��Ŀ����
6������Ԫ��W��X��Y��Z��ԭ��������������W��ԭ�������������Ǵ�����������3����X��Y��Z������ͬ�����ڣ����ǵ�ԭ������֮����Wԭ��������5��������Ԫ��W��X��Y��Z��ɵ����п��ܵĶ���ֻ������У���Ԫ��W��Y�γɵĻ�����M���۵���ߣ���ش��������⣺��1��WԪ��ԭ�ӵ�L������Ų�ʽΪ2s22p4��W3���ӵĿռ乹��ΪV�Σ�
��2��X������ˮ������Ҫ��Ӧ�Ļ�ѧ����ʽΪ2F2+2H2O�T4HF+O2��
��3��������M�Ļ�ѧʽΪMgO���侧��ṹ��NaCl��ͬ�����۵����NaCl��M�۵�ϸߵ�ԭ����MgO�ľ����ܴ���NaCl�ľ����ܣ���һ�����Ļ�����ZX������M�Ͽ��Ƶ�ZX/M���������ڴ�̼����������¹��������ϳ�̼����¹�������̼������������У�̼ԭ�Ӳ��õ��ӻ���ʽ��sp2��sp3��O-C-O�ļ���ԼΪ120�㣮
��4��X��Y��Z���γ���������ṹ�Ļ�����侧����Xռ������������ģ�Yλ�ڶ��ǣ�Z��������λ�ã���þ�������ΪX��Y��Z=3��1��1��
��5������Ԫ��Z���ε���ɫ��ӦΪ��ɫ����������ζ����Է�����ɫ��Ӧ����ԭ��������ʱ���Ӵӻ�̬������̬�����Ӽ���̬�ص���̬ʱ�ų������Թ����ʽ�ų����Ӷ����ֲ�ͬ����ɫ��
���� ����Ԫ��W��X��Y��Z��ԭ��������������W��ԭ�������������Ǵ�����������3����Wԭ��ֻ����2�����Ӳ㣬����������Ϊ6����WΪOԪ�أ�X��Y��Z������ͬ�����ڣ����ǵ�ԭ������֮����Wԭ��������5��������Ԫ��ԭ������֮��Ϊ40��X��Ϊ��������Ԫ�أ�X��Y��Z��ԭ������֮�ʹ���Wԭ��������5�������Կ��Զ϶�XҲ�ڵڶ����ڣ���XΪFԪ�أ���Y��Zԭ������֮��=40-9=31��ƽ��ԭ������Ϊ15.5�������ƶ�Y���ڵ������ڡ�Z���ڵ������ڣ�Z��ԭ����������18����YΪNaԪ�أ���ZΪCaԪ�أ���YΪMgԪ�أ���ZΪKԪ�أ�X��ԭ�������������������⣬����Ԫ��W��Y�γɵĻ�����M���۵���ߣ���YΪMgԪ�أ�ZΪKԪ�أ�������MΪMgO���ݴ˽��
��� �⣺����Ԫ��W��X��Y��Z��ԭ��������������W��ԭ�������������Ǵ�����������3����Wԭ��ֻ����2�����Ӳ㣬����������Ϊ6����WΪOԪ�أ�X��Y��Z������ͬ�����ڣ����ǵ�ԭ������֮����Wԭ��������5��������Ԫ��ԭ������֮��Ϊ40��X��Ϊ��������Ԫ�أ�X��Y��Z��ԭ������֮�ʹ���Wԭ��������5�������Կ��Զ϶�XҲ�ڵڶ����ڣ���XΪFԪ�أ���Y��Zԭ������֮��=40-9=31��ƽ��ԭ������Ϊ15.5�������ƶ�Y���ڵ������ڡ�Z���ڵ������ڣ�Z��ԭ����������18����YΪNaԪ�أ���ZΪCaԪ�أ���YΪMgԪ�أ���ZΪKԪ�أ�X��ԭ�������������������⣬����Ԫ��W��Y�γɵĻ�����M���۵���ߣ���YΪMgԪ�أ�ZΪKԪ�أ�������MΪMgO��
��1��WΪ��Ԫ�أ����ݺ�������Ų����ɣ�Oԭ�ӵ�L������Ų�ʽΪ2s22p4��O3������������ԭ�Ӽ۲���Ӷ���=2+$\frac{6-2��2}{2}$=3������1�Թµ��Ӷԣ���O3�ռ乹��ΪV�Σ�
�ʴ�Ϊ��2s22p4��V�Σ�
��2��XΪFԪ�أ���Ӧ�ĵ��ʾ���ǿ�����ԣ�����ˮ��Ӧ����HF����������Ӧ�ķ���ʽΪ��2F2+2H2O�T4HF+O2��
�ʴ�Ϊ��2F2+2H2O�T4HF+O2��
��3��������������֪��MΪMgO���侧��ṹ��NaCl��ͬ�����۵����NaCl������MgO���������ӵĵ�ɶ࣬�����ܴ�MgO�۵�ϸߣ���̼��Ľṹ��֪̼��������ĽṹΪ ��-CH3��Cԭ���γ�4����������ȡsp3�ӻ���������
��-CH3��Cԭ���γ�4����������ȡsp3�ӻ���������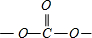 �У�Cԭ���γ�2��C-O���������ڦҼ���1��C=O˫����˫�����������㣬������Cԭ�ӵ��ӻ������Ϊ3����ȡsp2�ӻ���Ϊƽ���������Σ�����Ϊ120�㣬��O-C-O�ļ���ԼΪ120�㣬
�У�Cԭ���γ�2��C-O���������ڦҼ���1��C=O˫����˫�����������㣬������Cԭ�ӵ��ӻ������Ϊ3����ȡsp2�ӻ���Ϊƽ���������Σ�����Ϊ120�㣬��O-C-O�ļ���ԼΪ120�㣬
�ʴ�Ϊ��MgO��MgO�ľ����ܴ���NaCl�ľ����ܣ�sp2��sp3��120�㣻
��4��F��Mg��K�γ���������ṹ�Ļ����������Fռ������������ģ�������Fԭ����ĿΪ12��$\frac{1}{4}$=3��Mgλ�ڶ��ǣ�������Mgԭ����ĿΪ8��$\frac{1}{8}$=1��K��������λ�ã������к���1��Kԭ�ӣ���þ�������ΪF��Mg��K=3��1��1��
�ʴ�Ϊ��3��1��1��
��5������Ԫ��K���ε���ɫ��ӦΪ��ɫ����������ζ����Է�����ɫ��Ӧ����ԭ��Ϊ������ʱ���Ӵӻ�̬������̬�����Ӽ���̬�ص���̬ʱ�ų������Թ����ʽ�ų����Ӷ����ֲ�ͬ����ɫ��
�ʴ�Ϊ���ϣ�����ʱ���Ӵӻ�̬������̬�����Ӽ���̬�ص���̬ʱ�ų������Թ����ʽ�ų����Ӷ����ֲ�ͬ����ɫ��
���� ���⿼�����ʽṹ�����ʣ��漰Ԫ���ƶϡ����ӽṹ�����ʡ�����ṹ�ȣ��Ѷ��еȣ��ƶ�Ԫ���ǽ���Ĺؼ���Ҫ�������ԭ�������Ĺ�ϵ������ڱ��Ľṹ�����ж�Ԫ�أ�

| A�� | ���ʷе�ߵ�˳��Ϊ��W��X��Y��Z | |
| B�� | ԭ�Ӱ뾶��С˳��Ϊ��W��X��Y��Z | |
| C�� | Ԫ�صķǽ�����ǿ��˳��Ϊ��W��X��Y��Z | |
| D�� | ����Ԫ�ؿ��Թ�����һ�����ӻ������� |
| IA | IIA | IIIA | IVA | VA | VIA | VIIA | 0 | |
| 2 | �� | �� | ||||||
| 3 | �� | �� | �� | �� | �� | �� | �� | |
| 4 | �� | ⑪ | ⑫ |
��2������ЩԪ���У�����õĽ���Ԫ����K������õķǽ���Ԫ����F������õ�Ԫ����Ar��
��3������ЩԪ���У�ԭ�Ӱ뾶��С����F��ԭ�Ӱ뾶������K��
��4������ЩԪ�ص�����������Ӧˮ�����У�������ǿ����KOH�������Ե�����������Al��OH��3��
| ���� ���� | IA | IIA | IIIA | IVA | VA | VIA | VIIA | 0 |
| �� | �� | �� | ||||||
| �� | �� | �� | �� | �� | �� | �� | ||
| �� | �� | �� |
��2���ڢۢ�����Ԫ�ص�ԭ�Ӱ뾶�Ӵ�С��˳����Na��Mg��F ����Ԫ�ط��ű�ʾ��
��3���õ���ʽ��ʾ������γɻ�����Ĺ���
 ��
����4���ڢ١���Ԫ���У�Ԫ�ص�����������Ӧ��ˮ������������ǿ����HClO4��������ǿ����NaOH�����ѧʽ��
��5���ڢ����ĵ����У���ѧ���ʽϻ��õ���Cl2���ѧʽ��������ʲô��ѧ��Ӧ˵����д����Ӧ�Ļ�ѧ����ʽ����Cl2+2Br-=2Cl-+Br2��
��Fe2O3��s��+3CO��g��=2Fe��s��+3CO2��g����H=-25kJ•mol-1
��3Fe2O3��s��+CO��g��=2Fe3O4��s��+CO2��g����H=-47kJ•mol-1
�����й���Fe3O4��s����CO��ԭ��Fe��s����CO2���Ȼ�ѧ����ʽ����д����ȷ���ǣ�������
| A�� | Fe3O4+4CO=3Fe+4CO2��H=-14kJ•mol-1 | |
| B�� | Fe3O4��s��+4CO��g��=3Fe��s��+4CO2��g����H=-22kJ•mol-1 | |
| C�� | Fe3O4��s��+4CO��g��=3Fe��s��+4CO2��g����H=+14kJ•mol-1 | |
| D�� | Fe3O4��s��+4CO��g��=3Fe��s��+4CO2��g����H=-14kJ•mol-1 |
 ��
��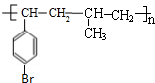 ��
��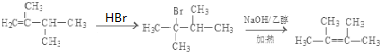 ��
��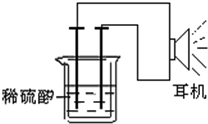 ���������л�ʵ�����г��õ���Ʒ������������ԭ��Ӧ֪ʶ�͵�ѧ֪ʶ���Լ��������һ��ԭ��أ�����д���пհף�
���������л�ʵ�����г��õ���Ʒ������������ԭ��Ӧ֪ʶ�͵�ѧ֪ʶ���Լ��������һ��ԭ��أ�����д���пհף�