��Ŀ����
��ij�㶨�¶��£����ݻ�Ϊ1L��������Ͷ��1mol CO��2mol H2O���������·�Ӧ��CO��g��+H2O��g��?CO2��g��+H2��g����ƽ��ʱ����CO2
mol���������¶Ⱥ��ݻ����䣬����������2mol H2O��g����ʹ��Ӧ�����µ�ƽ�⣬����˵������ȷ���ǣ�������
| 2 |
| 3 |
| A���¡���ƽ��ʱ�����������ѹǿ֮����5��3 |
| B����ƽ��ʱH2O��ת����Ϊ20% |
| C����ƽ��ʱCO��Ũ����0.2 mol?L-1 |
| D���¡���ƽ��ʱ�����������ܶ�֮��Ϊ5��3 |
���㣺��ѧƽ��ļ���
ר�⣺
�������������⣺CO+H2O��g��?CO2 +H2��
��ʼ���ʵ�����1 2 0 0
�仯���ʵ�����
ƽ�����ʵ�����
ƽ�ⳣ��K=
=1
��ͨ��2molˮ����ʱ���������ɶ�����̼xmol����
CO+H2O��g��?CO2 +H2��
��ʼ���ʵ�����
+2
�仯���ʵ�����x x x x
ƽ�����ʵ�����
-x
+2-x
+x
+x
���Ը����¶�û�䣬��K=1=
�����x=
mol��
�ݴ˷����жϣ�
��ʼ���ʵ�����1 2 0 0
�仯���ʵ�����
| 2 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
ƽ�����ʵ�����
| 1 |
| 3 |
| 4 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
ƽ�ⳣ��K=
| ||||
|
��ͨ��2molˮ����ʱ���������ɶ�����̼xmol����
CO+H2O��g��?CO2 +H2��
��ʼ���ʵ�����
| 1 |
| 3 |
| 4 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
�仯���ʵ�����x x x x
ƽ�����ʵ�����
| 1 |
| 3 |
| 4 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
���Ը����¶�û�䣬��K=1=
(
| ||||
(
|
| 2 |
| 15 |
�ݴ˷����жϣ�
���
�⣺�������⣺CO+H2O��g��?CO2 +H2��
��ʼ���ʵ�����1 2 0 0
�仯���ʵ�����
ƽ�����ʵ�����
ƽ�ⳣ��K=
=1
��ͨ��2molˮ����ʱ���������ɶ�����̼xmol����
CO+H2O��g��?CO2 +H2��
��ʼ���ʵ�����
+2
�仯���ʵ�����x x x x
ƽ�����ʵ�����
-x
+2-x
+x
+x
���Ը����¶�û�䣬��K=1=
�����x=
mol��
A���¡���ƽ��ʱ�����������ѹǿ֮�ȵ������ʵ���֮�ȣ�����Ϊ��Ӧǰ���������û�б仯������Ϊ��1+2������1+2+2��=3��5����A��ȷ��
B����ƽ��ʱH2O��ת����Ϊ
��100%=20%����B��ȷ��
C����ƽ��ʱCO��Ũ��Ϊ
-
=0.2 mol?L-1����C��ȷ��
D����Ϊ��Ӧǰ�����û�䣬�����ܶ�֮�ȵ�������֮�ȣ������¡���ƽ��ʱ����������������Ϊ��1��28+2��18������1��28+4��18��=64��100=16��25����D����
��ѡD��
��ʼ���ʵ�����1 2 0 0
�仯���ʵ�����
| 2 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
ƽ�����ʵ�����
| 1 |
| 3 |
| 4 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
ƽ�ⳣ��K=
| ||||
|
��ͨ��2molˮ����ʱ���������ɶ�����̼xmol����
CO+H2O��g��?CO2 +H2��
��ʼ���ʵ�����
| 1 |
| 3 |
| 4 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
�仯���ʵ�����x x x x
ƽ�����ʵ�����
| 1 |
| 3 |
| 4 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
���Ը����¶�û�䣬��K=1=
(
| ||||
(
|
| 2 |
| 15 |
A���¡���ƽ��ʱ�����������ѹǿ֮�ȵ������ʵ���֮�ȣ�����Ϊ��Ӧǰ���������û�б仯������Ϊ��1+2������1+2+2��=3��5����A��ȷ��
B����ƽ��ʱH2O��ת����Ϊ
| ||||
| 2+2 |
C����ƽ��ʱCO��Ũ��Ϊ
| 1 |
| 3 |
| 2 |
| 15 |
D����Ϊ��Ӧǰ�����û�䣬�����ܶ�֮�ȵ�������֮�ȣ������¡���ƽ��ʱ����������������Ϊ��1��28+2��18������1��28+4��18��=64��100=16��25����D����
��ѡD��
���������⿼���˻�ѧƽ��Ӱ�����ط����жϼ�����㣬ע��ƽ�ⳣ�������¶��йأ����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
���и������ʣ�ǰ����εμӵ�������ֱ���������ȳ��ְ�ɫ������������������ʧ���ǣ�������
| A��AlCl3��Һ����NaOH��Һ�� |
| B��Ba��OH��2��Һ����Al2��SO4��3��Һ�� |
| C���������NaAlO2��Һ�� |
| D����ˮ����AlCl3��Һ�� |
����֪Ũ�ȵ�����ζ�δ֪Ũ�ȵ�NaOH��Һ�ᵼ�²�õ�NaOH��ҺŨ��ƫ�ߵ��ǣ�������
| A���ζ�ǰ�ζ����������ݣ��ζ���������� |
| B����ʽ�ζ�����ȡNaOH��Һʱ��δ������ϴ���� |
| C���ζ�ʱ�ﵽ�ζ��յ�ʱ���Ӷ��� |
| D����ƿʢװNaOH����Һǰ������ˮϴ�� |
��������ijЩ�������ص�Ӱ�죬��ռ�ṹ�����仯�������������ʺ�������Եı仯����Ϊ�����ʵģ�������
| A��ˮ�� | B������ | C������ | D���۳� |
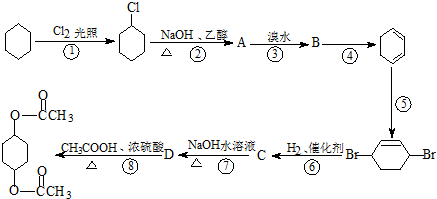
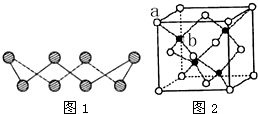 ��A�������������Se�����ڣ�Te����Ԫ���ڻ������г����ֳ���������̬������A��Ԫ�صĻ��������о�����������������Ҫ��;����ش��������⣺
��A�������������Se�����ڣ�Te����Ԫ���ڻ������г����ֳ���������̬������A��Ԫ�صĻ��������о�����������������Ҫ��;����ش��������⣺
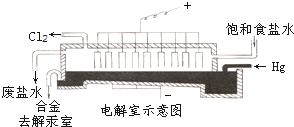 ���������ɵ���Ȼ��Ʊ�����Һ�Ƶã�������һ�ֵ�ⷨ�й���������������������������Һ̬������ѭ��ʹ�ã�����������ʯī�������������ɵ����������������ɣ���ͼ����
���������ɵ���Ȼ��Ʊ�����Һ�Ƶã�������һ�ֵ�ⷨ�й���������������������������Һ̬������ѭ��ʹ�ã�����������ʯī�������������ɵ����������������ɣ���ͼ����