��Ŀ����
20��Ϊ��֤������Cl2��Fe3+��SO2��ijС������ͼ��ʾװ�ý���ʵ�飨�г�������A�м���װ�����ԣ��������Ѽ��飩��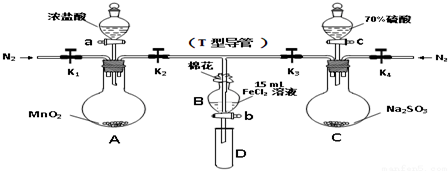
ʵ����̣�
���ɼ�K1��K4��ͨ��һ��ʱ��N2���ٽ�T�͵��ܲ���B�У�����ͨ��N2��Ȼ��ر�K1��K3��K4��
����a���μ�һ������Ũ���ᣬ����A��
��B����Һ���ʱ��ֹͣ���ȣ��н�K2��
��������b��ʹԼ2mL����Һ����D�Թ��У����������е����ӣ�
������K3�ͻ���c������70%�����ᣬһ��ʱ���н�K3��
���������Թ�D���ظ����̢�������B��Һ�е����ӣ���
��1�����̢��Ŀ�����ų�װ���еĿ�������ֹ���ţ�
��2������ڢ�B�еĻ�ɫ��Һ��ͨ��H2S���壬��۲쵽�е���ɫ�������ɣ�д���÷�Ӧ�����ӷ���ʽ2Fe3++H2S�T2Fe2++2H++S��
��3��������ȡ��SO2ͨ�������ữ�ĸ��������Һ��ʹ��Һ��ɫ���䷴Ӧ�Ļ�ѧ����ʽΪ2KMnO4+5SO2+2H2O=2MnSO4+K2SO4+2H2SO4
��4���ס��ҡ�����λͬѧ�ֱ����������ʵ�飬�������±���ʾ�����ǵļ����һ���ܹ�֤��������Cl2��Fe3+��SO2���Ǽף���ס����ҡ���������
| ���̢�B��Һ�к��е����� | ���̢�B��Һ�к��е����� | |
| �� | ��Fe3+��Fe2+ | ��SO42- |
| �� | ����Fe3+����Fe2+ | ��SO42- |
| �� | ��Fe3+��Fe2+ | ��Fe2+ |
���� ʵ����̣�
���ɼ�K1��K4��ͨ��һ��ʱ��N2���ٽ�T�͵��ܲ���B�У�����ͨ��N2��Ȼ��ر�K1��K3��K4��
����a���μ�һ������Ũ���ᣬ����A��
��B����Һ���ʱ��ֹͣ���ȣ��н�K2��
��������b��ʹԼ2mL����Һ����D�Թ��У����������е����ӣ�
������K3�ͻ���c������70%�����ᣬһ��ʱ���н�K3��
���������Թ�D���ظ����̢�������B��Һ�е����ӣ���
��1��ͨ��һ��ʱ��N2���ų�װ���е�������
��2���ڢ�B�з������������������ⷴӦ���������������������ˮ��
��3��SO2ͨ�����Ը��������Һ�ж��߿��Է���������ԭ��Ӧ��
��4�����е�һ�Σ�˵���������㣬���������Դ��������ӣ��ڶ�������������ӣ�˵���������������������ӵķ�Ӧ���������������Ӵ��ڶ��������е�һ����Fe3+����Fe2+���������������Դ��������ӣ��ڶ������������ӣ�˵���������������������ӵķ�Ӧ���������������Ӵ��ڶ�������
��5�����ݵ����غ���м��㣮
��� �⣺��1����K1��K4���ر�K5��K6��ͨ��һ��ʱ��N2��Ŀ�����ų�װ���е�������
�ʴ�Ϊ���ų�װ���еĿ�������ֹ���ţ�
��2�����������������ⷴӦ���������������������ˮ�����ӷ���ʽΪ��2Fe3++H2S�T2Fe2++2H++S����
�ʴ�Ϊ��2Fe3++H2S�T2Fe2++2H++S����
��3��SO2���л�ԭ�ԣ����Ը��������Һ����ǿ�������ԣ�����Ϻ���Է���������ԭ��Ӧ��2KMnO4+5SO2+2H2O=2MnSO4+K2SO4+2H2SO4��ʹ���������ɫ��
�ʴ�Ϊ��2KMnO4+5SO2+2H2O=2MnSO4+K2SO4+2H2SO4��
��4�������ֻ��֤��������Cl2 ��Fe3+������֤��Fe3+��SO2����ΪCl2Ҳ������SO2����SO42-�����е�һ�Σ�˵���������㣬���������Դ��������ӣ��ڶ�������������ӣ�˵���������������������ӵķ�Ӧ���������������Ӵ��ڶ��������е�һ����Fe3+����Fe2+���������������Դ��������ӣ��ڶ������������ӣ�˵���������������������ӵķ�Ӧ���������������Ӵ��ڶ������������ֻ��֤��������Cl2 ��Fe3+������֤��Fe3+��SO2����ΪCl2Ҳ������SO2����SO42-�������һ�����ܹ�֤��������Cl2��Fe3+��SO2���Ǽף�
�ʴ�Ϊ���ף�
��5����FeBr2�����ʵ���Ũ��Ϊc���ɵ����غ��֪��$\frac{1.12L}{22.4L/mol}$��2��1=c��0.1L����3-2��+c��0.1L��2��$\frac{1}{2}$����1-0�������c=0.5mol/L��
�ʴ�Ϊ��0.5��
���� ���⿼������ʵ�鷽������ƣ��漰��������ԭ��Ӧ���жϡ���ѧʵ����������������������Ʊ����������Ȼ�����SO2���ʵ�̽���Լ����������ƽ��״̬��Ӱ���֪ʶ����Ŀ�Ѷ��еȣ���ȷװ�õ����ü������ķ�Ӧ�ǽ��Ĺؼ���ע�������ԵıȽ��ǽ����ѵ㣮

| A�� | ������������������Һ������ˮ | B�� | �����ȷ�ұ�������� | ||
| C�� | ����֬�Ʒ��� | D�� | ��ˮ���� |
| A�� | �ĸ�C-H����ȫ��ͬ | B�� | CH4��һ�ȴ���ֻ��һ�� | ||
| C�� | CH2Cl2ֻ��һ�ֿռ�ṹ | D�� | �ڳ��³�ѹ��CH2Cl2ΪҺ�� |
| A�� | ������Һ���¶ȼ�Ҫһֱ������Һ�� | |
| B�� | Ӧ�����ᣨ��������μӼ�ᣩ | |
| C�� | ��Ӧǰ��Ҫ���¶ȼƲ����ᣨ��¶� | |
| D�� | ��Ӧ������Ӧ�ʵ����� |
| ѡ�� | Ŀ�� | ���뷽�� | ԭ�� |
| A | ʹ��CCl4��ȡ��ˮ�еĵ� | ��ȡ����Һ | ����CCl4�е��ܽ�Ƚϴ� |
| B | ���������������Ҵ� | ��Һ | �����������Ҵ����ܶȲ�ͬ |
| C | ��ȥ���۽����е�NaCl | ���� | ���۽��岻������ֽ |
| D | �����Ȼ��ƺ��Ȼ�� | ���� | �Ȼ�������� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | $\frac{1}{9a}$ mol-1 | B�� | 9a mol-1 | C�� | 2a mol-1 | D�� | 18a mol-1 |
| A�� | 14.2g | B�� | 7.3g | C�� | 3.65g | D�� | 7.1g |
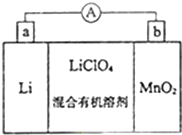 ��̵�ص����С�������������dz��õ�һ�ε�أ��õ�ط�Ӧԭ����ͼ��ʾ�����е����LiClO4���ڻ���л��ܼ��У�Li+ ͨ�������Ǩ����MnO2�����У�����LiMnO2���ش��������⣺
��̵�ص����С�������������dz��õ�һ�ε�أ��õ�ط�Ӧԭ����ͼ��ʾ�����е����LiClO4���ڻ���л��ܼ��У�Li+ ͨ�������Ǩ����MnO2�����У�����LiMnO2���ش��������⣺