��Ŀ����
�ɼ������ӻ�������ɵĻ����������������е������֣�K+��Cl-��NH4+��Mg2+��Ba2+��CO32-��SO42-�����û��������ˮ��ó�����Һ����ȡ����100mL����Һ�ֱ��������ʵ�飺
��ش��������⣺
��1������ʵ��1��3�ж�ԭ�������һ�������ڵ������� ��
��2����ȷ����Һ��һ�����ڵ������Ӽ������ʵ���Ũ�ȣ��ɲ���������
��3����ȷ��K+ �Ƿ���ڣ� ���жϵ������� ��
| ʵ����� | ʵ������ | ʵ���� |
| 1 | ��AgNO3 ��Һ | �а�ɫ�������� |
| 2 | ������NaOH ��Һ������ | �ռ�������1.12L��������ɱ�״���µ������ |
| 3 | ������BaCl2 ��Һ�������ó�������ϴ�ӡ������������������м�����ϡ���ᣬȻ�������� | ��һ�γ�������Ϊ6.27g���ڶ��γ�������Ϊ2.33g |
��1������ʵ��1��3�ж�ԭ�������һ�������ڵ�������
��2����ȷ����Һ��һ�����ڵ������Ӽ������ʵ���Ũ�ȣ��ɲ���������
| �����ӷ��� | ���ʵ���Ũ�ȣ�mol?L-1�� |
���㣺���������ӵļ���,���������ӵļ���
ר�⣺���ʼ��������
��������1����̼���������������Ȼ������ǰ�ɫ���������ʵ��1��ȷ���Ƿ��������Ӳ���ȷ��������ʵ��2��֪��������ӣ�����ʵ��3��֪����һ����̼�ᱵ�����ᱵ������Һ��һ������CO32-��SO42-������Һ��һ������Ba2+��Mg2+��
��2��̼�ᱵ���������ᣬ���ᱵ���������ᣬ��˼��������ʣ��2.33g����ΪBaSO4��������̼�غ㼴���������Һ��c��SO42-������CO32-�������ʵ���Ũ�ȣ�
��3�����ǰ����������ݿ�֪����Һ�п϶����ڵ�������NH4+��CO32-��SO42-�������㣬NH4+�����ʵ���Ϊ0.05 mol��CO32-��SO42-�����ʵ����ֱ�Ϊ0.02 mol��0.01 mol�����ݵ���غ��K+һ�����ڣ�
��2��̼�ᱵ���������ᣬ���ᱵ���������ᣬ��˼��������ʣ��2.33g����ΪBaSO4��������̼�غ㼴���������Һ��c��SO42-������CO32-�������ʵ���Ũ�ȣ�
��3�����ǰ����������ݿ�֪����Һ�п϶����ڵ�������NH4+��CO32-��SO42-�������㣬NH4+�����ʵ���Ϊ0.05 mol��CO32-��SO42-�����ʵ����ֱ�Ϊ0.02 mol��0.01 mol�����ݵ���غ��K+һ�����ڣ�
���
�⣺��1��̼���������������Ȼ������ǰ�ɫ���������ʵ��1�õ�������ȷ�����Ȼ�������ʵ��1��Cl-�Ƿ���ڵ��ж��ǣ�����ȷ��������ʵ��2��֪��������ӣ�����ʵ��3��֪����һ����̼�ᱵ�����ᱵ������Һ��һ������CO32-��SO42-��̼�ᱵ��̼��þ�����ᱵ�ȶ��Dz�����ˮ�ij������ʿ���֪��Һһ�������ڵ������ǣ�Ba2+��Mg2+���ʴ�Ϊ��Ba2+��Mg2+��
��2����ϣ�1���з�����֪��Һ��һ�����е�������ΪCO32-��SO42-����̼�ᱵ���������ᣬ���ᱵ�������������֪���������ʣ��2.33g����ΪBaSO4���������غ��֪��Һ��n��SO42-��=
=0.01mol��c��SO42-��=
=0.1mol/L��6.27g������̼�ᱵ������Ϊ6.27g-2.33g=3.94g������̼�غ��֪��Һ��n��CO32-��=
=0.02mol��c��CO32-��=
=0.2mol/L��
�ʴ�Ϊ��SO42-��0.1mol/L��CO32-��0.2mol/L��
��3����Һ�п϶����ڵ�������NH4+��CO32-��SO42-�������㣬NH4+�����ʵ���Ϊ
=0.05 mol�����ã�2���з����������֪CO32-��SO42-�����ʵ����ֱ�Ϊ0.02 mol��0.01 mol�����ݵ���غ㣬n��+��=n��NH4+��=0.05mol��n��-��=2n��CO32-��+2n��SO42-��=0.06mol��������һ�����ڣ�
�ʴ�Ϊ�����ڣ�ͨ��ʵ���֪��Һ��һ�����ڵ�������NH4+��SO42-��CO32-�������㣬笠����ӵ����ʵ���Ϊ0.05mol��̼�������Ϊ0.01mol�����������Ϊ0.02mol�����ݵ���غ㣬n��+��=n��NH4+��=0.05mol��n��-��=2n��CO32-��+2n��SO42-��=0.06mol��������һ�����ڣ�
��2����ϣ�1���з�����֪��Һ��һ�����е�������ΪCO32-��SO42-����̼�ᱵ���������ᣬ���ᱵ�������������֪���������ʣ��2.33g����ΪBaSO4���������غ��֪��Һ��n��SO42-��=
| 2.33g |
| 233g/mol |
| 0.01mol |
| 0.1L |
| 3.94g |
| 197g/mol |
| 0.02mol |
| 0.1L |
�ʴ�Ϊ��SO42-��0.1mol/L��CO32-��0.2mol/L��
��3����Һ�п϶����ڵ�������NH4+��CO32-��SO42-�������㣬NH4+�����ʵ���Ϊ
| 1.12L |
| 22.4L/mol |
�ʴ�Ϊ�����ڣ�ͨ��ʵ���֪��Һ��һ�����ڵ�������NH4+��SO42-��CO32-�������㣬笠����ӵ����ʵ���Ϊ0.05mol��̼�������Ϊ0.01mol�����������Ϊ0.02mol�����ݵ���غ㣬n��+��=n��NH4+��=0.05mol��n��-��=2n��CO32-��+2n��SO42-��=0.06mol��������һ�����ڣ�
���������⿼�����Ӽ��飬���ʱ���϶������㣬˼ά����������ֵ���о���������Ľⷨ�������⾳��

��ϰ��ϵ�д�
�����Ŀ
�����£�������Һ�е���Ũ�ȹ�ϵ��ȷ���ǣ�������
| A��������ˮ�м������NaOH��[Na+]=[Cl-]+[ClO-]+[OH-] |
| B��pH=8.3��NaHCO3��Һ��[Na+]��[HCO3-]��[CO32-]��[H2CO3] |
| C��pH=11�İ�ˮ��pH=3������������ϣ�[Cl-]=[NH4+]��[OH-]=[H+] |
| D��0.2mol?L-1CH3COOH��Һ��0.1mol?L-1NaOH��Һ�������ϣ�2[H+]-2[OH-]=[CH3COO-]-[CH3COOH] |
�ں��¡����ݵ������н��з�Ӧ��2HI?H2+I2������ӦΪ���ȷ�Ӧ������Ӧ���Ũ����0.1mol/L����0.06mol/L��Ҫ20s����ô��0.06mol/L����0.036mol/L����ʱ��Ϊ��������
| A������10 s |
| B������12 s |
| C������12 s |
| D����12 s |
����ʳ�õĴ����ղ�����������Ҫ�ɷ��ǣ�������
| A�������� | B������ |
| C�������� | D����֬ |
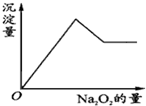 ��һ����Һ�����ܺ���Al3+��Fe3+��K+��NH4+��Mg2+��Cu2+�������е�һ�ֻ��֣��ּ���Na2O2��ĩֻ����ɫ��ζ������ų�����ͬʱ������ɫ����������Na2O2���������ɰ�ɫ��������֮��Ĺ�ϵ��ͼ��
��һ����Һ�����ܺ���Al3+��Fe3+��K+��NH4+��Mg2+��Cu2+�������е�һ�ֻ��֣��ּ���Na2O2��ĩֻ����ɫ��ζ������ų�����ͬʱ������ɫ����������Na2O2���������ɰ�ɫ��������֮��Ĺ�ϵ��ͼ��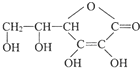
 ���Ǵ����к����ḻ��һ��Ԫ�أ���ش����к����仯�����������⣺
���Ǵ����к����ḻ��һ��Ԫ�أ���ش����к����仯�����������⣺