��Ŀ����
������Һ�У�������Ũ�ȵĴ�С˳����ȷ���� �� ��
A. 10 mL 0.1 mol��L-1���ᣬc��Cl-����c(OH-)��c(H+)
B. 10 mL 0.1 mol��L-1��ˮ��c(OH-)��c(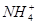 )��c(H+)
)��c(H+)
C. 5 mL 0.2 mol��L-1NaOH��Һ��c(Na+)��c(OH-)��c(H+)
D.10 mL 0.5 mol��L-1NaOH��Һ��6 mL 1 mol��L-1�����ϣ�c(Cl-)��c(Na+)��c(OH-)��c(H+)
���𰸡�
B
��������
���������������Һ�����ԣ�A���������������Һ��c(OH-)��c(Na+)��c(H+)��C�����.10 mL 0.5 mol��L-1NaOH��Һ��6 mL 1 mol��L-1�����ϣ���Һ�����ԣ�D�����ѡB��
���㣺����Ũ�ȴ�С�Ƚ�
����������Ũ�ȴ�С��Ҫ���������غ㶨�ɺ���Һ������Խ��бȽϡ�

��ϰ��ϵ�д�
 �±�Сѧ��Ԫ�Բ���ϵ�д�
�±�Сѧ��Ԫ�Բ���ϵ�д� �ִʾ��ƪϵ�д�
�ִʾ��ƪϵ�д�
�����Ŀ
 �ش����и��⣺
�ش����и��⣺ HCO3-+OH-
HCO3-+OH-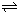 C2D4��g������H��0���ں��º��������£���һ����CD2��C2D4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ��ͼ��ʾ��
C2D4��g������H��0���ں��º��������£���һ����CD2��C2D4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ��ͼ��ʾ��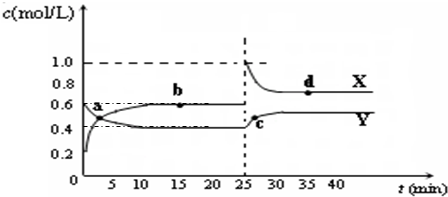
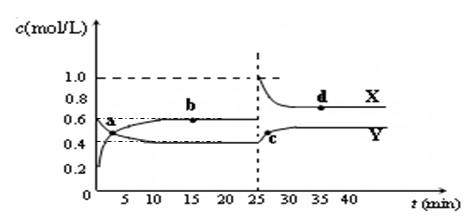
 C2D4(g)����H��0���ں��º��������£���һ����CD2��C2D4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ����ͼ��ʾ��
C2D4(g)����H��0���ں��º��������£���һ����CD2��C2D4�Ļ������ͨ���ݻ�Ϊ2L���ܱ������У���Ӧ�����и����ʵ����ʵ���Ũ��c��ʱ��t�ı仯��ϵ����ͼ��ʾ��