��Ŀ����
3�����Ȼ����ǻ�ѧ�ϳ��еij������ʣ����Ȼ������������ڸ������ܱ������������Ʊ����Ȼ�����������ͼ��ʾ��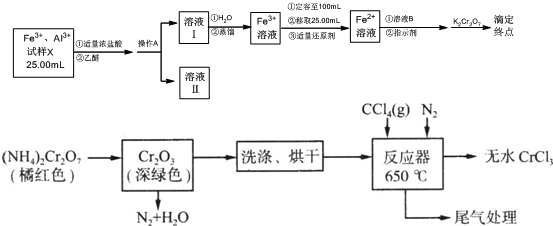
��1���ظ���立ֽ������������������Cr2O3������ˮ����������ˮϴ�ӣ�����ü����ж�����ϴ�Ӹɾ����Ͼ���Ӧװ���е�����������ʹ��Ӧ������ʽ¯�н��з�Ӧ��
��2����֪CCl4�е�Ϊ76.8�棬Ϊ��֤�ȶ���CCl4���������˵ļ��ȷ�ʽ��ˮԡ���ȣ������¶ȼ�ָʾ�¶ȣ���
��3������ͼװ���Ʊ�CrCl3ʱ��
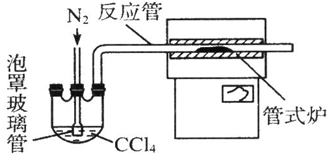
��Ӧ���з�������Ҫ��ӦΪ��Cr2O3+3CCl4�T2CrCl3+3COCl2������������ƿ��ͨ��N2�����ã�
�Ͼ���Ӧװ���е�����������ʹ��Ӧ������ʽ¯�н��з�Ӧ��
��4����Ʒ�����Ȼ������������IJⶨ����ȡ��Ʒ0.3000g����ˮ�ܽⲢ������250mL����ƿ�У���ȡ25.00mL�ڵ���ƿ��һ�ִ�������ƿ���У��������к����1g Na2O2����ּ�����У��ʵ�ϡ�ͣ�Ȼ��������2mol•L-1H2SO4����Һ��ǿ���ԣ���ʱ����Cr2O72-���ڣ��ټ���1.1g KI������ҡ�ȣ���ַ�Ӧ�����Cr3+���ڣ��ڰ�������5min����1mLָʾ������0.0250mol•L-1��Na2S2O3��Һ�ζ����յ㣬ƽ�вⶨ���Σ�ƽ�����ı�Na2S2O3��Һ21.00mL������֪��2Na2S2O3+I2�TNa2S4O6+2NaI��
�ٵζ�ʵ���ѡ�õ�ָʾ������Ϊ���ۣ��ж��յ�����������һ�ε���ʱ����ɫǡ����ȫ��ȥ���Ұ�����ڲ��ָ�ԭɫ�����ζ�ʱ����֣��տ����ֲ���ɫ��ֹͣ�ζ������ʹ��Ʒ����ˮ���Ȼ��������������IJ������_ƫ�ͣ��ƫ�ߡ���ƫ�͡�����Ӱ�족����
�ڼ���Na2O2��Ҫ������У�����Ҫԭ���dz�ȥ�����ܽ����������ֹ������I-����������ƫ�ߵ���
�ۼ���KIʱ������Ӧ�����ӷ���ʽΪCr2O72-+6I-+14H+�T2Cr3++3I2+7H2O��
����Ʒ����ˮ���Ȼ�������������Ϊ92.5%�����������һλС����
���� ��������ͼ֪���ظ���立ֽ������������������Cr2O3������ˮ����������ˮ��Ȼ��ϴ�ӡ���ɵõ�Cr2O3���ڼ��������£��ڷ�Ӧ���з�����ӦCr2O3+3CCl4�T2CrCl3+3COCl2�����Ȼ������������ڸ������ܱ�����������Ϊ��ֹ���Ȼ�����������ͨ�뵪�����ҳ���ĵ�����ʹ��Ӧ������ʽ¯�н��з�Ӧ�����õ���ˮ���Ȼ�����
��1���ظ���立ֽⲻ��ȫ�������ܺ����������������ʣ���Ϊ��NH4��2Cr2O7�Խۺ�ɫ�����Կ����ṩ��ɫ���жϣ�
��2����ΪCCl4�е�Ϊ76.8�棬�¶ȱȽϵͣ���˱�֤�ȶ���CCl4����������ͨ��ˮԡ������������������
��3�����������������Ȼ����ҳ��뵪����ʹ��Ӧ������ʽ¯�н��з�Ӧ��
��4��������Na2S2O3�ζ�����I2��I2����������ɫ���������һ��ʱ�������Һ��ɫ�ڰ�����ڲ���ɫ����ﵽ�ζ��յ㣻
���ζ�ʱ����֣��տ����ֲ���ɫ��ֹͣ�ζ������ʹ��Ʒ����ˮ���Ȼ��������������IJ������ƫ�ͣ�
����Һ�����ܽ��������������������I-������
��Cr2O72-��I-����������ԭ���ɵⵥ�ʺ����ӣ�
����CrԪ���غ㼰����ʽ�ɵù�ϵʽ2Cr3++��Cr2O72-��3I2��6Na2S2O3�����ݹ�ϵʽ���㣮
��� �⣺��������ͼ֪���ظ���立ֽ������������������Cr2O3������ˮ����������ˮ��Ȼ��ϴ�ӡ���ɵõ�Cr2O3���ڼ��������£��ڷ�Ӧ���з�����ӦCr2O3+3CCl4�T2CrCl3+3COCl2�����Ȼ������������ڸ������ܱ�����������Ϊ��ֹ���Ȼ�����������ͨ�뵪�����ҳ���ĵ�����ʹ��Ӧ������ʽ¯�н��з�Ӧ�����õ���ˮ���Ȼ�����
��1����Ϊ��NH4��2Cr2O7�Խۺ�ɫ�����һ��ϴ�ӵ�����Һ����ɫ��˵��ϴ�Ӹɾ���
�ʴ�Ϊ�����һ��ϴ�ӵ�����Һ����ɫ��
��2����ΪCCl4�е�Ϊ76.8�棬�¶ȱȽϵͣ���˱�֤�ȶ���CCl4����������ͨ��ˮԡ�����������������������¶ȼ�ָʾ�¶ȣ�
�ʴ�Ϊ��ˮԡ���ȣ������¶ȼ�ָʾ�¶ȣ���
��3�����������������Ȼ����ҳ��뵪����ʹ��Ӧ������ʽ¯�н��з�Ӧ��Ϊ��ֹ���Ȼ�����������ʹ��Ӧ������ʽ¯�н��з�Ӧ�����뵪����
�ʴ�Ϊ���Ͼ���Ӧװ���е�����������ʹ��Ӧ������ʽ¯�н��з�Ӧ��
��4��������Na2S2O3�ζ�����I2��I2����������ɫ�����Կ����õ�����ָʾ�����������һ��ʱ�������Һ��ɫ�ڰ�����ڲ���ɫ����ﵽ�ζ��յ㣻
���ζ�ʱ����֣��տ����ֲ���ɫ��ֹͣ�ζ������ʹ��Ʒ����ˮ���Ȼ�����Ӧ����ȫ�����Ե����������������IJ������ƫ�ͣ�
�ʴ�Ϊ�����ۣ����һ�ε���ʱ����ɫǡ����ȫ��ȥ���Ұ�����ڲ��ָ�ԭɫ��ƫ�ͣ�
����Һ�����ܽ��������������������I-������������ȥ�����ܽ������ʹ���ɵ�I2����������ƫ�ߵ����ʼ�����У�����Ҫԭ���ǣ���ȥ�����ܽ����������ֹ������I-����������ƫ�ߵ���
�ʴ�Ϊ����ȥ�����ܽ����������ֹ������I-����������ƫ�ߵ���
��Cr2O72-��I-����������ԭ���ɵⵥ�ʺ����ӣ����ӷ���ʽΪCr2O72-+6I-+14H+�T2Cr3++3I2+7H2O��
�ʴ�Ϊ��Cr2O72-+6I-+14H+�T2Cr3++3I2+7H2O��
����25.00mL��Һ��n��Cr3+������CrԪ���غ㼰����ʽ�ɵù�ϵʽ2Cr3++��Cr2O72-��3I2��6Na2S2O3�����ݹ�ϵʽ���㣮
2Cr3++��Cr2O72-��3I2��6Na2S2O3��
2 6
n��Cr3+�� 0.0250mol/L��0.021L
��n��Cr3+��=0.0250mol/L��0.021L��$\frac{1}{3}$������250mL��Һ��n�䣨Cr3+��=0.0250mol/L��0.021L��$\frac{1}{3}$��$\frac{250mL}{25mL}$=0.00175mol������CrԪ���غ��֪n��CrCl3��=n�䣨Cr3+��=0.00175mol��������Ʒ��m��CrCl3��=0.00175mol��158.5g/mol=0.2774g������Ʒ����ˮ���Ȼ�������������Ϊ$\frac{0.2774g}{0.3g}$��100%=92.5%��
�ʴ�Ϊ��92.5%��
���� ���⿼��ѧ���Ե�ʵ�鷽��ԭ�������������ۡ�������ɺ����IJⶨ���ζ�Ӧ�á���ѧ����ȣ���Ŀ�ѶȽϴ�����ʵ��ԭ���ǹؼ����ѵ��ǣ�4������㣬�״����ǻ���ʵ���������Ŀ�Ѷ��еȣ�

 ��ĩ���䵥Ԫ�����ิϰ��ϵ�д�
��ĩ���䵥Ԫ�����ิϰ��ϵ�д�| A�� | ��ϩ�����뱽������̼̼����ͬ�������߶��ܷ����ӳɷ�Ӧ | |
| B�� | ��ϩ��������ϩ������ϩ���ӽṹ�ж����в����ͼ� | |
| C�� | �Ҵ��ȿ�����ȼ�ϣ�Ҳ�������ճ�������ʹ�ã�����ˮ�Ҵ�������ɱ����������Ư�� | |
| D�� | ʯ�͵ķ����ѻ���ú�ĸ����ǻ�ѧ�仯 |
| A�� | ${\;}_{6}^{12}$C��${\;}_{6}^{14}$Cԭ�ӵ���������� | |
| B�� | ��ˮ��20�����90��ʱ��pHֵС | |
| C�� | ������ˮPH����7�������PHС��7 | |
| D�� | ��������һ��ǿ����������һ������ |
| A�� | �� 2 min �ڵķ�Ӧ���ʣ��� C ��ʾ�� 0.4 mol/��L•min�� | |
| B�� | �ֱ��� B��C��D ��ʾ��ѧ��Ӧ�������ֵ�� 3��2��1 | |
| C�� | �ﵽ��ѧ��Ӧ��ʱ��B ����ת����Ϊ 100% | |
| D�� | ������������ A ��Ͷ�������Լӿ�÷�Ӧ������ |
| A�� | ��ϩ���ͻ�������Ϊͬ���칹�� | |
| B�� | ����ʽ���һ��������CH2�����л���һ����Ϊͬϵ�� | |
| C�� | ��ϩ����ͨʽ��CnH2n�����ϩ����ͨʽΪCnH2n-2 | |
| D�� | n��2ʱ��CnH2n+2��ͬ���칹���CnH2n��ͬ���칹��� |
| A�� | ��Ȳ�ĵ���ʽ��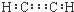 | B�� | �Լ����ӵĽṹ��ʽ��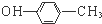 | ||
| C�� | ȩ���Ľṹ��ʽ��-CHO | D�� | ������ӵı���ģ��Ϊ��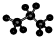 |
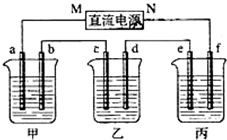 ��ͼ��ʾװ���У��ס��ҡ��������ձ����ηֱ�ʢ��100g 5.00%��NaOH��Һ��������CuSO4��Һ��100g 10.00%��K2SO4��Һ���缫��Ϊʯī�缫��
��ͼ��ʾװ���У��ס��ҡ��������ձ����ηֱ�ʢ��100g 5.00%��NaOH��Һ��������CuSO4��Һ��100g 10.00%��K2SO4��Һ���缫��Ϊʯī�缫��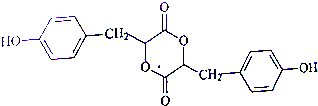
 ��
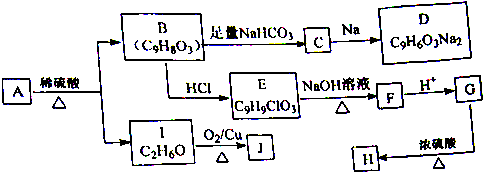
��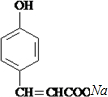 +
+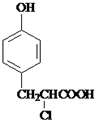 $��_{��}^{����}$
$��_{��}^{����}$ +HCl��
+HCl��