��Ŀ����
 ��������Ȼ������Ҫ�ɷ֣�������������Ӧ�÷dz��㷺��һ�ֻ�ѧ���ʣ�
��������Ȼ������Ҫ�ɷ֣�������������Ӧ�÷dz��㷺��һ�ֻ�ѧ���ʣ���1��һ�������£��ü�����������������NOx������Ⱦ����֪��
CH4��g��+4NO��g��=2N2��g��+CO2��g��+2H2O��g������H1
CH4��g��+4NO2��g��=4NO��g��+CO2��g��+2H2O��g������H2
����һ������ͬ�����¶�H2������ܶ�Ϊ17��NO��NO2�Ļ�����壬��16g�����������ԭ�û�����壬ǡ�����ɵ�����������̼�����ˮ���������ų�1042.8kJ������
�ٸû��������NO��NO2�����ʵ���֮��Ϊ
����֪�����Ȼ�ѧ����ʽ�С�H1=-1160kJ/mol�����H2=
����һ��������NO������Էֽ�ΪNO2�����N2���壬д���÷�Ӧ���Ȼ�ѧ����ʽ
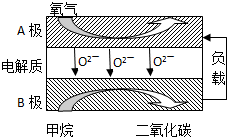
��2���Լ���Ϊȼ�ϵ����͵�أ���ɱ�����������Ϊȼ�ϵĴ�ͳȼ�ϵ�أ�Ŀǰ�õ��㷺���о�����ͼ��Ŀǰ�о��϶��һ�����������ȼ�ϵ�ع���ԭ��ʾ��ͼ���ش��������⣺
��B�����
�����ø�ȼ�ϵ������Դ����ʯī���缫���100mL 1mol/L������ͭ��Һ��д�������ĵ缫��Ӧʽ
���㣺�йط�Ӧ�ȵļ���,�Ȼ�ѧ����ʽ,ԭ��غ͵��صĹ���ԭ��,���ԭ��
ר�⣺��ѧ��Ӧ�е������仯
��������1������ͬ�����£���ͬ�����Ħ������֮�ȵ������ܶ�֮�ȣ��ݴ˼��㵪������ƽ��Ħ���������ٸ���ƽ��Ħ����������NO�Ͷ������������ʵ���֮�ȣ�
�ڸ��ݸ�˹����ȷ������Ͷ�������֮��ķ���ʽ�����ݼ����NO��NO2֮��ķ�Ӧȷ���ֱ��NO��NO2֮�䷴Ӧ��Ҫ��������ʵ���֮�ȣ��ٸ����ʱ���м��㣻
�۸��ݸ�˹������д�Ȼ�ѧ����ʽ��
��2����BΪ�������ܷ�ӦʽΪCH4+2O2+=CO2+2H2O��������ӦʽΪ��2O2+8e-=4O2-����ʽ����ɵø����缫��Ӧʽ��
�ݿ�ʼ�η�����Ӧ��2Cu2++2H2O
2Cu+O2��+4H+��ͭ������ȫ�ŵ������Ӧ2H2O
2H2��+O2���������ĵ缫��Ӧʽ�����������ӷŵ磬�������ռ���������������ʱ���������������������ȣ���������Ϊxmol�����ݵ���ת���غ��з��̼��㣬�ٸ��ݵ���ת���غ�������ĵļ��飻���ݵ���е�����ת���ʷ�����
�ڸ��ݸ�˹����ȷ������Ͷ�������֮��ķ���ʽ�����ݼ����NO��NO2֮��ķ�Ӧȷ���ֱ��NO��NO2֮�䷴Ӧ��Ҫ��������ʵ���֮�ȣ��ٸ����ʱ���м��㣻
�۸��ݸ�˹������д�Ȼ�ѧ����ʽ��
��2����BΪ�������ܷ�ӦʽΪCH4+2O2+=CO2+2H2O��������ӦʽΪ��2O2+8e-=4O2-����ʽ����ɵø����缫��Ӧʽ��
�ݿ�ʼ�η�����Ӧ��2Cu2++2H2O
| ||
| ||
���
�⣺��1��������ͬ�����¶�H2������ܶ�Ϊ17��NO��NO2�Ļ�����壬��ͬ�����£�������ܶ�֮�ȵ�����Ħ������֮�ȣ����Ի�������Ħ������Ϊ34g/mol����n��NO��Ϊx��n��NO2��Ϊy��
�������Ħ������=
=34g/mol��x��y=3��1��
�ʴ�Ϊ��3��1��
��16g��������ʵ���=
=1mol��
���ݸ�˹����֪��CH4��g��+4NO��g���T2N2��g��+CO2��g��+2H2O��g����H1
CH4��g��+2NO2��g���TN2��g��+CO2��g��+2H2O��g����H=0.5����H1+��H2��
NO��NO2�����ʵ���֮��Ϊ3��1�����ݷ���ʽ֪����NO��NO2��ȫת��Ϊ����ʱ���ֱ��NO��NO2��Ӧ�ļ�������ʵ���֮��=
��
=3��2������0.6mol�����NO��Ӧ��0.4mol�ļ����NO2��Ӧ��0.6mol�����NO��Ӧ�ų�������=0.6mol��1160kJ/mol=696kJ��0.4mol�����NO2��ȫ��Ӧ�ų�������=1042.8kJ-696kJ=346.8kJ����0.4mol�����NO2��ȫ��Ӧ�ų�������=0.5����H1+��H2����0.4mol=0.2mol����1160kJ/mol+��H2��=346.8kJ�����H2=
=-574 kJ/mol��
�ʴ�Ϊ��-574 kJ/mol��
��CH4��g��+4NO��g��=2N2��g��+CO2��g��+2H2O��g����H1=-1160kJ/mol��
CH4��g��+4NO2��g��=4NO��g��+CO2��g��+2H2O��g����H1=-574 kJ/mol��
���ݸ�˹���ɣ�
�ã�4NO��g��=2NO2��g��+N2��g������H=-293 kJ/mol��
�ʴ�Ϊ��4NO��g��=2NO2��g��+N2��g����H=-293 kJ/mol��
��2����BΪ�������ܷ�ӦʽΪCH4+2O2+=CO2+2H2O��������ӦʽΪ��2O2+8e-=4O2-����ʽ�����������ӦΪ��CH4-8e-+4O2-=CO2+2H2O��
�ʴ�Ϊ������CH4-8e-+4O2-=CO2+2H2O��
������ͭ�����ʵ���=0.1L��1mol/L=0.1mol����ʼ�η�����Ӧ��2Cu2++2H2O
2Cu+O2��+4H+��ͭ������ȫ�ŵ������Ӧ2H2O
2H2��+O2���������ĵ缫��Ӧʽ��4OH--4e-=O2��+2H2O���������ռ���������������ʱ���������������������ȣ���������Ϊxmol�����ݵ���ת���غ㣬��0.1mol��2+2x=4x�����x=0.1�����ݵ���ת���غ㣬��֪���ĵļ������ʵ���=
=0.05mol�������ļ�������=0.05mol��22.4L/mol=1.12L��ԭ����з�����Ӧʱ��ѧ�ܲ�ȥȫ��ת��Ϊ���ܣ����������ת���ʴﲻ��100%������ʵ�������ĵļ��������������ϴ�
�ʴ�Ϊ��4OH--4e-=O2��+2H2O��1.12L���������ת���ʴﲻ��100%��
�������Ħ������=
| 30xg+46yg |
| (x+y)mol |
�ʴ�Ϊ��3��1��
��16g��������ʵ���=
| 16g |
| 16g/mol |
���ݸ�˹����֪��CH4��g��+4NO��g���T2N2��g��+CO2��g��+2H2O��g����H1
CH4��g��+2NO2��g���TN2��g��+CO2��g��+2H2O��g����H=0.5����H1+��H2��
NO��NO2�����ʵ���֮��Ϊ3��1�����ݷ���ʽ֪����NO��NO2��ȫת��Ϊ����ʱ���ֱ��NO��NO2��Ӧ�ļ�������ʵ���֮��=
| 3 |
| 4 |
| 1 |
| 2 |
| 346.8kJ |
| 0.2mol |
�ʴ�Ϊ��-574 kJ/mol��
��CH4��g��+4NO��g��=2N2��g��+CO2��g��+2H2O��g����H1=-1160kJ/mol��
CH4��g��+4NO2��g��=4NO��g��+CO2��g��+2H2O��g����H1=-574 kJ/mol��
���ݸ�˹���ɣ�
| ��-�� |
| 2 |
�ʴ�Ϊ��4NO��g��=2NO2��g��+N2��g����H=-293 kJ/mol��
��2����BΪ�������ܷ�ӦʽΪCH4+2O2+=CO2+2H2O��������ӦʽΪ��2O2+8e-=4O2-����ʽ�����������ӦΪ��CH4-8e-+4O2-=CO2+2H2O��
�ʴ�Ϊ������CH4-8e-+4O2-=CO2+2H2O��
������ͭ�����ʵ���=0.1L��1mol/L=0.1mol����ʼ�η�����Ӧ��2Cu2++2H2O
| ||
| ||
| 0.1mol��4 |
| 8 |
�ʴ�Ϊ��4OH--4e-=O2��+2H2O��1.12L���������ת���ʴﲻ��100%��
�����������ۺϿ����˷�Ӧ�ȵļ��㡢�绯ѧ֪ʶ�ȣ���Ŀ����������ϴ����ڶԻ���֪ʶ�ۺ�Ӧ�õĿ��飬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ