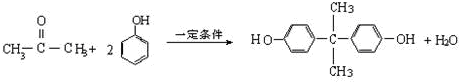
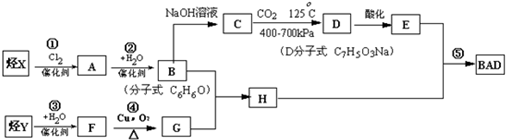
��Ŀ����
���÷�����X������Y���Ժϳ����������ռ�BAD����֪G���ܷ���������Ӧ��BAD
�ṹ��ʽΪ��
BAD�ĺϳ�·�����£�
�Իش�
(1) д���ṹ��ʽY________________________ D____________________��
(2) ����ȡ����Ӧ����_____________________________����������ţ� ��
(3) 1molBAD�����뺬____molNaOH����Һ��ȫ��Ӧ
(4) д����Ӧ�ܷ���ʽ________________________________________________
�� д��B+G��H����ʽ________________________________________________��
(6) E��ͬ���칹�����������ʣ�����FeCl3��Ӧ����Һ����ɫ�������ܷ���������Ӧ�����ͬ���칹�干�� ��
��15�֣�
(1)CH3CH=CH2��2�֣� ��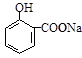 ��2�֣� (2) �٢ڢ� ��2�֣� (3) 6��2�֣�
��2�֣� (2) �٢ڢ� ��2�֣� (3) 6��2�֣�
(4) 2 ![]() + O2
+ O2  2
2 ![]() +2H2O��2�֣�
+2H2O��2�֣�
(5)2![]() +
+ ![]() ��
�� 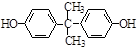 + H2O��2�֣�
+ H2O��2�֣�
��6���������֣�
����:

��ϰ��ϵ�д�
 ��ǰ����ϵ�д�
��ǰ����ϵ�д�
�����Ŀ