��Ŀ����
10��������ͭ�Ǵ���ˮ�潢ͧ����Ϳ�����Ҫԭ�ϣ���������Һ�������绯��Ӧ��ijС��ͨ���������ϣ����������о���I��ʵ���ҿ��������Ƶ�������ͭ����Һ�������Ǽ�����ȡCu2O��
��1��ʵ�����ô˷�����ȡ���������Cu2O���壬��Ҫ�IJ����������Թܡ��ƾ��ơ��ձ�
�⣬����Ҫ©������������
II��������Ʒ���Ƿ���CuO
����1�����Ƶõ�Cu2O��Ʒ������ϡ���ᷴӦ��
��1����ͬѧ��Ϊ����Һ��Ϊ��ɫ����˵����Ʒ�к���CuO���ʣ���ͬѧ��Ϊ�����۲��������û�ѧ�������ԭ��Cu2O+H2SO4=CuSO4+Cu+H2O��
��2����ͬѧͨ����˼����Ϊ���������Լ����Ϊ�����ⶨ����ȷ����Ʒ���Ƿ���CuO���ʣ�Ӧ������������Cu2O��Ʒ����������Ӧ��ʣ����壨Cu����������
����2����ͬѧ��Ϊѡ��������װ�ã�����ҩƷ�����������ж���ʵ�飬�Ӷ�ȷ����Ʒ���Ƿ�������ͭ��
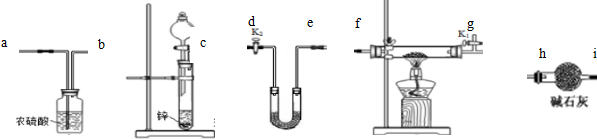
���3��װ�õ�����˳��Ϊcabfgdeh��ʵ�鿪ʼǰ���Ž�װ���п������ſ�������������K1��K2Ӧ��ε��ڴ�K1���ر�K2��װ�ø�����м�ʯ�ҵ������Ƿ�ֹ�����е�ˮ����U�ιܣ�
��4������������Ϊ8.00g����ַ�Ӧ��U������1.08g��Ӳ�ʲ�����������ֻ�к�ɫ����ʣ�࣬�Լ���ԭ������������ͭ����������Ϊ0.90���������С�������λ����
��5����װ�������װ�ô���һ��ȱ�ݣ��Ľ���ʩΪ��װ������һ���ƾ��ƣ���ȼβ����
���� ��1��������������������ͭ��Ӧ��Ҫ���ȣ�����������ͭ��������Ҫ�������˲������з��룻
������Ʒ���Ƿ���CuO������Ʒ���ᷴӦ����ͭ���ӣ������ж��Ƿ�������ͭ����Ϊ������ͭҲ�����ᷴӦ����ͭ���ӣ�����ͨ�������ķ����жϣ����ⶨ������Ʒ������������Ʒ��������Ԫ�ص�����������ͨ���з�����ķ����������Ʒ������������ͭ��
����1����1��������ͭҲ�����ᷢ��������ԭ��Ӧ��������ͭ����Һ����ɫ��ͬʱ����Cu���ʣ�
��2�������Լ����Ϊ�����ⶨ����ȷ����Ʒ���Ƿ���CuO���ʣ�Ӧ������������������ͭ��Ʒ�����ͷ�Ӧ����ͭ��������ͨ����ѧ����ʽ���������
����2����3������ʵ��װ��ͼ��֪��ʵ������п��ϡ���ᷴӦ������������Ũ�������������������������ͭ�������ﷴӦ�û���Cu�����ɵ�ˮ��U�ι��м�ʯ�����գ������θ������еļ�ʯ�ҷ�ֹ�����е�ˮ����U�ιܣ�Ӱ��ˮ�����ⶨ���ݴ˴��⣻
��4������Ԫ���غ㣬����ˮ������ȷ����������Ԫ�ص��ʣ��з�����ɼ�����������ɣ�����ȷ���ʷ�����
��5�������ǿ�ȼ�����壬����ֱ���ŷŵ������У�Ҫ�þƾ��Ƶ�ȼ��
��� �⣺��1��������������������ͭ��Ӧ��Ҫ���ȣ������貣������Ϊ�Թܡ��ƾ��ƣ��������ɵ�������ͭ��������Ҫ���й��˲��������貣������Ϊ©�������������ձ���
�ʴ�Ϊ��©������������
������Ʒ���Ƿ���CuO������Ʒ���ᷴӦ����ͭ���ӣ������ж��Ƿ�������ͭ����Ϊ������ͭҲ�����ᷴӦ����ͭ���ӣ�����ͨ�������ķ����жϣ����ⶨ������Ʒ������������Ʒ��������Ԫ�ص�����������ͨ���з�����ķ����������Ʒ������������ͭ��
����1����1��������ͭҲ�����ᷢ��������ԭ��Ӧ��������ͭ����Һ����ɫ��ͬʱ����Cu���ʣ���Ӧ�Ļ�ѧ����ʽΪ��Cu2O+H2SO4=CuSO4+Cu+H2O��
�ʴ�Ϊ��Cu2O+H2SO4=CuSO4+Cu+H2O��
��2�������Լ����Ϊ�����ⶨ����ȷ����Ʒ���Ƿ���CuO���ʣ�Ӧ������������Cu2O��Ʒ����������Ӧ��ʣ����壨Cu����������
�ʴ�Ϊ��Cu2O��Ʒ����������Ӧ��ʣ����壨Cu����������
����2����3������ʵ��װ��ͼ��֪��ʵ������п��ϡ���ᷴӦ������������Ũ�������������������������ͭ�������ﷴӦ�û���Cu�����ɵ�ˮ��U�ι��м�ʯ�����գ������θ������еļ�ʯ�ҷ�ֹ�����е�ˮ����U�ιܣ�Ӱ��ˮ�����ⶨ������װ�õ�����˳��Ϊcabfgdeh��ʵ�鿪ʼǰ���Ž�װ���п������ſ�������������K1��K2Ӧ��K1���ر�K2��
�ʴ�Ϊ��cabfgdeh����K1���ر�K2����ֹ�����е�ˮ����U�ιܣ�
��4������������Ϊ8.00g����ַ�Ӧ��U������1.08g����ˮ������Ϊ1.08g�����������Ԫ�ص����ʵ���Ϊ0.06mol����ԭ������������ͭ�����ʵ���Ϊxmol������ͭ�����ʵ���Ϊymol����$\left\{\begin{array}{l}{x+y=0.06}\\{144x+80y=8.0}\end{array}\right.$�����x=0.05��y=0.01��������������Ϊ $\frac{0.05��144}{8.0}$=0.90��
�ʴ�Ϊ��0.90��
��5�������ǿ�ȼ�����壬����ֱ���ŷŵ������У�Ҫ�þƾ��Ƶ�ȼ�����ԸĽ���ʩΪ��װ������һ���ƾ��ƣ���ȼβ����
�ʴ�Ϊ����װ������һ���ƾ��ƣ���ȼβ����
���� ���⿼��ʵ���Ʊ��������Ƕ�ѧ���ۺ������Ŀ��飬���ؿ���ѧ����ԭ����װ�÷������ۣ��Ѷ��еȣ�

| A�� | KClO3��MnO2�����ȣ� | B�� | KMnO4�����ȣ� | C�� | H2O2��MnO2�� | D�� | HgO�����ȣ� |
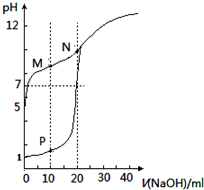
| A�� | �ζ�ʱ�����÷�̪��ָʾ�� | |
| B�� | M��c��HX��-c��X-����c��OH-��-c��H+�� | |
| C�� | ��P���N�����Һ��ϣ�$\frac{{K}_{W}}{c��{H}^{+}��}$��1.0��10-7mol/L | |
| D�� | ��N�����Һ��ͨ��HCl��pH=7��c��Na+����c��HX��=c��Cl-�� |
| A�� | AlCl3��Һ�м���NaOH��Һ | B�� | AlCl3��Һ�м��������İ�ˮ | ||
| C�� | AlCl3��Һ�м���ʯ��ˮ | D�� | Al2O3������������ˮ�� |
| A�� | ��״���£�22.4L������ȫȼ�գ����ɶ�����̼������Ϊ8NA | |
| B�� | ��״���£�11.2L�嵥�ʺ���NA����ԭ�� | |
| C�� | ���³�ѹ�£�28g��ϩ�ͱ�ϩ�Ļ�����庬�е�ԭ������Ϊ6NA | |
| D�� | 1mol�Ҵ���1mol�����ϳ�ַ�Ӧ��������������������ΪNA |
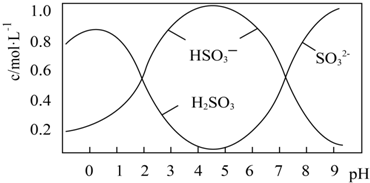
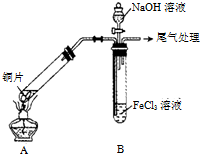 ijУ��ѧ��ȤС��̽��SO2��FeCl3��Һ�ķ�Ӧ������װ����ͼ��ʾ���г���������ȥ����
ijУ��ѧ��ȤС��̽��SO2��FeCl3��Һ�ķ�Ӧ������װ����ͼ��ʾ���г���������ȥ����