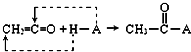��Ŀ����
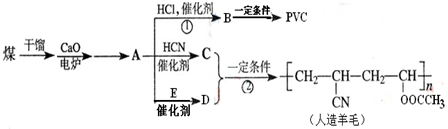
������������ʯ�ͼ۸����ǣ���úΪԭ���Ʊ�һЩ������Ʒ��ǰ���ֱ����ã���ͼ����úΪԭ������������ϩ��PVC����������ë�ĺϳ�·�ߣ�
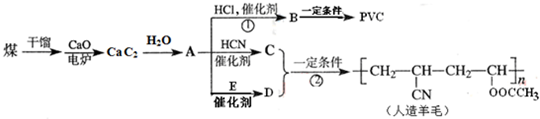
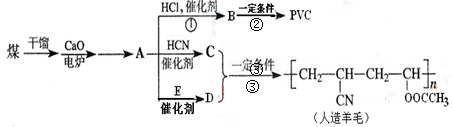
��ش��������⣺
��1����������A�Ľṹ��ʽ��______��
��2��A��C��A��D������Ӧ��ԭ�������ʶ���100%�����ǵķ�Ӧ������______��C�Ľṹ��ʽ��______��E�Ľṹ��ʽ��______��
��3��д��B��PVC�Ļ�ѧ��Ӧ����ʽ______��
��4����D��Ϊͬ���칹���ҿɷ�������ˮ���������______�֣���������״�������д������һ�ֵĽṹ��ʽ______��

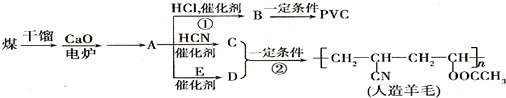
��ش��������⣺
��1����������A�Ľṹ��ʽ��______��
��2��A��C��A��D������Ӧ��ԭ�������ʶ���100%�����ǵķ�Ӧ������______��C�Ľṹ��ʽ��______��E�Ľṹ��ʽ��______��
��3��д��B��PVC�Ļ�ѧ��Ӧ����ʽ______��
��4����D��Ϊͬ���칹���ҿɷ�������ˮ���������______�֣���������״�������д������һ�ֵĽṹ��ʽ______��
CaC2��ˮ��Ӧ����������������Ȳ����ת����ϵ��֪AΪCH��CH��CH��CH��HCl�����ӳɷ�Ӧ����CH2�TCHCl��CH2�TCHCl�����Ӿ۷�Ӧ���ɾ�����ϩ

����BΪCH2�TCHCl��PVCΪ

��CH��CH��HCN�����ӳɷ�Ӧ����CH2�TCHCN��CH2�TCHCN��Dͨ���Ӿ۷�Ӧ�ϳ�������ë����DΪCH2=CHOOCCH3����Ȳ��E��Ӧ����CH2=CHOOCCH3����EΪCH3COOH��
��1��������������֪��A�Ľṹ��ʽ��CH��CH��
�ʴ�Ϊ��CH��CH��
��2��A��C��A��D��Ӧ��ԭ�������ʶ���100%�����ǵķ�Ӧ�����Ǽӳɷ�Ӧ��������������֪��C�Ľṹ��ʽΪH2C=CH-CN��E�Ľṹ��ʽ��CH3COOH��
�ʴ�Ϊ���ӳɷ�Ӧ��H2C=CH-CN��CH3COOH��
��3��B��PVC��CH2�TCHCl�����Ӿ۷�Ӧ���ɾ�����ϩ

����Ӧ����ʽΪ��nH2C=CHCl

��
�ʴ�Ϊ��nH2C=CHCl

��
��4����CH2=CHOOCCH3 ��Ϊͬ���칹���ҿɷ�������ˮ������ʣ������ʺ�����������������״����������ܽṹ�У�CH2=CHCOOCH3��CH2=CHCH2OOCH��CH3CH=CHOOCH��CH2=C��CH3��OOCH��
�ʴ�Ϊ��4��CH2=CHCOOCH3 ��CH2=CHCH2OOCH��CH3CH=CHOOCH��CH2=C��CH3��OOCH��

����BΪCH2�TCHCl��PVCΪ

��CH��CH��HCN�����ӳɷ�Ӧ����CH2�TCHCN��CH2�TCHCN��Dͨ���Ӿ۷�Ӧ�ϳ�������ë����DΪCH2=CHOOCCH3����Ȳ��E��Ӧ����CH2=CHOOCCH3����EΪCH3COOH��
��1��������������֪��A�Ľṹ��ʽ��CH��CH��
�ʴ�Ϊ��CH��CH��
��2��A��C��A��D��Ӧ��ԭ�������ʶ���100%�����ǵķ�Ӧ�����Ǽӳɷ�Ӧ��������������֪��C�Ľṹ��ʽΪH2C=CH-CN��E�Ľṹ��ʽ��CH3COOH��
�ʴ�Ϊ���ӳɷ�Ӧ��H2C=CH-CN��CH3COOH��
��3��B��PVC��CH2�TCHCl�����Ӿ۷�Ӧ���ɾ�����ϩ

����Ӧ����ʽΪ��nH2C=CHCl
| ���� |

��
�ʴ�Ϊ��nH2C=CHCl
| ���� |

��
��4����CH2=CHOOCCH3 ��Ϊͬ���칹���ҿɷ�������ˮ������ʣ������ʺ�����������������״����������ܽṹ�У�CH2=CHCOOCH3��CH2=CHCH2OOCH��CH3CH=CHOOCH��CH2=C��CH3��OOCH��
�ʴ�Ϊ��4��CH2=CHCOOCH3 ��CH2=CHCH2OOCH��CH3CH=CHOOCH��CH2=C��CH3��OOCH��

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
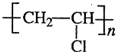
 [��ѧ--�л���ѧ����
[��ѧ--�л���ѧ����
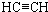 +CH3COOH
+CH3COOH