol=0.56L����B����
C���������ͭ��Һ���缫X���������ĵ缫��Ӧ����ʽ��Cu2++2e-�TCu������Y��4OH--4e-�TO2��+2H2O�������ɽ���ͭΪ1.6gʱ�������ɽ���ͭ�����ʵ�����0.025mol����ʱ������������ʵ�����0.025mol��������Ũ����
| 0.025mol��2 |
| 0.5L |
D�������������ݳ�ʲô��ʲôԭ������������ͭ������������Ҫʹ������Һ�ָ������ǰ��״̬��������ͭ��̼��ͭ���Իָ���ҺŨ�ȣ���D��ȷ��
��ѡB��
���������⿼��ѧ�����صĹ���ԭ���Լ�Ӧ��֪ʶ��ע��֪ʶ�Ĺ��ɺ������ǽ���Ĺؼ����ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
������������ѧ��ʹ����ͨ�����Ӻʹ�����ɵ����������������4����ԭ�ӹ��ɵ�������O4������������̽��������ڵ�֤�ݣ�����˵����ȷ���ǣ�������
| A��O4����Ԫ�ص�һ��ͬλ�� |
| B��O4���ܺ��м��Լ� |
| C��ͬ��ͬѹ�£��������O4�����O2������ͬ�ķ����� |
| D��O4ת��O2Ϊ�����仯 |
������NO2ͨ�����и���Һ�У��������ӻ��ܴ���������ǣ�������
| A��Fe2+��Cl-��SO42-��Na+ |
| B��NH4+��Al3+��NO3-��Cl- |
| C��Ca2+��K+��Cl-��HCO3- |
| D��K+��Na+��SO32-��SO42- |
 þȼ�ϵ�ؾ��б������ߡ���ȫ���㡢�ɱ��͵��ص㣬���õ���þ-��������ȼ�ϵ�أ�þ-��������ȼ�ϵ�صȣ�ͼΪþ-��������ȼ�ϵ�صĹ���ԭ��ͼ������˵������ȷ���ǣ�������
þȼ�ϵ�ؾ��б������ߡ���ȫ���㡢�ɱ��͵��ص㣬���õ���þ-��������ȼ�ϵ�أ�þ-��������ȼ�ϵ�صȣ�ͼΪþ-��������ȼ�ϵ�صĹ���ԭ��ͼ������˵������ȷ���ǣ�������| A����ص��ܷ�ӦʽΪMg+ClO-+H2O�TMg��OH��2+Cl- |
| B���ŵ������OH-�������� |
| C��þ�����þ��Ϊ����������������Ӧ |
| D��þ-��������ȼ�ϵ�أ����Ե������������ӦʽΪH2O2+2H++2e-�T2H2O |

 �Ʊ���
�Ʊ��� ��2����ԭ�ӵķ������ĺϳ�·��
��2����ԭ�ӵķ������ĺϳ�·��
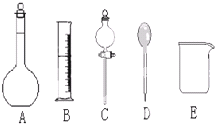 ��У������ȤС���ڴ�����ˮ��Ʒʱ��������������Ϊ37%��Ũ���ᣨ�ܶ�Ϊ1.19g/cm3�����Ƴ�250mL 0.1mol?L-1��������Һ��
��У������ȤС���ڴ�����ˮ��Ʒʱ��������������Ϊ37%��Ũ���ᣨ�ܶ�Ϊ1.19g/cm3�����Ƴ�250mL 0.1mol?L-1��������Һ��