��Ŀ����
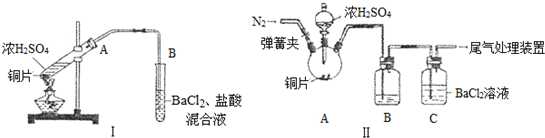
19��ij�о���ѧϰС����̽��ͭ��Ũ���ᷴӦ�������ɼ����ʣ���ͬѧ��װ��I����ʵ�飺���Ⱥ��Թ�A��Һ���ϲ�������������ͭƬ�����ȱ�ں�ʼ�ܽ⣬ͬʱ�����Թ�B�г��ְ�ɫ��������Ӧ�������Թ�A�еõ�����ɫ��Һ���ײ�����������ɫ���壬δ�ܽ��ͭƬ�������к�ɫ���ʸ��ţ��㵹��Һ������ɫ������С�ļ�ˮ���ܽ����Һ����ɫ����ϲ������ϣ����������գ����������ϡ�����Cu2S������ϡ���ᣬ�ܹ������ȵ�ϡ�����У��Ҳ���SO42-���ӣ�
��98.3%Ũ����ķе㣺339�棻 �۾ƾ��ƻ�����¶�Ϊ400〜500��
��1���Թ�B�а�ɫ�����Ļ�ѧʽ��BaSO4���������ӷ���ʽ��ʾ���ɸð�ɫ�����Ŀ���ԭ��Ba2++SO42-=BaSO4����2Ba2++2SO2+O2+2H2O=2BaSO4��+4H+��
��2����ͬѧ�ϼ�ͬѧ��װ�ò����ƣ�����˸Ľ�װ��II����ʵ�飨�г�װ�ú�A�м���װ�����ԣ��������Ѽ��飩���ٴ��ɼУ�ͨ��N2һ��ʱ���رյ��ɼ�
�ڵμ�һ����Ũ���ᣬ����A��һ��ʱ���C��δ���������ɣ�
�����ٵ�Ŀ�����ų�װ���ڵĿ�����O2����ϴ��ƿB�е��Լ���H2O��
��3����ͬѧȡ��ʵ����C����Һ�������μ�һ����ɫ��Һ��Ҳ��������������İ�ɫ���������μӵ��Լ�������C��
A��NaOH��Һ B��NaAlO2��Һ C��H2O2��Һ D������ KMnO4��Һ
��4��̽������ʵ�������ͭ��������ĺ�ɫ������ɣ�ijͬѧ�������ΪCuO��Cu2S��һ�֣���δ�ܽ�ı����к�ɫ���ʸ��ŵ�ͭ�˳�����ϴ�����ĥ�飮����ʵ������ܹ�ȷ������ͬѧ�������BC��
A��ȡ������Ʒ����ϡ������ȹ۲��Ƿ��ܽ⣬�Թܿ��Ƿ�����������ɫ
B��ȡ������Ʒ����ϡ���������۲���Һ�Ƿ����ɫ
C��ȡ������Ʒ��������ϡ������ȣ��ܽ⣬����BaCl2�����Ƿ����SO42-����
��5��ȡ������Ӧ����ƿ�в������Ʊ�CuSO4•5H2O���壬������ȥ����������л�����������ˮ���ܽ⡢����Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�������صøþ��壮
���� ��1��Cu��ŨH2SO4��������Ҫ��ӦΪCu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O������������+4�۵�����л�ԭ�ԣ��ױ������е�����������+6����������������ӣ���������Ӻͱ������γ����ᱵ��������״Ũ����ӷ�������������B�Թ��е�ˮ��Ӧ�������ᣬ��������Ӻͱ����ӷ�Ӧ�������ᱵ������
��2�����������ȶ����ž�װ���еĿ����е�����������֤����ԭ���Ƿ���ȷ��Ϊ��ȥ��Ӧ�д�������״���ᣬ���Խ�����ͨ��H2O���գ�
��3����ʵ����C�е���Һ������SO2�����в���������İ�ɫ����Ӧ��BaSO4������Ҫ��һ����ɫ��������
��4��Cu�ܹ���ϡ���ᷴӦ���ɵ�������������Ϣ��Cu2S������ϡ���ᣬ�ܹ������ȵ�ϡ�����У��Ҳ���SO42-���ӷ�����
��5��������ͭ��Һ����ȡCuSO4•5H2O���壬��Ҫ����Ũ������ȴ�ᾧ������ϴ�ӡ�������أ�
��� �⣺��1����ͬѧ��װ��I����ʵ�飺���Ⱥ��Թ�A��Һ���ϲ�������������Ϊ�ӷ�������������ͭƬ�����ȱ�ں�ʼ�ܽ⣬��Ҫ��ӦΪ��Cu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��ͬʱ�����Թ�B�г��ְ�ɫ��������Ӧ�������Թ�A�еõ�����ɫ��Һ��Ϊ����ͭ��Һ���ײ�����������ɫ����Ϊ����ͭ���壬δ�ܽ��ͭƬ�������к�ɫ���ʸ��ţ�����Ϊ��ͭ���������ʣ��㵹��Һ������ɫ������С�ļ�ˮ���ܽ����Һ����ɫ����һ��˵����ɫ����������ͭ����״Ũ����ӷ��������������B�Թ��У����������������Ӻͱ����ӷ�Ӧ��Ba2++SO42-=BaSO4����Cu��ŨH2SO4��Ӧ���ɵĶ���������+4�۵�����л�ԭ�ԣ��ױ������е�����������+6����������������ӣ���������Ӻͱ������γ����ᱵ��������ӦΪ��2Ba2++2SO2+O2+2H2O=2BaSO4��+4H+��
�ʴ�Ϊ��BaSO4��Ba2++SO42-=BaSO4����2Ba2++2SO2+O2+2H2O=2BaSO4��+4H+��
��2������aͨ��N2Ŀ�����ų�װ���е�O2��C��δ���������ɣ�˵��B�е��Լ������ջӷ�����H2SO4�Ҳ������µ����ʣ���������SO2��Ӧ���ʸ��Լ�Ӧ��H2O��
�ʴ�Ϊ���ų�װ���ڵĿ�����O2���� H2O��
��3����ʵ����C�е���Һ������SO2�����в���������İ�ɫ����Ӧ��BaSO4������Ҫ��һ�����������μӵ���һ����ɫ��Һ������KmnO4��Һ����ɫ��NaOH��Һ��NaAlO2��Һ�������ԣ���ֻ����H2O2��Һ��
�ʴ�Ϊ��C��
��4��A��Cu�ܹ���ϡ���ᷴӦ���ɵ���������˳����к���Cu�������Թܿ�����������ɫҲ����֤��������ͭ����A����
B��Cu2S��Cu��������ϡ���ᣬ��CuO�ܹ�����ϡ���ᣬ����ȡ������Ʒ����ϡ�����������۲���Һ����ɫ��˵������CuO����B��ȷ��
C��CuO��Cu�����ᷴӦ����������������ӣ�Cu2S�ܹ������ȵ�ϡ�����У��Ҳ���SO42-���ӣ�����ȡ������Ʒ��������ϡ��������ܽ⣬������BaCl2������SO42-���Ӳ�����֤��������ͭ����C��ȷ��
�ʴ�Ϊ��BC��
��5����ᾧˮ�����ֹ�ᾧˮ��ʧ������ȴ�ᾧ�����Դ�����ͭ��Һ����ȡCuSO4•5H2O���壬��Ҫ����Ũ������ȴ�ᾧ������ϴ�ӡ�������أ�
�ʴ�Ϊ������Ũ������ȴ�ᾧ��
���� ���������о�̽��ͭ��Ũ���ᷴӦ�������ɼ�����Ϊ���忼����ѧ���ķ�����������������ѧʵ���������漰���ӷ���ʽ����д����������ѡ�����ʵļ���ȣ�������ѧ���������֪ʶ����������Ŀ�Ѷ��еȣ�

 �������ϵ�д�
�������ϵ�д�| ѡ�� | ʵ����������� | ���� |
| A | �ò�˿պȡij����Һ�ھƾ������������գ��������ɫ������ɫ�ܲ����� | ������Һ�п��ܺ���Ԫ�� |
| B | ��ij��ɫ��Һ��ͨ�˹�����CO2���壬�а�ɫ�������� | ����Һ��һ������SiO32- |
| C | ��Ʒ����Һ��ͨ��ij�������Һ��ɫ | ������һ����SO2 |
| D | ��NaOH��Һ�еμ�����MgCl2��Һ��������ɫ�����������μ�FeCl3��Һ�������ɫ���� | Fe��OH��3���ܽ��С��Mg��OH��2���ܽ�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| ѡ�� | ʵ�� | ���ͻ���� |
| A | �ýྻ��Ptպȡij��Һ������ɫ��Ӧ������ʻ�ɫ | ����Һ��һ������Na+����k+ |
| B | �ýྻ�IJ����������Na2O2����֬��������֬��ȼ�� | CO2��H2O��Na2O2��Ӧ�Ƿ��ȷ�Ӧ |
| C | ����ˮ�е���ֲ���ͣ����Ͳ�����ɫ | �岻������֬ |
| D | �������ữ��H2O2����Fe��NO3��2��Һ����Һ���ɫ | H2O2�������Ա�Fe3+ǿ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ���뾶��С�Ƚϣ�r��N3-����r��O2-�� | |
| B�� | NԪ������Ԫ���γɵĻ�����ֻ������ | |
| C�� | �ڸ÷�Ӧ�����£�NH3�Ļ�ԭ�����ڵ�H2O��ԭ�� | |
| D�� | �ڸ÷�Ӧ�У���Ӧ���������С��������������� |
| A�� | ��1molSO2��1molO2��ַ�Ӧ��ת�Ƶĵ�����Ϊ2��6.02��1023 | |
| B�� | �ڸ�������Ϳˢ�����۵ķ����ᣬ������Ч��ֹ������ʴ | |
| C�� | �������£��������Һ�в���ͨ��NH3��$\frac{c��C{H}_{3}COOH��}{c��{H}^{+}��}$���ϼ�С | |
| D�� | �����£���ӦCl2��g��+H2O��l���THCl��aq��+HClO��aq�� ���Է����У����H��0 |
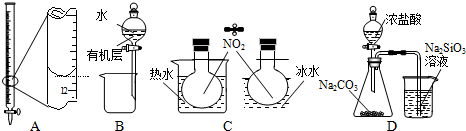
| A�� | װ��A��¼�ζ��յ����Ϊ12.20 mL | |
| B�� | װ��B��������ȡI2��CCl4��Һ�е�I2 | |
| C�� | װ��C������̽���¶ȶ�2NO2?N2O4ƽ����ϵ��Ӱ�� | |
| D�� | װ��D�����ڱȽ�HCl��H2CO3��H2SiO3������ǿ�� |
| A�� | 0.1 mol•L-1 CH3COONa��Һ��0.1 mol•L-1 CaCl2��Һ�������ϣ�c��Na+��+2c��Ca2+��=c��CH3COO-��+c��CH3COOH��+c��Cl-�� | |
| B�� | Ũ�Ⱦ�Ϊ0.1 mol•L-1��С�մ���Һ���ռ���Һ�������ϣ�c��OH-��-c��H+��=c��HCO3-��+c��H2CO3�� | |
| C�� | pH��ͬ�İ�ˮ��KOH��Һ��Ba��OH��2��Һ�У�c��NH4+��=c��K+��=2c��Ba2+�� | |
| D�� | �����£�0.2 mol•L-1 NH4NO3��Һ��0.1 mol•L-1 NaOH��Һ�������ϣ�����pH=9.6����Һ�У�c��NO3-����c��Na+����c��NH4+����c��NH3•H2O����c��OH-����c��H+�� |
| A�� | ʹ�ú��ʵĴ������Լӿ췴Ӧ���� | |
| B�� | ����O2��Ũ�ȿ�ʹSO3��Ũ�ȼ�С | |
| C�� | �����¶ȿ��Լ�С��Ӧ���� | |
| D�� | ����SO2��Ũ�ȿ�������Ӧ���� |
| A�� | ���ȼ���ֻ��һ�֣�˵���������������ṹ | |
| B�� | 2-��ϩ�ĸ�̼ԭ�ӹ�ƽ�棬˵����ϩ������ƽ��ṹ | |
| C�� | ����Ȳ����������ԭ�ӹ�ƽ�棬˵����Ȳ������ԭ����һ��ֱ���� | |
| D�� | �ڶ��ױ�ֻ��һ�֣�˵���������в����ڵ�˫�������̼̼���� |