��Ŀ����
��1��ʵ�����м���CO2�������ķ����ǽ�CO2����ͨ�뱥�͵ij���ʯ��ˮ�У���Ӧ�����ӷ�Ӧ����ʽΪ��2��������ɴ���̫��վ�й������Ա�����ܱյĹ���������Ҫ��������������CO2���ɴ��ڿ���ʹ��һ�����ʣ���������CO2����������������������
��3����Fe��OH��3�У�����������HIϡ��Һ����д���й����ӷ���ʽΪ
��������1�����������������̼��Ӧ���ɰ�ɫ��̼��Ƴ������ݴ˼��������̼�Ƿ���ڣ���������̼���������ɵİ�ɫ����̼����������̼��Ӧ���ɿ����Ե�̼����ƣ�
��2�����ݹ��������������̼��Ӧ����̼���ƺ��������з������ݴ�д����Ӧ�Ļ�ѧ����ʽ��
��3�������������������������Ӿ��������ԣ��ܹ��������������ɵⵥ�ʣ�
��2�����ݹ��������������̼��Ӧ����̼���ƺ��������з������ݴ�д����Ӧ�Ļ�ѧ����ʽ��
��3�������������������������Ӿ��������ԣ��ܹ��������������ɵⵥ�ʣ�
����⣺��1��ʵ�����м���CO2�������ķ����ǽ�CO2����ͨ�뱥�͵ij���ʯ��ˮ�У���Ӧ�����ӷ�Ӧ����ʽΪ��CO2+Ca2++2OH-�TCaCO3+H2O��������ͨ�������CO2���壬̼��Ƴ������������̼��Ӧ����̼����ƣ��������ܽ⣬��Ӧ�����ӷ���ʽΪ��CaCO3+CO2+H2O�TCa2++2HCO3-��
�ʴ�Ϊ��CO2+Ca2++2OH-�TCaCO3+H2O�������ܽ⣻CaCO3+CO2+H2O�TCa2++2HCO3-��
��2���ܹ���������̼ת��������������Ϊ�������ƣ��÷�Ӧ�Ļ�ѧ����ʽΪ��2CO2+2Na2O2�T2NaCO3+O2��
�ʴ�Ϊ���������ƣ�2CO2+2Na2O2�T2NaCO3+O2��
��3����Fe��OH��3�У�����������HIϡ��Һ���������ܹ��������������ɵⵥ�ʣ���Ӧ�����ӷ���ʽΪ��2Fe��OH��3+6H++2I-�T2Fe2++I2+6H2O��
�ʴ�Ϊ��2Fe��OH��3+6H++2I-�T2Fe2++I2+6H2O��
�ʴ�Ϊ��CO2+Ca2++2OH-�TCaCO3+H2O�������ܽ⣻CaCO3+CO2+H2O�TCa2++2HCO3-��
��2���ܹ���������̼ת��������������Ϊ�������ƣ��÷�Ӧ�Ļ�ѧ����ʽΪ��2CO2+2Na2O2�T2NaCO3+O2��
�ʴ�Ϊ���������ƣ�2CO2+2Na2O2�T2NaCO3+O2��
��3����Fe��OH��3�У�����������HIϡ��Һ���������ܹ��������������ɵⵥ�ʣ���Ӧ�����ӷ���ʽΪ��2Fe��OH��3+6H++2I-�T2Fe2++I2+6H2O��
�ʴ�Ϊ��2Fe��OH��3+6H++2I-�T2Fe2++I2+6H2O��
���������⿼���˳�������ļ��顢�Ƶ���Ҫ��������ӷ���ʽ����д��֪ʶ����Ŀ�Ѷ��еȣ�ע�����ճ�������ļ��鷽������ȷ���ӷ���ʽ����д����������������ѧ�����Ӧ����ѧ֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
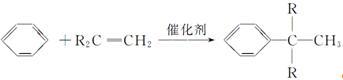 ��R�������
��R�������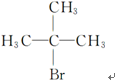
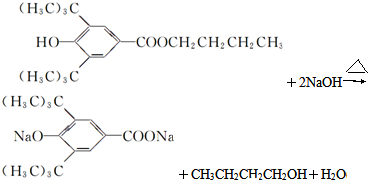




 ��R�������
��R�������