��Ŀ����
9��ij��Һ�п��ܺ���Na+��NH4+��Cl-��SO42-��NO3-��CO32-���ֽ�������ʵ�飺��1��ȡһ��������Һ���Թ��У����������Ba��NO3��2��Һ�а�ɫ�������ɣ����˳��ij�����������ϡHNO3�����ų�5.6L������£���ɫ��ζ�����壬ʣ�����4.66g��
��2��ȡ������Һ�����������NaOH��Һ�����ȣ�����13.6g��ɫ�д̼�����ζ�����壬��������ʹʪ��ĺ�ɫʯ����ֽ������
��3����ȡ������Һ������AgNO3��Һ������������
���������������жϣ�
����Һ�п϶����ڵ�������CO32-��SO42-��NH4+��NO3-��
����Һ�п϶������ڵ�������Cl-��
����Һ�п��ܴ��ڵ�������Na+��
��д��ʵ�飨1���г�����������ϡHNO3���ų���ɫ��ζ��������ӷ���ʽ��BaCO3+2H+=Ba2++H2O+CO2��
��ʵ�飨2���в�����������ӷ���ʽΪNH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O��
���� ij��Һ�п��ܺ���Na+��NH4+��Cl-��SO42-��NO3-��CO32-��
��1��ȡһ��������Һ���Թ��У����������Ba��NO3��2��Һ�а�ɫ�������ɣ����˳��ij�����������ϡHNO3��˵��ԭ��Һ�м���SO42-����CO32-�����ᱵ������ϡ���ᣬ̼�ᱵ����ϡ���ᣬ����������̼����5.6L������£������ʵ���Ϊ0.25mol������ԭ���غ㣬ԭ��Һ�е�CO32-�����ʵ���Ϊ0.25mol��ʣ����������ᱵ��������4.66g�����ᱵ��Ħ��������233g/mol���������ʵ���Ϊ0.02mol��ͬ������ԭ���غ��֪ԭ��Һ�е�SO42-�����ʵ���Ϊ0.02mol��
��2��ȡ������Һ�����������NaOH��Һ�����ȣ�����13.6g��ɫ�д̼�����ζ�����壬��������ʹʪ��ĺ�ɫʯ����ֽ�����������������ǰ�����13.6g���������ʵ���Ϊ0.8mol������ԭ���غ��֪ԭ��Һ��NH4+�����ʵ���Ϊ0.8mol��
��3����ȡ������Һ������AgNO3��Һ������������֤��ԭ��Һ���������ӣ�
��� �⣺����������ó�ԭ��Һ��һ���е������ǣ�NH4+��SO42-��CO32-��һ������Cl-����Һ��NH4+��0.8mol�������������������0.8mol��SO42-��0.02mol��CO32-��0.25mol���������������������0.04mol+0.5mol=0.54mol�����ݵ���غ�˵����Һ�б��뻹�����������ӣ�����NO3-һ���У���Na+�����У�
����Һ�п϶����ڵ�������CO32-��SO42-��NH4+��NO3-��
�ʴ�Ϊ��CO32-��SO42-��NH4+��NO3-��
����Һ�п϶������ڵ�������Cl-��
�ʴ�Ϊ��Cl-��
����Һ�п��ܴ��ڵ�������Na+��
�ʴ�Ϊ��Na+��
��ʵ�飨1���г���������̼�ᱵ����ϡHNO3���ų���ɫ��ζ��������ӷ���ʽ��BaCO3+2H+=Ba2++H2O+CO2����
�ʴ�Ϊ��BaCO3+2H+=Ba2++H2O+CO2����
��ʵ�飨2���в�����������ӷ���ʽΪ��NH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O��
�ʴ�Ϊ��NH4++OH-$\frac{\underline{\;\;��\;\;}}{\;}$NH3��+H2O��
���� ���⿼�鳣�����ӵļ��飬������ʵ����ж��⣬���ж���ʵ������ݷ�����ע�����غ�����ã������״��⣮

| A��Ŀ�ģ�Ũ�ȶԻ�ѧ��Ӧ���ʵ�Ӱ�� | B��Ŀ�ģ�����������Һ |
���� 1mol/L 1mL���ᡡ��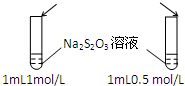 | 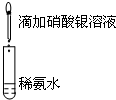 ���� ���� |
| C��Ŀ�ģ��Ƚ�Al��Fe��Cu��� | D��Ŀ�ģ�ʵ������ȡ���� |
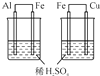 |  |
| A�� | A | B�� | B | C�� | C | D�� | D |
 �ݱ����������ض�����һ������£������˴�����ġ���ȼ����������Ҫ�Ǽ����ˮ�γɵ�ˮ���CH4•nH2O����
�ݱ����������ض�����һ������£������˴�����ġ���ȼ����������Ҫ�Ǽ����ˮ�γɵ�ˮ���CH4•nH2O������1���ڳ��³�ѹ�£�����ȼ�����ᷢ���ֽⷴӦ���仯ѧ����ʽ��CH4•nH2O=CH4��+nH2O��
��2��������Ƴɺϳ�����CO��H2�������Ƴɼ״����������湩Ӧ���ŵ�ȼ�ͣ�
����101KPaʱ��1.6g CH4��g����H2O��g����Ӧ����CO��H2������20.64kJ���������H2O��g����Ӧ���Ȼ�ѧ����ʽ��CH4��g��+H2O��g��=CO��g��+3H2��g����H=+206.4 kJ•mol-1��
��CH4����ȫȼ��Ҳ���Ƶúϳ�����CH4��g��+$\frac{1}{2}$O2��g���TCO��g��+2H2��g����
��H=-35.4kJ•mol-1�����ԭ��ѡ�����Դ���ýǶȣ��ȽϷ����ٺ͢ڣ��ϳɼ״������˷���Ϊ�ڣ�����ţ���ԭ����ѡ��CH4����ȫȼ�գ��ƺϳ�����ʱ���ų�������ͬʱ�õ�CO��H2Ϊ1��2����ǡ����ȫ��Ӧ�ϳɼ״���
��3�����úϳ�������Ҫ�ɷ�ΪCO��CO2��H2���ڴ��������ºϳɼ״�����������Ҫ��Ӧ���£�
��CO��g��+2H2��g��=CH3OH��g����H1
��CO2��g��+3H2��g��=CH3OH��g��+H2O��g����H2
��CO2��g��+H2��g��=CO��g��+H2O��g����H3
�ش��������⣺
��֪��Ӧ������صĻ�ѧ�������������£�
| ��ѧ�� | H-H | C-O | C��O | H-O | C-H |
| E/��kJ•mol-1�� | 436 | 343 | 1 076 | 465 | 413 |
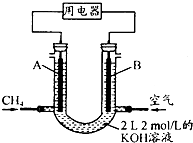
��4����ȼ����CH4��������;�ǣ���CH4��Ƴ�ȼ�ϵ�أ��������ʸ��ߣ�װ��ʾ��ͼ��A��BΪ�����̼����������ͨ�˼��飬�ڱ�״���£����ļ������VL��
��O��V��44.8Lʱ������ܷ�Ӧ����ʽΪCH4+2O2+2KOH=K2CO3+3H2O��
��44.8L��V��89.6Lʱ�������缫��ӦΪCH4-8e-+9CO32-+3H2O=10HCO3-��
| A�� | K | B�� | Ca | C�� | I | D�� | Ne |

| A�� | ȡ����Ӧ | B�� | �ӳɷ�Ӧ | C�� | �ۺϷ�Ӧ | D�� | �Ӿ۷�Ӧ |
| A�� | ����Һ������˿������������ | B�� | ��ˮ�������ԣ���ʹ��̪��Һ��� | ||
| C�� | ����������ˮ����˰�ˮ�Ƚ��ȶ� | D�� | ����������ˮ����˿�����Ȫʵ�� |
| A�� | NaOH��aq��+$\frac{1}{2}$H2SO4��aq���T$\frac{1}{2}$Na2SO4��aq��+H2O��l������H1 | |
| B�� | NaOH��aq��+$\frac{1}{2}$H2SO4��Ũ���T$\frac{1}{2}$Na2SO4��aq��+H2O��l������H4 | |
| C�� | NaOH��aq��+HCl��aq���TNaCl��aq ��+H2O��l������H2 | |
| D�� | CH3COOH��aq��+NaOH��aq���TCH3COONa ��aq ��+H2O��l������H3 |
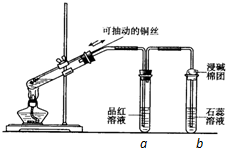 ijͬѧ���ʵ��֤��ͭ��Ũ�����ܷ�����Ӧ��������������������ʣ���ͼ��ʾ�����Թ������2mLŨ���ᣬ�ô����ܺ�һ��С�Ľ����������ӿ��в���һ��ͭ˿�����ȣ��ѷų�����������ͨ��Ʒ����Һ��ʯ����Һ�У���ش��������⣺
ijͬѧ���ʵ��֤��ͭ��Ũ�����ܷ�����Ӧ��������������������ʣ���ͼ��ʾ�����Թ������2mLŨ���ᣬ�ô����ܺ�һ��С�Ľ����������ӿ��в���һ��ͭ˿�����ȣ��ѷų�����������ͨ��Ʒ����Һ��ʯ����Һ�У���ش��������⣺