��Ŀ����
Ϊ��̽����ҵ���ϵ������ã�ij��ѧ��ȤС�����������ʵ�����̣�ͨ�����ַ����ú���Cu��Ag�ĺϽ������ȡAgNO3���壺

��֪2AgNO3
2Ag+2NO2+O��2Cu��NO3��2
2CuO+4NO2+O2
��ش�
��1��д������ I��������Ӧ�����ӷ���ʽ
��2���Լ�x�� ����������ָ ����������ָ
��3������һ���ɹ���A�Ʊ�����Bʱ�����¶ȷ�ΧӦ������ ֮�ڣ�
��4���Լ�y�����Ƶ�Ag2O�������������� ���û�ѧ����ͱ�Ҫ������˵������ԭ�� [��֪Ag2O+H2O?2AgOH����AgOH���ܽ�ȴ���Cu��OH��2]
��5��������һ�Ʊ���AgNO3����Ϊm1�ˣ��������Ʊ���AgNO3����Ϊm2�ˣ���Ͻ���Ag����������Ϊ ���ú���m��m1��m2�Ĵ���ʽ��ʾ����

��֪2AgNO3
| ||
| ||
��ش�
��1��д������ I��������Ӧ�����ӷ���ʽ
��2���Լ�x��
��3������һ���ɹ���A�Ʊ�����Bʱ�����¶ȷ�ΧӦ������
��4���Լ�y�����Ƶ�Ag2O��������������
��5��������һ�Ʊ���AgNO3����Ϊm1�ˣ��������Ʊ���AgNO3����Ϊm2�ˣ���Ͻ���Ag����������Ϊ
���㣺�����Ļ����뻷������Դ����,�Ʊ�ʵ�鷽�������
ר�⣺������Ҫ�Ľ������仯����
��������1��Cu��Ag���ܽ�ϡ�������������Ρ�һ��������ˮ��
��2������B��Һ��B����Ҫ��ˮ�ܽ⣻�������õ��������Һ���ʲ�����Ϊ���ˣ�
��3��Ϊʹ����ͭ�ֽ⣬���������������ֽ⣬Ӧ�����¶���473K��713K��
��4������Ag2Oʱ������ƽ�⣺Ag2O+H2O 2AgOH
2AgOH Ag++OH-��Cu2++2OH-
Ag++OH-��Cu2++2OH- Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2�������Ӷ���ȥ����Cu��
Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2�������Ӷ���ȥ����Cu��
��5���������м�����Ag2O����Ӧ�÷���һ���м��㣮
��2������B��Һ��B����Ҫ��ˮ�ܽ⣻�������õ��������Һ���ʲ�����Ϊ���ˣ�
��3��Ϊʹ����ͭ�ֽ⣬���������������ֽ⣬Ӧ�����¶���473K��713K��
��4������Ag2Oʱ������ƽ�⣺Ag2O+H2O
 2AgOH
2AgOH Ag++OH-��Cu2++2OH-
Ag++OH-��Cu2++2OH- Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2�������Ӷ���ȥ����Cu��
Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2�������Ӷ���ȥ����Cu����5���������м�����Ag2O����Ӧ�÷���һ���м��㣮
���
�⣺��1��Cu��Ag���ܽ�ϡ�������������Ρ�һ��������ˮ���ʴ�Ϊ��3Ag+4H++NO3-=3Ag++NO��+2H2O��3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��
��2������B��Һ��B����Ҫ��ˮ�ܽ⣻�������õ��������Һ���ʲ�����Ϊ���ˣ��������õ�AgNO3���壻�ʴ�Ϊ��H2O�����ˣ�����Ũ������ȴ�ᾧ��
��3��Ϊʹ����ͭ�ֽ⣬���������������ֽ⣬Ӧ�����¶���473K��713K���ʴ�Ϊ��473K��713K��
��4������Ag2Oʱ������ƽ�⣺Ag2O+H2O 2AgOH
2AgOH Ag++OH-��Cu2++2OH-
Ag++OH-��Cu2++2OH- Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2�������Ӷ���ȥ����Cu���ʴ�Ϊ��Ag2O+H2O
Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2�������Ӷ���ȥ����Cu���ʴ�Ϊ��Ag2O+H2O 2AgOH
2AgOH Ag++OH-��Cu2++2OH-
Ag++OH-��Cu2++2OH- Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2������
Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2������
��5���������м�����Ag2O����Ӧ�÷���һ���м��㣻�Ͻ���Ag����������Ϊ
=
���ʴ�Ϊ��
��
��2������B��Һ��B����Ҫ��ˮ�ܽ⣻�������õ��������Һ���ʲ�����Ϊ���ˣ��������õ�AgNO3���壻�ʴ�Ϊ��H2O�����ˣ�����Ũ������ȴ�ᾧ��
��3��Ϊʹ����ͭ�ֽ⣬���������������ֽ⣬Ӧ�����¶���473K��713K���ʴ�Ϊ��473K��713K��
��4������Ag2Oʱ������ƽ�⣺Ag2O+H2O
 2AgOH
2AgOH Ag++OH-��Cu2++2OH-
Ag++OH-��Cu2++2OH- Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2�������Ӷ���ȥ����Cu���ʴ�Ϊ��Ag2O+H2O
Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2�������Ӷ���ȥ����Cu���ʴ�Ϊ��Ag2O+H2O 2AgOH
2AgOH Ag++OH-��Cu2++2OH-
Ag++OH-��Cu2++2OH- Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2������
Cu��OH��2������Cu��OH��2���ܽ��С��AgOH������ƽ�������ƶ���Ag2O�ܽ⣬Cu2+��ת��ΪCu��OH��2��������5���������м�����Ag2O����Ӧ�÷���һ���м��㣻�Ͻ���Ag����������Ϊ
| 108m1 |
| 170m |
| 54m1 |
| 85m |
| 54m1 |
| 85m |
�����������Ƕ����ʵķ�����ᴿ֪ʶ��Ŀ��飬ע��������������ͭ���仯����������ǽ����Ĺؼ�����Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
��NA��ʾ�����ӵ�������������������ȷ���ǣ�������
| A����25�棬1.01��105Paʱ��11.2L����������ԭ����ĿΪNA |
| B��48g O3 ��O2�������������ԭ����ĿΪ3NA |
| C��1mol���������ĵ�����ĿΪ4NA |
| D��2.7g���������������ʱʧȥ�ĵ�����ĿΪ0.1NA |
�����й�ʵ���˵����ȷ���ǣ�������
| A����NH3?H2O��Һ�ܼ���Al3+��Mg2+��Ag+��Cu2+ |
| B������һ�����ʵ���Ũ�ȵ�ϡH2SO4ʱ����Ͳ��ȡŨ��Һʱ�����Ӷ��������ƫ�� |
C����ͼʵ�� ���رյ��ɼУ�������ס��ƿ���������¶�������ð����˵������������ ���رյ��ɼУ�������ס��ƿ���������¶�������ð����˵������������ |
| D���ⶨ�к���ʱ��ȡ25 mL 10 mol/L��Ũ�����25 mL 10 mol/L��NaOH��Һ�кͣ��ⶨ��Ӧ�ų������� |
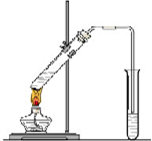 �����dzµ��㡱��������Ϊ���ڴ������������������ζ��������������ʵ��������Ҳ������ͼ��ʾ��װ����ȡ�����������ش���������
�����dzµ��㡱��������Ϊ���ڴ������������������ζ��������������ʵ��������Ҳ������ͼ��ʾ��װ����ȡ�����������ش���������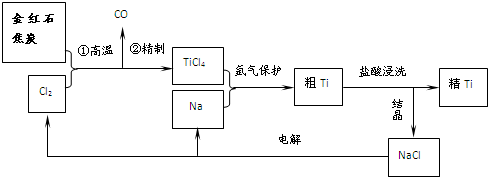

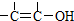 �ṹ�����ʲ����ȶ����ڣ�
�ṹ�����ʲ����ȶ����ڣ�