��Ŀ����
��ҵ���������β���к��е�������NOx��NO��NO2�Ļ������費��N2O4��������̬���������ཡ�������ϴ����в��
��1����ҵ�Ͽ��ð������շ�����NOx����Ӧԭ��Ϊ��4xNH3+6NOx
��2x+3��N2+6xH2O��ij��ѧ��ȤС��ģ��ô������̵�ʵ��װ����ͼ1���г�װ������ȥ����
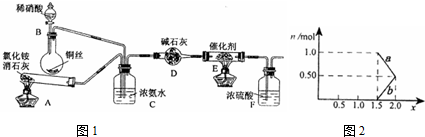
��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ ��
��װ��D�м�ʯ�ҵ������� ��
��2����ҵ��Ҳ����Na2CO3��Һ���շ�����NOx����֪��NO������Na2CO3��Һ��Ӧ��NO+NO2+Na2CO3=2NaNO2+
CO2��2NO2+Na2CO3=NaNO2+NaNO3+CO2��
�ٵ�NOx��Na2CO3��Һ��ȫ����ʱ��x��ֵ�������� ������ĸ����
A��1.3 B��1.6 C��1.8
�ڽ�1mol NOxͨ��Na2CO3��Һ�У�����ȫ����ʱ����Һ�����ɵ�NO-3��NO-2�������ӵ����ʵ�����x�仯��ϵ��ͼ2��ʾ��ͼ���߶�a��ʾ ������xֵ�仯�Ĺ�ϵ������������������Ϊ21.2%��Na2CO3��Һ���գ�����ҪNa2CO3��Һ���� g��
��1����ҵ�Ͽ��ð������շ�����NOx����Ӧԭ��Ϊ��4xNH3+6NOx
| ||

��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪ
��װ��D�м�ʯ�ҵ�������
��2����ҵ��Ҳ����Na2CO3��Һ���շ�����NOx����֪��NO������Na2CO3��Һ��Ӧ��NO+NO2+Na2CO3=2NaNO2+
CO2��2NO2+Na2CO3=NaNO2+NaNO3+CO2��
�ٵ�NOx��Na2CO3��Һ��ȫ����ʱ��x��ֵ��������
A��1.3 B��1.6 C��1.8
�ڽ�1mol NOxͨ��Na2CO3��Һ�У�����ȫ����ʱ����Һ�����ɵ�NO-3��NO-2�������ӵ����ʵ�����x�仯��ϵ��ͼ2��ʾ��ͼ���߶�a��ʾ
���㣺��������������ʼ���Ի�����Ӱ��
ר�⣺����Ԫ��
��������1�����ڼ��������£��Ȼ�狀��������Ʒ�Ӧ�����Ȼ��ơ�������ˮ��
�ڰ������ڼ������壬���ü������ʸ��
��2���ٵ�NOx��Na2CO3��Һ��ȫ����ʱ����n��NO2����n��NO����
�����ü������غ㷨���������
�ڰ������ڼ������壬���ü������ʸ��
��2���ٵ�NOx��Na2CO3��Һ��ȫ����ʱ����n��NO2����n��NO����
�����ü������غ㷨���������
���
�⣺��1�����ڼ��������£��Ȼ�狀��������Ʒ�Ӧ�����Ȼ��ơ�������ˮ��2NH4Cl+Ca��OH��2
CaCl2+2NH3��+2H2O��
�ʴ�Ϊ��2NH4Cl+Ca��OH��2
CaCl2+2NH3��+2H2O��
�ڼ�ʯ��������ˮ�ֶ�����������������ڼ������壬�������ü�ʯ�Ҹ��
�ʴ�Ϊ����ȥ�����к��е�ˮ������
��2���ٵ�NOx��Na2CO3��Һ��ȫ����ʱ����n��NO2����n��NO������n��NO2����n��NO��=1ʱxֵ��С��x��СֵΪ
=1.5����Ϊ����NO������x���ֵ��2����x��ȡֵ��ΧΪ1.5��x��2������x��ֵ��������1.3��
�ʴ�Ϊ��A��
���ü�������x=1.5����ӦΪNO��NO2�������ʵ�����Ϊ1��1������ʽ��Ӧ��û��NO3-����aӦ�ñ�ʾNO2-��
���غ㷨����Ӧ���ɵ�NaNO3��NaNO2�е�Ԫ������Ԫ��֮��Ϊ1��1������1mol NOx����ȫ����������̼����0.5mol������Ϊ53g�������̼������Һ������Ϊ250g��
�ʴ�Ϊ��NO2-��250��
| ||
�ʴ�Ϊ��2NH4Cl+Ca��OH��2
| ||
�ڼ�ʯ��������ˮ�ֶ�����������������ڼ������壬�������ü�ʯ�Ҹ��
�ʴ�Ϊ����ȥ�����к��е�ˮ������
��2���ٵ�NOx��Na2CO3��Һ��ȫ����ʱ����n��NO2����n��NO������n��NO2����n��NO��=1ʱxֵ��С��x��СֵΪ
| 2+1 |
| 2 |
�ʴ�Ϊ��A��
���ü�������x=1.5����ӦΪNO��NO2�������ʵ�����Ϊ1��1������ʽ��Ӧ��û��NO3-����aӦ�ñ�ʾNO2-��
���غ㷨����Ӧ���ɵ�NaNO3��NaNO2�е�Ԫ������Ԫ��֮��Ϊ1��1������1mol NOx����ȫ����������̼����0.5mol������Ϊ53g�������̼������Һ������Ϊ250g��
�ʴ�Ϊ��NO2-��250��
�����������Ե�������Ϊ���忼�������ʼ�ķ�Ӧ����ȷ���ʵ������ǽⱾ��ؼ����ѵ��ǣ�2����ļ��㣬Ҫ��Ϸ���ʽ�и���������֮��Ĺ�ϵʽ��𣬻���������Ϣ����xֵ���Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
���е��뷽��ʽ�У���ȷ���ǣ�������
| A��NaNO3=Na++NO3- | ||
| B��Ba��OH��2=Ba2++OH- | ||
C��MgSO4�TMg+2+S
| ||
| D��H2SO4=H2++SO42- | ||
| E��H2SO4=H2++SO42- |
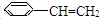 ��5���л���ֱ���һ����������H2��ַ�Ӧ�������и���Ҫ�����
��5���л���ֱ���һ����������H2��ַ�Ӧ�������и���Ҫ�����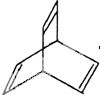 I���л���Ľṹ���á�����ʽ����ʾ��CH3-CH=CH-CH3�ɼ�д
I���л���Ľṹ���á�����ʽ����ʾ��CH3-CH=CH-CH3�ɼ�д Ϊ���л���X�ļ���ʽ��ͼ��
Ϊ���л���X�ļ���ʽ��ͼ��