��Ŀ����
7�����ͼ��ѡ�ñ�Ҫ��װ�ý��е�ⱥ��ʳ��ˮ��ʵ�飬Ҫ��ⶨ���������������������25mL���������������������ԣ�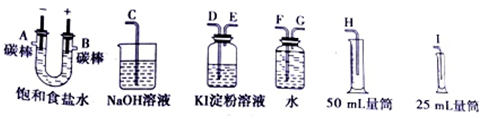
��1��B�������ĵ缫��Ӧʽ��2Cl--2e-=Cl2����
��2�������������ʵ��װ��ʱ�����ӿڵ���ȷ����˳��Ϊ��
A��G��F��H��B��D��E��C��
��3����ʵ���У�ʢ��KI������Һ�������з�����Ӧ�����ӷ���ʽΪCl2+2I-=I2+2Cl-��
��4����֪�����ò�����H2�����Ϊ44.8mL���Ѿ�����ɱ�״������������Һ�����Ϊ50mL����ʱ��Һ��NaOH�����ʵ���Ũ��Ϊ��0.08mol/L��
���� ��1��Ҫ��������̼�����缫����Ȼ�����Һ��ȡ������������������̼���������������������ӷŵ����������������������ӷŵ�����������
��2����Դ�����ӵ��ص�������̼���ӵ����ơ�-���ˣ���+���˽ӵ�Դ������
��3���õ⻯����Һ���������������ԣ�������������Һ����������β��������ˮ���ռ���������50mL��Ͳʢ���ų���ˮ��ע�������ѭ�������̳���ԭ��
��4�������������������ƵĹ�ϵ�Ǽ����������Ƶ����ʵ������ٸ���c=n/V������������Ũ�ȣ�
��� �⣺��1��Ҫ��������̼����������Ȼ�����Һ��ȡ������������������̼�����������������������ӵõ��ӷ�����ԭ��Ӧ���缫��ӦʽΪ2H++2e-�TH2��������̼����������ʧ���ӷ���������Ӧ���缫��ӦʽΪ2Cl--2e-�TCl2����ͬʱ��Һ�л������������ƣ����Ե�ط�ӦʽΪ 2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+H2��+Cl2����
�ʴ�Ϊ��2Cl--2e-�TCl2����
��2���������A���ܿڲ���H2���ұ�B���ܿڲ���Cl2���Ե���Ϊ���ģ���Ӧװ�õ����ã� ����������˳���ǣ�H��F��G��A��B��D��E��C��
����������˳���ǣ�H��F��G��A��B��D��E��C��
�ʴ�Ϊ��G��F��H��D��E��C��
��3�������ܽ�����������Ϊ�ⵥ�ʣ���ʵ���У�ʢ��KI������Һ�������з�����Ӧ�����ӷ���ʽΪCl2+2I-=I2+2Cl-���ʴ�Ϊ��Cl2+2I-=I2+2Cl-��
��4������2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+H2��+Cl2�����������ƺ������Ĺ�ϵʽ֪��n��NaOH��=2n��H2��=2��$\frac{0.0448L}{22.4L/mol}$=0.004mol��
����ԭ���غ��n��OH-��=0.004mol��c��OH-��=$\frac{0.004mol}{0.05L}$=0.08mol/L���ʴ�Ϊ��0.08 mol•L-1��
���� �����Ե��ԭ��Ϊ���忼�����������ȡ�����ʵļ����֪ʶ�㣬���ݵ��ԭ�������ʵ����ʡ����ʼ�Ĺ�ϵ����������ɣ��ѵ�����������˳����������ȡװ�á�����װ�á��ռ�װ�á�β������װ�������ɣ��Ѷ��еȣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ԭ�Ӱ뾶��Z��Y��X | |
| B�� | ����������Ӧˮ��������ԣ�Z��X | |
| C�� | ����⻯����ȶ��ԣ�W��Z��X | |
| D�� | Y�������ﲻ�������������� |
| A�� | CBr2F2 | B�� | CH3CH2OH | C�� | CH3CH2CH3 | D�� | CH3CH��OH��COOH |
| n��SO32-����n��HSO3-�� | 91��9 | 1��1 | 1��91 |
| pH | 8.2 | 7.2 | 6.2 |
A��c��Na+��=2c��SO32-��+c��HSO3-��
B��c��Na+����c��HSO3-����c��SO32-����c��H+��=c��OH-��
C��c��Na+��+c��H+��=c��SO32-��+c��HSO3-��+c��OH-��
�ڵ�����Һ��pH����ԼΪ6ʱ����������������������ʾ��ͼ���£�
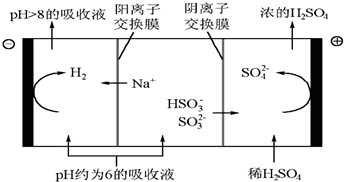
��д��HSO3-�������ŵ�ĵ缫��Ӧʽ��HSO3-+H2O-2e-=SO42-+3H+��
��2����֪��CH4��g��+2O2��g��=CO2 ��g��+2H2O ��l����H=-Q1 kJ/mol
2H2��g��+O2��g��=2H2 O ��g����H=-Q2 kJ/mol
2H2��g��+O2��g��=2H2 O ��l����H=-Q3 kJ/mol��
�����£�ȡ�����4��1�ļ���������Ļ������11.2L�������������ȫȼ�ջָ����£��ų�����Ϊ0.4Q1+0.05Q3��
| A�� |  ������ƿ��ת����Һ | B�� |  �к��ȵIJⶨ | ||
| C�� |  Ũ������ͭ�ķ�Ӧ | D�� | 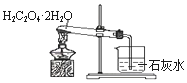 �Ҷ��ᾧ�����ȷֽ� |
| A�� | ��Ԫ��λ��Ԫ�����ڱ��е������ڢ�B�� | |
| B�� | ��Ԫ��λ��s�� | |
| C�� | ��Ԫ�ص�����ϼ�Ϊ+2�� | |
| D�� | ��Ԫ�ػ�̬ԭ����������ߵ���3d���� |
| A�� | 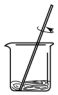 | B�� | 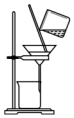 | C�� | 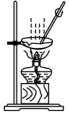 | D�� | 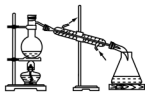 |
| A�� | XΪ��̬��ZΪ��̬ | B�� | XΪ��̬��ZΪ��̬ | C�� | XΪ��̬��ZΪ��̬ | D�� | XΪ��̬��ZΪ��̬ |