��Ŀ����
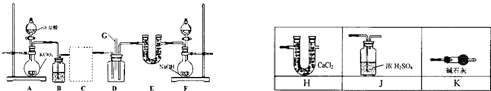
 �Ҵ��ķ������ΪC2H6O���ֲ���ʵ�鷽��ȷ��һ���Ҵ��������м�����ԭ���ܱ����û�������װ����ͼ��ʾ��
�Ҵ��ķ������ΪC2H6O���ֲ���ʵ�鷽��ȷ��һ���Ҵ��������м�����ԭ���ܱ����û�������װ����ͼ��ʾ��ʵ�����Ҫ���ǣ�ȫ��ʵ��װ�ñ��뱣֤���������ã�װ��AҪ�����ʽ�ζ����м�����ˮ�Ҵ����������������ڶ��ױ������ױ����������Ʒ�Ӧ���з�ɢ��������С�Ŀ���������B��C�IJ�����������Ҫ����ˮ���ֲ��ܷ����������ӵζ����л���������ˮ�Ҵ�V mL��ͬʱ�ɿ����ɼУ�������ˮ��Bѹ��C������ƿ�ָ������º��C��������ˮ�����ΪV1 mL�����������������ɱ�״�����������ش��������⣺
��1�����ʵ�鿪ʼǰ����B��C�IJ�������δ����ˮ����ʵ������
��2���������ͨ�ƾ�������ˮ�Ҵ���ʵ�飬������ʵ������
��3������A��B�ĵ��ܵ���δ�ֱ����Ҫ�൱������������
��4���ѽ����Ʒ�ĩ��ɢ��С������Ŀ����
��5������ˮ�Ҵ����ܶ�Ϊ�� g/mL�����Ҵ������б����û�����ԭ�Ӹ���Ϊ
���㣺����ʵ�鷽�������
ר�⣺
�������ζ����л���������ˮ�Ҵ�V mL��������B�еĿ���ΪVmL�����C��������ˮ�����ΪV1 mL�������B�����������ΪV��H2��=��V1-V�� mL������n����H2��=
=
mol��
�Ҵ������ʵ�����n��C2H6O��=
mol������ÿ���Ҵ������пɱ��û�����ԭ����N��H��=
=
��
��1�����ʵ�鿪ʼǰ����B��C�IJ�������δ����ˮ�������C�е�ˮƫ�٣�����C��������ˮ�����ƫС���������ϼ���ʽ������
��2����ͨ�ƾ��к���ˮ��ˮ����������ƫ�ࣻ
��3���Ҵ��ӷ���
��4�������Ʒ�ĩ��ɢ��С���������������Ҵ��ĽӴ������
��5���������Ϸ����ش�
| (V1-V)��10-3 |
| 22.4 |
| V1-V |
| 22400 |
�Ҵ������ʵ�����n��C2H6O��=
| V�� |
| 46 |
| ||
|
| 23(V1-V) |
| 5600��V |
��1�����ʵ�鿪ʼǰ����B��C�IJ�������δ����ˮ�������C�е�ˮƫ�٣�����C��������ˮ�����ƫС���������ϼ���ʽ������
��2����ͨ�ƾ��к���ˮ��ˮ����������ƫ�ࣻ
��3���Ҵ��ӷ���
��4�������Ʒ�ĩ��ɢ��С���������������Ҵ��ĽӴ������
��5���������Ϸ����ش�
���
�⣺�ζ����л���������ˮ�Ҵ�V mL��������B�еĿ���ΪVmL�����C��������ˮ�����ΪV1 mL�������B�����������ΪV��H2��=��V1-V�� mL������n����H2��=
=
mol��
�Ҵ������ʵ�����n��C2H6O��=
mol������ÿ���Ҵ������пɱ��û�����ԭ����N��H��=
=
��
��1�����ʵ�鿪ʼǰ����B��C�IJ�������δ����ˮ�������C�е�ˮƫ�٣�����C��������ˮ�����ƫС����V1ƫС����
��֪��ʵ������ƫС��
�ʴ�Ϊ��ƫС��
��2����ͨ�ƾ��к���ˮ��ˮ����������ƫ�࣬�����C�е�ˮƫ�࣬���C��������ˮ�����ƫ��V1ƫ����
��֪��ʵ������ƫ��
�ʴ�Ϊ��ƫ��
��3���Ҵ��ӷ�������A��B�ĵ��ܵ���δ�ֱ����Ҫ�൱��������ʹ�Ҵ���������������ʹ�Ҵ�ȫ�����Ʒ�Ӧ��
�ʴ�Ϊ��ʹ�Ҵ���������������
��4�������Ʒ�ĩ��ɢ��С���������������Ҵ��ĽӴ������ʹ�Ҵ���ַ�Ӧ��
�ʴ�Ϊ��ʹ�Ҵ���Ӧ��ȫ��
��5���������Ϸ�����֪��ÿ���Ҵ������пɱ��û�����ԭ����N��H��=
���ʴ�Ϊ��
��
| (V1-V)��10-3 |
| 22.4 |
| V1-V |
| 22400 |
�Ҵ������ʵ�����n��C2H6O��=
| V�� |
| 46 |
| ||
|
| 23(V1-V) |
| 5600��V |
��1�����ʵ�鿪ʼǰ����B��C�IJ�������δ����ˮ�������C�е�ˮƫ�٣�����C��������ˮ�����ƫС����V1ƫС����
| 23(V1-V) |
| 5600��V |
�ʴ�Ϊ��ƫС��
��2����ͨ�ƾ��к���ˮ��ˮ����������ƫ�࣬�����C�е�ˮƫ�࣬���C��������ˮ�����ƫ��V1ƫ����
| 23(V1-V) |
| 5600��V |
�ʴ�Ϊ��ƫ��
��3���Ҵ��ӷ�������A��B�ĵ��ܵ���δ�ֱ����Ҫ�൱��������ʹ�Ҵ���������������ʹ�Ҵ�ȫ�����Ʒ�Ӧ��
�ʴ�Ϊ��ʹ�Ҵ���������������
��4�������Ʒ�ĩ��ɢ��С���������������Ҵ��ĽӴ������ʹ�Ҵ���ַ�Ӧ��
�ʴ�Ϊ��ʹ�Ҵ���Ӧ��ȫ��
��5���������Ϸ�����֪��ÿ���Ҵ������пɱ��û�����ԭ����N��H��=
| 23(V1-V) |
| 5600��V |
| 23(V1-V) |
| 5600��V |
���������⿼����ʵ�鷽����ƺ�ʵ���������������ڿ���ѧ��Ӧ��ԭ��֪ʶ����Ƚ�İ����ʵ������������Ϳ���ѧ����˼ά���̶ܳȣ�ע�������������װ�õ����á�ҩƷ�Ĵ����ͻ�ѧ��������⣬��Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �������Ӳ�ϵ�д�
�������Ӳ�ϵ�д� ���ɿ��õ�Ԫ����AB��ϵ�д�
���ɿ��õ�Ԫ����AB��ϵ�д�
�����Ŀ
���ڻ�ѧ��Ӧ������жϣ�����˵������ȷ���ǣ�������
| A���¶ȡ�ѹǿһ��ʱ���ؼ�С�ķ��ȷ�Ӧһ�����Է����� |
| B���¶ȡ�ѹǿһ��ʱ�����оݹ�ͬ����һ����ѧ��Ӧ�Է����еķ��� |
| C����Ӧ�ʱ��Ǿ�����Ӧ�ܷ��Է����е�Ψһ���� |
| D��������ܽ�������ر��� |
����Ȳ����������������ɵĻ�����У���ú�̼Ԫ�ص���������Ϊ72%������������Ԫ�ص���������Ϊ��������
| A��24.9% | B��22% |
| C��19.6% | D�������� |
����˵����ȷ���ǣ�������
| A������������Һ�������Һ��Ӧ�����ӷ���ʽΪ��H++OH-�TH2O |
| B����ͬ���ʵ���Ũ�������백ˮ��Һ�������Ϻ�c��CI-����c��NH4+����c��OH-����c��H+�� |
| C�������£���ˮ�������c��H+��=1��10-12mol/L����Һ�У�Fe2+��NO3-��Na+��SO42-һ���ܴ������� |
| D����������Һ���ɺ�õ����������壬�Ȼ�����Һ���ɺ�õ������������� |
�������ӷ���ʽ��д��ȷ���ǣ�������
| A��0.1mol/LNH4CN��Һ�ʼ��ԣ�������Һ��ֻ���ڣ�CN-+H2O?HCN+OH- | ||
| B��CuSO4��Һ�м��������ˮ��Ӧ��Cu2++4NH3?H2O=[Cu��NH3��4]2++H2O | ||
| C��Fe��OH��3��ǿ������ᷴӦ��Fe��OH��3+3H+=Fe3++3H2O | ||
D����ȩ������������Һ��Ӧ��HCHO+2[Ag��NH3��2]++2OH-
|
�й�ҩ���������ȷ���ǣ�������
| A���˹��ϳ�ҩ��ֻ�а�˾ƥ�� |
| B��1853�꣬�¹��ݶ�ҩ���Ļ�ѧ�һ������ϳ�������ˮ���� |
| C����ù�����˹��ϳ�ҩ���е�����һ�� |
| D������ҩ���˺���̼�����ơ��������������������⣬�����е�ζ�����ϼ� |
 ij���������ᷴӦʱ������H2�����������Ľ������ʵع�ϵ��ͼ��ʾ���˽��������ǣ�������
ij���������ᷴӦʱ������H2�����������Ľ������ʵع�ϵ��ͼ��ʾ���˽��������ǣ�������| A����п | B������ |
| C��þп�Ͻ� | D��ͭ���Ͻ� |