��Ŀ����
7����������98%��ŨH2SO4����=1.84g/cm3�����Ƴ�0.5mol/L��ϡH2SO4 500mL�IJ������밴Ҫ����գ���1������ŨH2SO4�����Ϊ13.6ml��
��2�����ʵ������15mL��20mL��50mL��Ͳ��Ӧѡ��15mL��Ͳ��ã�
��3������ȡ��ŨH2SO4���ձ��ڱ�����ע��ʢ��Լ100mLˮ���ձ�������Ͻ��裬Ŀ���Ƿ�ֹŨ����ɽ���
��4������ȴ���������Һ�ز�����ע��500mL����ƿ�У�����50mL����ˮϴ���ձ�2��3�Σ�ϴ��ҺҲҪע������ƿ�У���ҡ�ȣ���δϴ���ձ�����������Һ��Ũ�Ȼ�ƫ�ͣ���ƫ�ߡ�ƫ�ͻ���Ӱ�죩��
��5����ˮ��Һ���̶�����1-2cmʱ�����ý�ͷ�ιܼ�ˮ��ʹ��Һ�İ�Һ�����ø��̶���ƽ��
���� ��1��ŨH2SO4�����ʵ���Ũ��c=$\frac{1000��w}{M}$���ٸ�����Һϡ��ǰ�����ʵ��������������Ũ����������
��2�����ݴ������ԭ��ѡ����ʹ����Ͳ��
��3������Ũ����ϡ�͵ķ���������
��4��������Һ����ȷ������𣻷������������ʵ����ʵ�������Һ�����Ӱ�죬����c=$\frac{n}{V}$������������
��5�����ݶ��ݵ���ȷ�������
��� �⣺��1��ŨH2SO4�����ʵ���Ũ��c=$\frac{1000��1.84��98%}{98}$mol/L=18.4mol/L��
����ϡ�Ͷ��ɣ�ϡ��ǰ�����ʵ����ʵ������䣬������Ũ������������Ũ��������ΪxmL������xmL��18.4mol/L=500mL��0.5mol/L��
��ã�x��13.6������Ӧ��ȡ��Ũ���������13.6mL��
�ʴ�Ϊ��13.6��
��2����ȡ13.6mLŨ���ᣬӦѡ��15mL��Ͳ��
�ʴ�Ϊ��15��
��3��Ũ����ϡ�͵ķ���Ϊ��ϡ��Ũ����ʱ��Ҫ��Ũ���Ỻ����������ע��ʢ��ˮ���ձ��У�ͬʱ�ò��������Ͻ��裬��ʹ������ʱ����ɢ����ֹŨ����ɽ����в��ɰ�ˮע��Ũ�����У�
�ʴ�Ϊ���ձ�����ֹŨ����ɽ���
��4����Һʱ������ȴ�����Һ���Ų�����ע��500mL����ƿ�У�����50mL����ˮϴ���ձ�2��3�Σ�ϴ��ҺҪת�Ƶ�����ƿ�У�ҡ�ȣ���δϴ���ձ������²���������ģ����ʵ����ʵ���ƫС����������Һ��Ũ�Ȼ�ƫ�ͣ�
�ʴ�Ϊ����������500mL����ƿ��ƫ�ͣ�
��5��������ȷ����Ϊ������������ƿ�м�������ˮ��Һ���̶�����1-2cm�����ý�ͷ�ιܼ�ˮ��ʹ��Һ�İ�Һ�����ø��̶���ƽ��
�ʴ�Ϊ��1-2cm��
���� ���⿼����һ�����ʵ���Ũ����Һ�����Ƶ�����������ȷ����ԭ�������������ǽ���ؼ���ע���c=$\frac{n}{V}$�������������ʵ����ʵ��������Һ�������Ӱ���жϣ�

| A�� | ���������ƹ��������ֽ�ϳ��� | |
| B�� | ��ȡ����ʱ��������������Ũ�����ǿ�ȣ��������������ռ����� | |
| C�� | ����0.1mol/L H2SO4��Һʱ������ȡ��ŨH2SO4��������ƿ��ϡ�� | |
| D�� | ����ɫ��Ӧʱ��ÿ��ʵ��ǰһ��Ҫ��ϡ����ϴ����˿�����ڻ��������յ�û����ɫ |
| A�� |  �Ʊ�Cl2 | B�� | 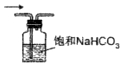 ȥ������ | C�� |  ����Cl2 | D�� |  ���չ���MnO2 |
| A�� | 1 mol ��ϩ�����к��е�̼�����Ϊ4NA | |
| B�� | 1 mol ������ȫȼ��ת�Ƶĵ�����Ϊ8NA | |
| C�� | 1 L 0.1 mol•L-1��������Һ�к�H+������Ϊ0.1NA | |
| D�� | 1 mol ��CO��N2��������к��е�������Ϊ14NA |
| A�� | ���³�ѹ�£�4 g H2���еķ�����Ϊ 2NA | |
| B�� | 8 g H2O�ڱ�״���µ������22.4 L | |
| C�� | 2.3 g�����Ʊ�Ϊ������ʱʧȥ�ĵ�����ΪNA | |
| D�� | ���ʵ���Ũ��Ϊ0.5 mol/L��MgCl2��Һ�У�����Cl-����ΪNA |
 �-ͭ����ȼ�ϵ�������ߡ��ɱ��ͣ����й����ķ�չǰ�����õ��ͨ��һ�ָ��ӵ�ͭ��ʴ�����������������зŵ����Ϊ2Li+Cu2O+H2O�T2Cu+2Li++2OH-������˵������ȷ���ǣ�������
�-ͭ����ȼ�ϵ�������ߡ��ɱ��ͣ����й����ķ�չǰ�����õ��ͨ��һ�ָ��ӵ�ͭ��ʴ�����������������зŵ����Ϊ2Li+Cu2O+H2O�T2Cu+2Li++2OH-������˵������ȷ���ǣ�������| A�� | �ŵ�ʱ�������ĵ缫��ӦʽΪCu2O+H2O+2e-=2Cu+2OH- | |
| B�� | �ŵ�ʱ������������������Li���ƶ� | |
| C�� | ͨ����ʱ��ͭ����ʴ���������Cu2O | |
| D�� | ������Ӧ�����У�������ΪO2 |
| A�� | X��Y��Z һ��λ��ͬһ���� | |
| B�� | X��Y�γɵĻ������и�ԭ�Ӳ�һ���ﵽ8�����ȶ��ṹ | |
| C�� | Y��Z������������Ӧˮ����֮��һ�������Ӧ | |
| D�� | W��X��Y�γɵĻ�������ȶ���һ����ǰ�ߴ��ں��� |
| A�� | 2��3-���������H-NMR����3��� | |
| B�� | ����������ڹ����µķ�Ӧ����ϩ��������Ȼ�̼��Һ�ķ�Ӧ����ͬһ��Ӧ���� | |
| C�� |  ��ͬ���칹���У������ϵ�һ�ȴ���ֻ��һ�ֵĽṹ��2�� ��ͬ���칹���У������ϵ�һ�ȴ���ֻ��һ�ֵĽṹ��2�� | |
| D�� | ��֬��ʳ���в���������ߵ�Ӫ������ |