��Ŀ����
7�� �£�N2H4���ǻ��ȼ�ϣ�����ӵ����ģ����ͼ��ʾ��������H2O2������Ӧ��N2H4+2H2O2�TN2+4H2O����NAΪ����٤��������ֵ������˵����ȷ���ǣ�������
�£�N2H4���ǻ��ȼ�ϣ�����ӵ����ģ����ͼ��ʾ��������H2O2������Ӧ��N2H4+2H2O2�TN2+4H2O����NAΪ����٤��������ֵ������˵����ȷ���ǣ�������| A�� | 32g N2H4�к��й��ۼ�������Ϊ6NA | |
| B�� | ��״���£�22.4LH2O2������ԭ������Ϊ4NA | |
| C�� | 28g N2�к���������Ϊ7NA | |
| D�� | ��17g H2O2�����Ǽ��Լ���Ŀ��ͬ��N2H4�ķ�������Ϊ0.5NA |
���� A������µ����ʵ�����Ȼ�����1mol���к�5mol���ۼ���������
B�������˫��ˮΪҺ̬��
C�����������к���14�����ӣ�28g���������ʵ���Ϊ1mol������14mol���ӣ�
D��˫��ˮ�����к���1���Ǽ��Լ���17g˫��ˮ�����ʵ���Ϊ0.5mol������0.5mol�Ǽ��Լ���N2H4�����к���1�����Լ����ݴ˼������ҪN2H4�����ʵ�����
��� �⣺A.32g�µ����ʵ���Ϊn=$\frac{32g}{32g/mol}$=1mol��1mol���к�5mol���ۼ������й��ۼ�������Ϊ5NA����A����
B�������˫��ˮΪҺ̬���ʲ��ܸ�������Ħ����������������ʵ����;��е�ԭ����������B����
C.28g N2�����ʵ���Ϊ��$\frac{28g}{28g/mol}$=1mol��1mol���������к���14mol���ӣ�����������Ϊ14NA����C����
D.17g˫��ˮ�����ʵ���Ϊ0.5mol������0.5mol�Ǽ��Լ���N2H4�����к���1�����Լ�����ҪN2H4�����ʵ���Ϊ0.5mol��N2H4�ķ�������Ϊ0.5NA����D��ȷ��
��ѡD��
���� ���⿼���˰���٤�������ļ������жϣ���Ŀ�Ѷ��еȣ���ȷ��״��������Ħ�������ʹ������Ϊ���ؼ���ע���������ʵ����밢��٤��������Ħ��������֮��Ĺ�ϵ������������ѧ���ķ������������Ӧ��������

| A�� | �ڳ��³�ѹ�£�1mol�������еķ�����ΪNA | |
| B�� | �ڳ��³�ѹ�£�11.2L�������еķ�����Ϊ0.5NA | |
| C�� | 15.6gNa2O2�����CO2��Ӧʱ��ת�Ƶĵ�����ΪNA | |
| D�� | ��4gNaOH����1Lˮ�У��ɵõ�0.1mol/LNaOH��Һ |
| A�� | ��ϩ������7���Ҽ���1���м� | |
| B�� | ��ϩ������3��̼ԭ�Ӷ���sp2�ӻ� | |
| C�� | ��ϩ������3��̼ԭ����ͬһֱ���� | |
| D�� | ��ϩ�����мȴ��ڷǼ��Լ��ִ��ڼ��Լ� |
| ѡ�� | ʵ����� | ���� | ���ͻ���� |
| A | ������AgNO3��Һ���ȵμ�����NaCl��Һ����μ�����KI��Һ | �Ȳ�����ɫ�������ֲ�����ɫ���� | AgI��AgCl������ |
| B | ��������ռ���Ҵ���Һ���� | �����������ʹ���Ը��������Һ��ɫ | ֤������ϩ���� |
| C | ������ˮ����ͨ�����ȵ����� | ���۱�� | ����ˮ�ڸ������ܷ�����Ӧ |
| D | Fe���м��������ϡHNO3����ַ�Ӧ����뼸��KSCN��Һ | ��Һ�ʺ�ɫ | ϡHNO3��Fe����ΪFe3+ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | 1L��Һ�к�0.3NA�������� | B�� | 1L��Һ�к�0.9NA�����ӣ�K+��SO42-�� | ||
| C�� | 2L��Һ�м�����Ũ����1.2mol/L | D�� | 2L��Һ�к�0.6 NA�����ӣ�K+��SO42-�� |
��֪����2NO+Na2O2�T2NaNO2��
��3NaNO2+3HCl�T3NaCl+HNO3+2NO��+H2O��
�����������£�NO��NO2������MnO4-��Ӧ����NO3-��Mn2+��Na2O2��ʹ���Ը��������Һ��ɫ��
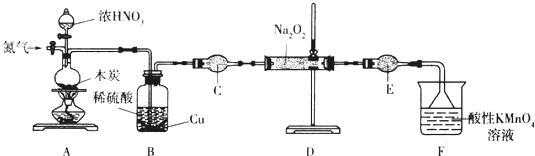
��1������װ��Aǰ����ͨһ��ʱ��N2��Ŀ�����ų�װ���еĿ�����
��2��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪC+4HNO3��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CO2��+4NO2��+2H2O��ʵ�������Bƿ�е���Һ������Ũ������ȴ�ᾧ����������ƣ������˿ɻ��CuSO4•5H2O��
��3������C������Ϊ����ܣ�����ʢ�ŵ�ҩƷΪ��ʯ�ң������ƣ���
��4����ַ�Ӧ����װ��D�в���ķ����ǣ�ȡ�������������Թ��У�����ϡ������Һ�������ݲ��������Թܿ��Ϸ����ֺ���ɫ���壬�������NaNO2��ע���Լ�������
��5��Ϊ�ⶨ�������Ƶĺ�������ȡ4.000g��Ʒ����ˮ���250mL��Һ��ȡ25.00mL��Һ����ƿ�У���0.1000mol•L-1����KMnO4��Һ���еζ���ʵ�������������±���ʾ��
| ����� | 1 | 2 | 3 | 4 |
| KmnO4��Һ���/mL | 20.60 | 20.02 | 20.00 | 19.98 |
a����ʽ�ζ���������ˮϴ����δ�ñ�Һ��ϴ
b����ƿϴ����δ����
c���ζ��յ�ʱ���Ӷ���
�ڸ��ݱ������ݣ��������ù������������Ƶ���������86.25%��
��6����ƺ���ʵ��Ƚ�0.1mol•L-1NaNO2��Һ��NO2-��ˮ��̶Ⱥ�0.1mol•L-1HNO2��Һ��HNO2�ĵ���̶���Դ�С������Ҫ˵��ʵ�鲽�衢����ͽ��ۣ�������ҩƷ��ѡ��25��C��0.1mol/LHNO2��0.1mol/LNaNO2��Һ�������ϣ����ⶨ��ҺPH��7��˵��HNO2�ĵ���̶ȴ���NO2-���ӵ�ˮ��̶ȣ����ⶨ��ҺpH��7��˵��NO2-����ˮ��̶ȴ���HNO2�ĵ���̶ȣ�
 ��
��