��Ŀ����
A��B��C��D��E�����ֶ���������Ԫ�أ����ǵ�ԭ�����������������У�A��Cͬ���壬B��Eͬ���壮��֪BԪ��ԭ�������������Ǵ�����������3����B��E��Ԫ��ԭ�Ӻ���������֮����A��C��Ԫ��ԭ�Ӻ���������֮�͵�2����D��������������������Ӳ�������ش��������⣮
��1��E�����ӽṹʾ��ͼ�� ����B��C�γɺ��зǼ��Լ������ӻ�����ĵ���ʽ�� ��
��2������A��B��Ԫ�صĵ����Ƴɵ�����ȼ�ϵ�أ���������ɴ���ʹ�ã������͵��ʹ�õĵ������ҺΪKOH��Һ�����������ĵ缫��ӦʽΪ ������·����3.01��1022�����ӷ���ת�ƣ��������ĵ�A���ʵ�����Ϊ ��
��3��D�����ܸ�C�����������ˮ�������Һ��Ӧ�����ӷ���ʽΪ ��
��4��D�γɵ�ij�����dz��õľ�ˮ�����仯ѧʽΪ �����ľ�ˮԭ���� ���������ӷ���ʽ��ʾ��
��5��A2E��ȼ���ȡ�H=-a kJ?mol-1��д��X2Eȼ�շ�Ӧ���Ȼ�ѧ����ʽ�� ��
��1��E�����ӽṹʾ��ͼ��
��2������A��B��Ԫ�صĵ����Ƴɵ�����ȼ�ϵ�أ���������ɴ���ʹ�ã������͵��ʹ�õĵ������ҺΪKOH��Һ�����������ĵ缫��ӦʽΪ
��3��D�����ܸ�C�����������ˮ�������Һ��Ӧ�����ӷ���ʽΪ
��4��D�γɵ�ij�����dz��õľ�ˮ�����仯ѧʽΪ
��5��A2E��ȼ���ȡ�H=-a kJ?mol-1��д��X2Eȼ�շ�Ӧ���Ȼ�ѧ����ʽ��
���㣺λ�ýṹ���ʵ����ϵӦ��
ר�⣺
������A��B��C��D��E�����ֶ���������Ԫ�أ����ǵ�ԭ����������������֪BԪ��ԭ�������������Ǵ�����������3���������㺬��2�����ӣ�����㺬��6�����ӣ���BΪOԪ�أ�B��Eͬ���壬��EΪSԪ�أ�B��E��Ԫ��ԭ�Ӻ���������֮��Ϊ��8+16=24����A��C��Ԫ��ԭ�Ӻ���������֮��Ϊ12����A��Cͬ���壬��AΪHԪ�ء�CΪNaԪ�أ�D��������������������Ӳ�����D��ԭ����������Na��11����D�ĵ��Ӳ���Ϊ3������������Ϊ3������DΪAlԪ�أ��ݴ˽��н��
���
�⣺A��B��C��D��E�����ֶ���������Ԫ�أ����ǵ�ԭ����������������֪BԪ��ԭ�������������Ǵ�����������3���������㺬��2�����ӣ�����㺬��6�����ӣ���BΪOԪ�أ�B��Eͬ���壬��EΪSԪ�أ�B��E��Ԫ��ԭ�Ӻ���������֮��Ϊ��8+16=24����A��C��Ԫ��ԭ�Ӻ���������֮��Ϊ12����A��Cͬ���壬��AΪHԪ�ء�CΪNaԪ�أ�D��������������������Ӳ�����D��ԭ����������Na��11����D�ĵ��Ӳ���Ϊ3������������Ϊ3������DΪAlԪ�أ�
��1��EΪSԪ�أ������ӵĺ����������Ϊ18�������ӽṹʾ��ͼΪ��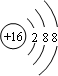 ����B��C�γɺ��зǼ��Լ������ӻ�����Ϊ�������ƣ���������Ϊ���ӻ���������ʽΪ��
����B��C�γɺ��зǼ��Լ������ӻ�����Ϊ�������ƣ���������Ϊ���ӻ���������ʽΪ�� ��
��
�ʴ�Ϊ�� ��
�� ��
��
��2��AΪH��BΪOԪ�أ�����H��O��Ԫ�صĵ��������������Ƴɵ�����ȼ�ϵ�أ���������ɴ���ʹ�ã������͵��ʹ�õĵ������ҺΪKOH��Һ���������������õ������������������ӣ���缫��ӦʽΪ��O2+4e-+2H2O=4OH-������·����3.01��1022�����ӷ���ת�ƣ����ӵ����ʵ���Ϊ0.05mol�����ݵ����غ㣬������A���ʵ�����Ϊ��2g/mol��
=0.05g��
�ʴ�Ϊ��O2+4e-+2H2O=4OH-��0.05g��
��3��D����ΪAl��C�����������ˮ����Ϊ�������ƣ���������������Һ��Ӧ����ƫ�����ƺ���������Ӧ�����ӷ���ʽΪ��2Al+2OH-+2H2O=2AlO2-+3H2����
�ʴ�Ϊ��2Al+2OH-+2H2O=2AlO2-+3H2����
��4��Al2��SO4��3������ˮ����������ˮ�����������������壬���ڽ�������ϴ�������ˮ���������ʣ����ӷ�Ӧ����ʽΪ��Al3++3H2O?Al��OH��3�����壩+3H+��
�ʴ�Ϊ��Al2��SO4��3��Al3++3H2O?Al��OH��3�����壩+3H+��
��5��A2EΪ���⣬H2S��ȼ���ȡ�H=-a kJ?mol-1��H2Sȼ�շ�Ӧ���Ȼ�ѧ����ʽΪ��H2S��g��+
O2��g��=SO2��g��+H2O��l����H=-a kJ?mol-1��
�ʴ�Ϊ��H2S��g��+
O2��g��=SO2��g��+H2O��l����H=-a kJ?mol-1��
��1��EΪSԪ�أ������ӵĺ����������Ϊ18�������ӽṹʾ��ͼΪ��
 ����B��C�γɺ��зǼ��Լ������ӻ�����Ϊ�������ƣ���������Ϊ���ӻ���������ʽΪ��
����B��C�γɺ��зǼ��Լ������ӻ�����Ϊ�������ƣ���������Ϊ���ӻ���������ʽΪ�� ��
���ʴ�Ϊ��
 ��
�� ��
����2��AΪH��BΪOԪ�أ�����H��O��Ԫ�صĵ��������������Ƴɵ�����ȼ�ϵ�أ���������ɴ���ʹ�ã������͵��ʹ�õĵ������ҺΪKOH��Һ���������������õ������������������ӣ���缫��ӦʽΪ��O2+4e-+2H2O=4OH-������·����3.01��1022�����ӷ���ת�ƣ����ӵ����ʵ���Ϊ0.05mol�����ݵ����غ㣬������A���ʵ�����Ϊ��2g/mol��
| 0.05mol |
| 2 |
�ʴ�Ϊ��O2+4e-+2H2O=4OH-��0.05g��
��3��D����ΪAl��C�����������ˮ����Ϊ�������ƣ���������������Һ��Ӧ����ƫ�����ƺ���������Ӧ�����ӷ���ʽΪ��2Al+2OH-+2H2O=2AlO2-+3H2����
�ʴ�Ϊ��2Al+2OH-+2H2O=2AlO2-+3H2����
��4��Al2��SO4��3������ˮ����������ˮ�����������������壬���ڽ�������ϴ�������ˮ���������ʣ����ӷ�Ӧ����ʽΪ��Al3++3H2O?Al��OH��3�����壩+3H+��
�ʴ�Ϊ��Al2��SO4��3��Al3++3H2O?Al��OH��3�����壩+3H+��
��5��A2EΪ���⣬H2S��ȼ���ȡ�H=-a kJ?mol-1��H2Sȼ�շ�Ӧ���Ȼ�ѧ����ʽΪ��H2S��g��+
| 3 |
| 2 |
�ʴ�Ϊ��H2S��g��+
| 3 |
| 2 |
���������⿼��ṹ����λ�ù�ϵӦ�ã���Ŀ�Ѷ��еȣ��ƶ�Ԫ���ǽ���ؼ���ע��Ի���֪ʶ���������գ������漰��֪ʶ��϶ࡢ�ۺ��Խ�ǿ����ֿ�����ѧ���ķ������������������Ӧ�û���֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
��NA��ʾ�����ӵ�������������������ȷ���ǣ�������
| A�����³�ѹ�£�11.2 LCO2������ԭ����Ϊ1.5NA | ||
| B�����³�ѹ�£�48 g O3���е���ԭ����Ϊ3NA | ||
| C����״���£�22.4 L SO3������ԭ����Ϊ4NA | ||
D����״���£�1 Lˮ����������Ϊ
|
����˵����ȷ���ǣ�������
| A�����к��Ȳⶨʵ���У�Ϊȷ��÷�Ӧǰ����²Ӧ���ѷֱ����¶ȵ�����������Һ�������ڸ��ȵ������п��ٻ�ϣ������Ͻ��裬���¶ȼƲ������Һ������¶� |
| B���������ҵĽ���Һ�м����������ˮ����֤I-��ȫ����ΪI2 |
| C�������������������ػ��Һ���ȣ��ٵμ���������Һ���۲��е���ɫ�������ɣ���֤���������к����� |
| D����ϴ��ʽ�ζ���ʱӦ�ӵζ����Ͽڼ���3��5mL��Ҫʢװ������Һ����б��ת���ζ��ܣ�ʹҺ����ʪ���ڱڣ��ٴ��Ͽڵ������ظ�2��3�� |
���ж���Ԫ�ػ�̬ԭ�ӵĵ����Ų�ʽ��Ԫ�صĵ縺�������ǣ�������
| A��1s22s22p2 |
| B��1s22s22p5 |
| C��1s22s22p63s23p4 |
| D��1s22s22p3 |
����˵����ȷ���ǣ�������
A����ϵͳ�������������� 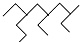 ������Ϊ2��4-���һ�-6-�������� ������Ϊ2��4-���һ�-6-�������� |
| B�������ֶ��Ļ�Ϊͬ���칹�壬����ߵ�ˮ�����һ������ͬ |
| C�������Ӳ֬�ụΪͬϵ�C2H6��C9H20Ҳһ����Ϊͬϵ�� |
D��1molij�л���Ľṹ��ʽΪ 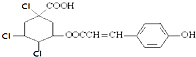 ͨ����ȥ��Ӧ��ȥ1 molHClʱ���ܵõ� 5 �ֲ�ͬ��������������칹�� ͨ����ȥ��Ӧ��ȥ1 molHClʱ���ܵõ� 5 �ֲ�ͬ��������������칹�� |
 ú����Ҫ���ܣ�Ҳ������������Ʒ����Ҫԭ�ϣ�������ѧ֪ʶ������������⣺
ú����Ҫ���ܣ�Ҳ������������Ʒ����Ҫԭ�ϣ�������ѧ֪ʶ������������⣺