��Ŀ����
11����1��д��ͼ1����Ţ١������������ƣ�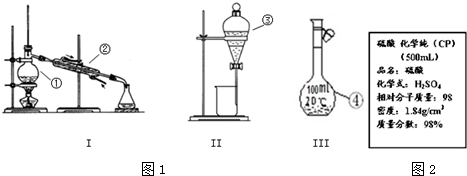
��������ƿ�� �������ܣ� �۷�Һ©����
��2�������١����У�ʹ��ʱ�������Ƿ�©ˮ���Тۢܣ�����������ţ�
��3����ͼ2��ijѧУʵ���Ҵӻ�ѧ�Լ��̵���ص�Ũ�����Լ���ǩ�ϵIJ������ݣ����ø�Ũ��������480mL 1mol•L-1��ϡ���ᣮ�ɹ�ѡ�õ������У�
�ٽ�ͷ�ιܢ���ƿ���ձ��ܲ�������ҩ�ע���Ͳ��������ƽ��
��ش��������⣺
a������������ʵ���Ũ��Ϊ18.4mol•L-1��
b������ϡ����ʱ����ȱ�ٵ�������500ml����ƿ ��д�������ƣ���
c�������㣬����480mL 1mol•L-1��ϡ������Ҫ����Ͳ��ȡ����Ũ��������Ϊ27.2mL��
d���������Ƶ�ϡ������вⶨ��������Ũ�ȴ���1mol•L-1�����ƹ��������и�������������������ԭ����A��B��
A������ʱ����������ƿ�̶��߽��ж��ݣ�
B����ϡ�ͺ��ϡ��������ת������ƿ�����žͽ����Ժ��ʵ�������
C��ת����Һʱ��������������Һ��������ƿ���森
D������ƿ������ˮϴ�Ӻ�δ���������������ˮ��
E�����ݺ�����ƿ����ҡ�Ⱥ���Һ����ڿ̶��ߣ��㲹�伸��ˮ���̶ȴ���
���� ��1�����ݳ��������Ĺ�����������ش�
��2����Һ©����100mL����ƿ��ʹ��ǰҪ����Ƿ�©ˮ��
��3��a��������Һ�����ʵ���Ũ��C=$\frac{1000�Ѧ�}{M}$�����㣻
b�����ݲ��������м��㡢��ȡ��ϡ�͡�ϴ�ӡ����ݡ�ҡ�ȵȲ�����ȷ��������
c��������Һϡ�Ͷ���CŨVŨ=CϡVϡ�����㣻
d�������������������ʵ�������Һ�����Ӱ�죬����c=$\frac{n}{V}$�ж϶�������ҺŨ�ȵ�Ӱ�죮
��� �⣺��1����٢ڢ����������Ʒֱ�Ϊ��������ƿ�������ܡ���Һ©����
�ʴ�Ϊ��������ƿ�������ܣ���Һ©����
��2�����Һ©����100mL����ƿ��ʹ��ǰҪ����Ƿ�©ˮ��������ƿ�������ܲ���Ҫ����Ƿ�©ˮ��
�ʴ�Ϊ���ۢܣ�
��3��a����Һ�����ʵ���Ũ��C=$\frac{1000�Ѧ�}{M}$=$\frac{1000��1.84g/ml��98%}{98g/mol}$=18.4mol/L��
�ʴ�Ϊ��18.4��
b�����������м��㡢��ȡ��ϡ�͡�ϴ�ӡ����ݡ�ҡ�ȵȲ�����һ������Ͳ��ȡŨ���ᣬ���ձ���ϡ�ͣ�������Ͳ��ȡˮ�����ձ��������ò��������裬��ȴ��ת�Ƶ�500mL����ƿ��������480ml����ƿ����ѡ��500ml����ƿ���У����ò�����������ϴ���ձ���������2-3�Σ�����ϴ��Һ��������ƿ�У���ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�����ݵߵ�ҡ�ȣ�����������������Ͳ���ձ�����������500mL����ƿ����ͷ�ιܣ�
�ʴ�Ϊ��500mL����ƿ��
c��������480ml����ƿ����ѡ��500ml����ƿ������480mL 1mol•L-1��ϡ���ᣬ����Ҫ��Ũ��������ΪVml��������Һϡ�Ͷ���CŨVŨ=CϡVϡ��֪��18.4mol/L��Vml=1mol•L-1��500mL�����V=27.2mL��
�ʴ�Ϊ��27.2��
d��A������ʱ����������ƿ�̶��߽��ж��ݣ��ʵ���ϡ��Һ���ƫС������c=$\frac{n}{V}$�жϿ�֪Ũ��ƫ�ߣ���A��ȷ��
B����ϡ�ͺ��ϡ��������ת������ƿ�����žͽ����Ժ��ʵ�������һ����ȴ����������ϡ��Һ���ƫС������c=$\frac{n}{V}$�жϿ�֪Ũ��ƫ�ߣ���B��ȷ��
C��ת����Һʱ��������������Һ��������ƿ���棬����Ũ��Һ���ʵ���ƫС������c=$\frac{n}{V}$�жϿ�֪Ũ��ƫ�ͣ���C����
D������ƿ������ˮϴ�Ӻ�δ���������������ˮ�����ƹ�������Ҫ�μ�����ˮ���ʴ���Ӱ�죬��D����
E�����ݺ�����ƿ����ҡ�Ⱥ���Һ����ڿ̶��ߣ��㲹�伸��ˮ���̶ȴ�������ϡ��Һ���ƫ����c=$\frac{n}{V}$�жϿ�֪Ũ��ƫ�ͣ���E����
��ѡAB��
���� ���⿼����һ�����ʵ���Ũ����Һ�����ƹ����еļ���������������ڻ�������Ŀ��ע��c=$\frac{n}{V}$����Ӧ�ã���Ŀ�ѶȲ���

 ��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�
��ĩ���100�ִ��½����ȫ�Ծ�ϵ�д�| A�� | pH=4.3��CH3COOH��CH3COONa�����Һ�У�c��Na+����c��CH3COO-�� | |
| B�� | Ũ��Ϊ0.2 mol/L��CH3COOH��Һ��Ũ��Ϊ0.1 mol/L��NaOH��Һ�������Ϻ�c��CH3COO -��-c��CH3COOH��=2[c��H+��-c��OH -��] | |
| C�� | ����Ũ��Һ������ˮϡ�ͣ�$\frac{c��C{H}_{3}COOH��}{{c}^{2}��{H}^{+}��}$�������� | |
| D�� | a mol/L CH3COOH��Һ��b mol/L NaOH��Һ�������ϣ�������Һ��c��Na+����c��CH3COO-������һ����a��b |
| A�� | ������Һ�е�KW��ͬ����ˮ�������c��H+������=��=��=�� | |
| B�� | ȡ�����ͬ����Һ�١��ڡ��۷ֱ����������۷�Ӧ������H2������������ | |
| C�� | ���������������Һ�ֱ�ϡ��100����������Һ��pH���ۣ��ܣ��ڣ��� | |
| D�� | �����£����ں͢۵������ϣ�c��CH3COO-��-c��Na+��=c��H+��-c��OH-�� |
| A�� | 0.05mol•L-1��H2SO4 | B�� | 0.1mol•L-1��KNO3 | ||
| C�� | 0.1mol•L-1��KOH | D�� | 0.1mol•L-1��NH4NO3 |
| A�� | �����ھƾ���ɵ�� | B�� | �� | ||
| C�� | Fe��OH��3���� | D�� | ���лҳ������Ŀ��� |
 ��֪���±�Ϊ25��ʱijЩ����ĵ���ƽ�ⳣ����
��֪���±�Ϊ25��ʱijЩ����ĵ���ƽ�ⳣ����| CH3COOH | HClO | H2CO3 |
| Ka=1.8��10-5 | Ka=3.0��10-8 | Ka1=4.4��10-7Ka2=4.7��10-11 |
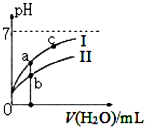
| A�� | ͼ��c��H+����c��R-����ֵ��a�㣾c�㣨HR����CH3COOH��HCIO�� | |
| B�� | pH��ͬ��������ҺŨ�ȹ�ϵ��c��CH3COONa��c��NaHC03��c��NaClO��c��Na2C03�� | |
| C�� | ͼ��a�������Ũ��С��b�������Ũ�� | |
| D�� | Ũ�Ⱦ�Ϊ0��l mol/L��CH3COONa��NaCIO�Ļ����Һ�У�c��OH-��=0��l mol/L-c��ClO-��+c��H+��+c��CH3COOH�� |
| ������ | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 1 | A | B | ||||||
| 2 | D | E | F | G | ||||
| 3 | C | H | I |
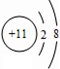 ��I��ԭ�ӽṹʾ��ͼ
��I��ԭ�ӽṹʾ��ͼ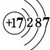 ��
����2����һ�������£�A��E���γ�һ�ּ�������ˮ����̬����������ʽΪ
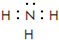 ������������ˮ�����Һ�ʼ��ԣ���ᡱ��������С�����
������������ˮ�����Һ�ʼ��ԣ���ᡱ��������С�������3��Ԫ�ص�����������Ӧ��ˮ�����У�������ǿ����NaOH���üѧ���������Ӽ����ۼ���������ǿ����HClO4�������Ե���Al��OH��3��
��4��A�ֱ���D��E��F��G H�γɵĻ�������ȶ���˳��Ϊ��CH4��NH3��H2O��HF��
 D��0.55 mol��L��1
D��0.55 mol��L��1