��Ŀ����
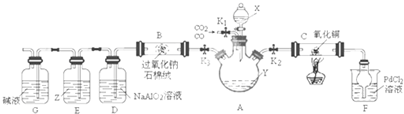
ij�о�С�������ͼװ�÷���CO��CO2�Ļ�����岢̽��CO��ԭ����ͭ�IJ���������Ƶ����ʣ���֪�Ȼ��٣�PdCl2����Һ��CO����������ɫ�������������ʣ����г�װ��δ������
��1���Լ�X��Y����ɫ��Ӧ���ʻ�ɫ����X��Y������ �� ��
��2������װ�á����������Բ�װ��ҩƷ�رշ�Һ©��������K3����K1��K2�������CO�����۲쵽 ����ʱ���ɶ�Cװ�ý��м��Ȳ�������Ӧ����Ϊ��ɫ���壮
��3����ѯ���ϻ��������Ϣ����Cu2OΪ��ɫ���壻�ڳ����£�Cu2+����Һ���ȶ���Cu+�������������·�����Ӧ��2Cu+=Cu2++Cu���ס�������ͬѧȡ������ɫ���壨��M����������ʵ�飬̽����ɷ֣�
��i������ͬѧ��Ϊ����ͬѧ���ý��۲��ɿ����������� ��
��ii������ͬѧ�������ͬѧ�����˼��㣬��Ϊ����ͬѧ�Ľ�������ȷ�ģ����ó�M��Cu��Cu2O�����ʵ���֮���� ��
��4��Cװ�÷�Ӧ���������ӻ�������з����CO2������е���Ҫ������ ��ʵ�������Dװ�ò�����ɫ���������ӷ���ʽΪ ��
��5��Z�Լ�Ϊ����KI�ı���Һ����Eװ���п��ܲ����������� ��

��1���Լ�X��Y����ɫ��Ӧ���ʻ�ɫ����X��Y������
��2������װ�á����������Բ�װ��ҩƷ�رշ�Һ©��������K3����K1��K2�������CO�����۲쵽
��3����ѯ���ϻ��������Ϣ����Cu2OΪ��ɫ���壻�ڳ����£�Cu2+����Һ���ȶ���Cu+�������������·�����Ӧ��2Cu+=Cu2++Cu���ס�������ͬѧȡ������ɫ���壨��M����������ʵ�飬̽����ɷ֣�
| ��� | ʵ����� | ʵ����������� | �� �� | ||
| �� | ����a g M�м���һ����ϡ���ᣬ��ֽ��裻 �ڼ����μ�ϡ��������������ַ�Ӧ�� |
�ٹ������Լ��٣� ����Ȼ��һ�������壬��Һ����ɫ |
��M��һ����Cu2O�� ��M��һ����Cu�� | ||
| �� | �ٽ���ʵ���������Һ���� �ڽ�����ϴ�ӡ�������� |
��������Ϊ
|
MΪCu��Cu2O�Ļ���� |
��ii������ͬѧ�������ͬѧ�����˼��㣬��Ϊ����ͬѧ�Ľ�������ȷ�ģ����ó�M��Cu��Cu2O�����ʵ���֮����
��4��Cװ�÷�Ӧ���������ӻ�������з����CO2������е���Ҫ������
��5��Z�Լ�Ϊ����KI�ı���Һ����Eװ���п��ܲ�����������
���㣺����ʵ�鷽�������
ר�⣺ʵ����
�����������̿�֪����K1��CO��CO2�Ļ������ͨ��A���رշ�Һ©��������K3��Y�����ն�����̼��Ӧ����K1��K2��CO������ͭ����������ԭ��Ӧ������֤CO�Ļ�ԭ�ԣ��Ȼ��٣�PdCl2����Һ��CO����������ɫ��������F�г��ֳ�����˵��CO����װ�ã�Ȼ��ɼ��ȣ�Cװ�÷�Ӧ�����ر�K1��K2����K3�ͷ�Һ©����������X����̼���η�Ӧ�������������̼��D�����ɳ���Ϊ������������B�з���������̼��������Ƶķ�Ӧ��������������Z�е�KI�ɱ��������ɵⵥ�ʣ��ܽ��ڱ���Ϊ�Ϻ�ɫ��G�еļ�Һ�ɷ�ֹ�����Ķ�����̼����װ�ã��Դ������
���
�⣺�����̿�֪����K1��CO��CO2�Ļ������ͨ��A���رշ�Һ©��������K3��Y�����ն�����̼��Ӧ����K1��K2��CO������ͭ����������ԭ��Ӧ������֤CO�Ļ�ԭ�ԣ��Ȼ��٣�PdCl2����Һ��CO����������ɫ��������F�г��ֳ�����˵��CO����װ�ã�Ȼ��ɼ��ȣ�Cװ�÷�Ӧ�����ر�K1��K2����K3�ͷ�Һ©����������X����̼���η�Ӧ�������������̼��D�����ɳ���Ϊ������������B�з���������̼��������Ƶķ�Ӧ��������������Z�е�KI�ɱ��������ɵⵥ�ʣ��ܽ��ڱ���Ϊ�Ϻ�ɫ��G�еļ�Һ�ɷ�ֹ�����Ķ�����̼����װ�ã�
��1���Լ�X��Y����ɫ��Ӧ���ʻ�ɫ������NaԪ�أ�X�����ԣ�Y�Լ��ԣ���XΪNaHSO4��YΪNaOH���ʴ�Ϊ��NaHSO4��NaOH��
��2��CO����ʱ���ȷ�����ը����Fװ���г���������ɫ������CO����װ�ÿɼ���Cװ�ã��ʴ�Ϊ��Fװ���г���������ɫ������
��3����i����ʵ����M����ֻ��Cu2O����Cu2O�����������·���������ԭ��Ӧ������Cu��ͬʱ��������ͭ��Һ����ʵ����۲��ɿ���
�ʴ�Ϊ��Cu2O�����������·���������ԭ��Ӧ������Cu��ͬʱ��������ͭ��Һ��
��ii��agM���壬�����ᷴӦ����������Ϊ
g����Cu��Cu2O�ֱ�Ϊxmol��ymol��
Cu+�������������·�����Ӧ��2Cu+=Cu2++Cu����
�����x��y=1��2���ʴ�Ϊ��1��2��
��4��������������֪�����ӻ�������з����CO2������е���Ҫ�����ǹر�K1��K2����K3�ͷ�Һ©��������Dװ�ò�����ɫ���������ӷ���ʽΪ2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��AlO2-+CO2+2H2O=Al��OH��3��+HCO3-��
�ʴ�Ϊ���ر�K1��K2����K3�ͷ�Һ©��������2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��AlO2-+CO2+2H2O=Al��OH��3��+HCO3-��
��5��Z�е�KI�ɱ��������ɵⵥ�ʣ��ܽ��ڱ���Ϊ�Ϻ�ɫ����Eװ���п��ܲ�������������Һ�ֲ㣬�ϲ�����Ϻ�ɫ���ʴ�Ϊ����Һ�ֲ㣬�ϲ�����Ϻ�ɫ��
��1���Լ�X��Y����ɫ��Ӧ���ʻ�ɫ������NaԪ�أ�X�����ԣ�Y�Լ��ԣ���XΪNaHSO4��YΪNaOH���ʴ�Ϊ��NaHSO4��NaOH��
��2��CO����ʱ���ȷ�����ը����Fװ���г���������ɫ������CO����װ�ÿɼ���Cװ�ã��ʴ�Ϊ��Fװ���г���������ɫ������
��3����i����ʵ����M����ֻ��Cu2O����Cu2O�����������·���������ԭ��Ӧ������Cu��ͬʱ��������ͭ��Һ����ʵ����۲��ɿ���
�ʴ�Ϊ��Cu2O�����������·���������ԭ��Ӧ������Cu��ͬʱ��������ͭ��Һ��
��ii��agM���壬�����ᷴӦ����������Ϊ
| a |
| 2 |
Cu+�������������·�����Ӧ��2Cu+=Cu2++Cu����
|
��4��������������֪�����ӻ�������з����CO2������е���Ҫ�����ǹر�K1��K2����K3�ͷ�Һ©��������Dװ�ò�����ɫ���������ӷ���ʽΪ2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��AlO2-+CO2+2H2O=Al��OH��3��+HCO3-��
�ʴ�Ϊ���ر�K1��K2����K3�ͷ�Һ©��������2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-��AlO2-+CO2+2H2O=Al��OH��3��+HCO3-��
��5��Z�е�KI�ɱ��������ɵⵥ�ʣ��ܽ��ڱ���Ϊ�Ϻ�ɫ����Eװ���п��ܲ�������������Һ�ֲ㣬�ϲ�����Ϻ�ɫ���ʴ�Ϊ����Һ�ֲ㣬�ϲ�����Ϻ�ɫ��
���������⿼������ʵ�鷽������ƣ�Ϊ��Ƶ���㣬��������ͼ��װ�õ����á������Ļ�ѧ��ӦΪ���Ĺؼ���ע��Aʵ�ַ���CO��CO2�Ļ�����壬�����������ʼ����������������������ۺϿ��飬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
���쳣�Ӱ����ֳ���������ζ����ȡ���õ�������������ζ�ijɷ��к��ж����л�����磺
��ʮ����
�ڻ�ʮ������
����������л������˵������ȷ���ǣ�������
��ʮ����
�ڻ�ʮ������
����������л������˵������ȷ���ǣ�������
| A���ٺ͢ڷ�����̼ԭ��������10 |
| B���ٺ͢ڳ��³�ѹ�²�Ϊ��̬ |
| C���ٺ͢ھ�����Na��Ӧ�ų����� |
| D���ڵķ���ʽΪC12H24O |
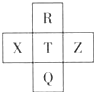 Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ����ͼ��ʾ������R�����ڰ�����H2���һ��ϲ�������ը���������ж���ȷ���ǣ�������
Ԫ��R��X��T��Z��Q��Ԫ�����ڱ��е����λ����ͼ��ʾ������R�����ڰ�����H2���һ��ϲ�������ը���������ж���ȷ���ǣ�������| A���ǽ����ԣ�Z��T��X |
| B��R��Qԭ�Ӻ�����������26 |
| C����̬�⻯���ȶ��ԣ�R��T��Q |
| D������������ˮ��������ԣ�X��T |

 ������AX3�͵���X2��һ�������·�Ӧ�����ɻ�����AX5���ش��������⣻
������AX3�͵���X2��һ�������·�Ӧ�����ɻ�����AX5���ش��������⣻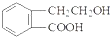 ������ͨ����ͬ��ѧ��Ӧ�ֱ��Ƶ�B��C��D��E�������ʣ�
������ͨ����ͬ��ѧ��Ӧ�ֱ��Ƶ�B��C��D��E�������ʣ�