��Ŀ����
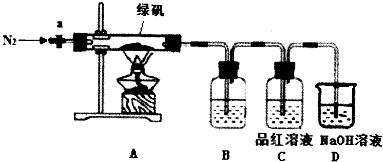
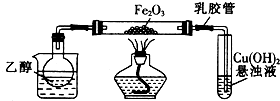
1��ˮ���£�N2H4•H2O������ˮ������������ɫ�������и�ʴ�Ժ�ǿ��ԭ�Եļ���Һ�壬����һ����Ҫ�Ļ����Լ����������ط�����ˮ���µ�ԭ��Ϊ��CO��NH2��2+2NaOH+NaClO=Na2CO3+N2H4•H2O+NaClʵ��һ���Ʊ�NaClO��Һ����ʵ��װ����ͼ1��ʾ��
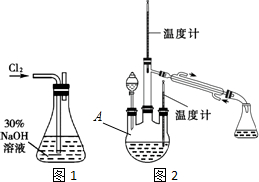
��1������30%NaOH��Һʱ�����貣����������Ͳ���BD�����ţ���
A������ƿ B���ձ� C����ƿ D�������� E����ͷ�ι�
��2����ƿ�з�����Ӧ�����ӷ���ʽ��Cl2+2OH-=ClO-+Cl-+H2O��
��3�����ʵ�鷽�������к͵ζ�ԭ���ⶨ��Ӧ����ƿ��ʣ��NaOH��Ũ�ȣ�ʵ���ṩ���Լ���H2O2��Һ��FeCl2��Һ��0.10mol•L-1���ᡢ��̪��Һ����ȡһ������ƿ�ڻ����Һ������������H2O2��Һ�μ�2��3�η�̪��Һ���� 0.10mol•L-1����ζ����ظ���������2��3�Σ����������������̵�ϸ�ڣ���
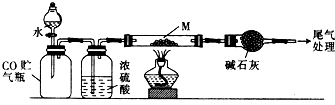
ʵ�������ȡˮ���£���ʵ��װ����ͼ2��ʾ��
��4��װ��A������Ϊ������ƿ���ٷ�Ӧ����������Ʒ�Ӧ�¶ȣ�ͬʱ����Һ©������Һ��������A�У�������ٹ�����ᵼ�²�Ʒ���ʽ��ͣ�ͬʱ��������������д���ù��̵Ļ�ѧ��Ӧ����ʽ��N2H4•H2O+2NaClO=N2��+3H2O+2NaCl���ʷ�Һ©���е���Һ��B�����ţ���
A��CO��NH2��2��Һ B��NaOH��NaClO
�ڳ�ַ�Ӧ��������A�ڵ���Һ���ռ�108��114����֣����ɵõ�ˮ���µĴֲ�Ʒ��
ʵ�������ⶨ������º�����
��5����ȡ���5.0g����������NaHCO3���壬��ˮ���250mL��Һ���Ƴ�25.00mL������ƿ�У����μ�2��3�ε�����Һ����0.10mo1•L-1��I2��Һ�ζ����ζ������У���Һ��pH������6.5���ң�����֪��N2H4•H2O+2I2=N2��+4HI+H2O��
�ٵζ�ʱ����ı���Һʢ������ʽ�����ʽ����ʽ�����ζ����У���ʵ��ζ��յ������Ϊ��Һ������ɫ�Ұ�����ڲ���ʧ��
��ʵ��������I2��Һ��ƽ��ֵΪ18.00mL�������ˮ���£�N2H4•H2O������������Ϊ9%��
���� ��1����������һ��������������Һ�IJ���ѡ��������
��2����ƿ���������������Ʒ�Ӧ�����Ȼ��ơ�����������ˮ��
��3���ù��������ȥ��Һ��NaClO���÷�̪��ָʾ����������ζ����ظ��ⶨ��������
��4������A�Ĺ����ж������ƣ�N2H4•H2O����ǿ��ԭ�ԣ����ױ�������NaClO������
��5���ٵ�ı���Һ���������ԣ����Ը�ʴ��Ƥ�ܣ�Ӧʢ������ʽ�ζ����У��·�Ӧ��ȫ���ٵ����ı���Һ�������۱���ɫ��
�ڸ��ݷ���ʽN2H4•H2O+2I2=N2��+4HI+H2O���㣮
��� �⣺��1������30%NaOH��Һʱ������ƽ�������������ձ��м�ˮ�ܽ⣬���ò��������裬��Ҫ���������У��ձ�����������
�ʴ�Ϊ��BD��
��2����ƿ���������������Ʒ�Ӧ�����Ȼ��ơ�����������ˮ����Ӧ���ӷ���ʽΪ��Cl2+2OH-=ClO-+Cl-+H2O��
�ʴ�Ϊ��Cl2+2OH-=ClO-+Cl-+H2O��
��3���ù��������ȥ��Һ��NaClO���÷�̪��ָʾ�������к͵ζ�ԭ���ⶨ��Ӧ����ƿ��ʣ��NaOH��Ũ�ȣ�ȡһ������ƿ�ڻ����Һ������������H2O2��Һ�μ�2��3�η�̪��Һ���� 0.10mol•L-1����ζ����ظ���������2��3�Σ�
�ʴ�Ϊ��ȡһ������ƿ�ڻ����Һ������������H2O2��Һ�μ�2��3�η�̪��Һ���� 0.10mol•L-1����ζ����ظ���������2��3�Σ�
��4������ͼʾ��֪������AΪ������ƿ��N2H4•H2O����ǿ��ԭ�ԣ����ױ�������NaClO�������ᷢ����Ӧ��N2H4•H2O+2NaClO=N2��+3H2O+2NaCl��Ӧ��NaOH��NaClO�����Һ����CO��NH2��2��Һ�У��ʷ�Һ©����ʢ�ŵ�ΪNaOH��NaClO�����Һ��
�ʴ�Ϊ��������ƿ��N2H4•H2O+2NaClO=N2��+3H2O+2NaCl��B��
��5���ٵ�ı���Һ���������ԣ����Ը�ʴ��Ƥ�ܣ�Ӧʢ������ʽ�ζ����У��·�Ӧ��ȫ���ٵ����ı���Һ�������۱���ɫ��ʵ��ζ��յ������Ϊ����Һ������ɫ�Ұ�����ڲ���ʧ��
�ʴ�Ϊ����ʽ����Һ������ɫ�Ұ�����ڲ���ʧ��
���������ˮ���£�N2H4•H2O������������Ϊa����
N2H4•H2O+2I2=N2��+4HI+H2O
50g 2mol
5g��a 0.018L��0.1mol/L��$\frac{250mL}{25mL}$����50g��5g��a=2mol��0.018L��0.1mol/L��$\frac{250mL}{25mL}$����ã�a=9%��
�ʴ�Ϊ��9%��
���� ���⿼�������Ʊ�ʵ�顢���ʺ����ⶨ����Ŀ�Ѷ��еȣ���ȷʵ��Ŀ��Ϊ���ؼ���ע��������Ŀ��Ϣ�ж���Һ����˳�ϺõĿ���ѧ����ʵ��ԭ�������⡢֪ʶǨ��Ӧ�ã�

 ��ĩ1�����ʽ���������ϵ�д�
��ĩ1�����ʽ���������ϵ�д�| A�� | ��Ũ������Cu��Ӧ��NO2 | B�� | ʵ������NH4Cl��Ca��OH��2�Ʊ�NH3 | ||
| C�� | NO2����ˮʱ����������ԭ��Ӧ | D�� | ������ͭ��Ũ���ᷴӦ��N0���� |
 �����������壨FeC2O4•2H2O���ʵ���ɫ��ij������Ϊ̽��������������Ļ�ѧ���ʣ�������һϵ��ʵ��̽����
�����������壨FeC2O4•2H2O���ʵ���ɫ��ij������Ϊ̽��������������Ļ�ѧ���ʣ�������һϵ��ʵ��̽������1����ʢ�в�������������Թ��е��뼸�������ữ��KMnO4��Һ����������Һ��ɫ��Ϊ�ػ�ɫ�������ж�����̼�������ɣ���˵����������������л�ԭ�ԣ�������ԡ�������ԭ�ԡ����ԡ���������Ӧ������1mol FeC2O4•2H2O����μӷ�Ӧ��KMnO4Ϊ0.6mol��
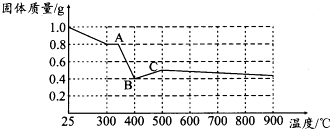
��2�����ϱ��������ܱ������м��ȵ�һ���¶�ʱ�����������������ȫ�ֽ⣬���ɼ��������������Ϊ��ɫ���壮��������ݿα��������ܵ���������������ʣ��Ժ�ɫ��������������¼��裬������ɼ�����ͼ�������
����һ��ȫ����FeO��
�������ȫ����Fe3O4��
����������FeO��Fe3O4�Ļ���
��3��Ϊ��֤��������һ�Ƿ��������������������о���
�������о���������ɱ������ݣ�
| ʵ�鲽�裨��Ҫ��д������������̣� | Ԥ��ʵ������ͽ��� |
| ȡ������ɫ���壬��������ϡ�����ϡ���ᣬ ����Һ�м������軯����Һ | ��Һ�����ɫ�������һ������ ��Һ��ɺ�ɫ�������һ������ |
| A�� | ���ʵķе㣺e��d | |
| B�� | ԭ�Ӱ뾶��r��e����r��d�� | |
| C�� | ����������Ӧˮ��������ԣ�c��d | |
| D�� | a��b�����γ����ӻ����� |
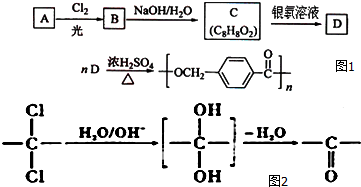
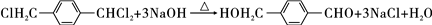 ���÷�Ӧ���������ɵIJ��ȶ��м���Ľṹ��ʽӦ��
���÷�Ӧ���������ɵIJ��ȶ��м���Ľṹ��ʽӦ��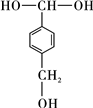 ��
��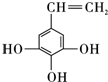 ��
��
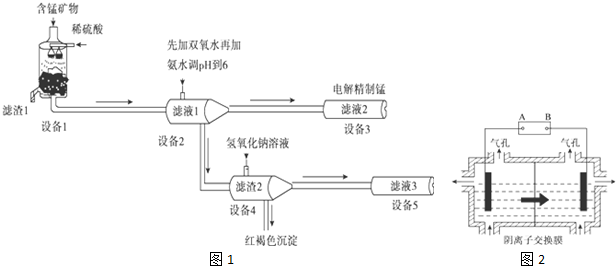
 ��Dԭ�ӵļ۲�����Ų�ʽΪ3s23p3��
��Dԭ�ӵļ۲�����Ų�ʽΪ3s23p3��