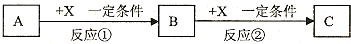
��Ŀ����
15��ij��ȤС����������ͼװ��̽����֤�����Cl2�ͳ�ʪ��Cl2����Ư���ԣ�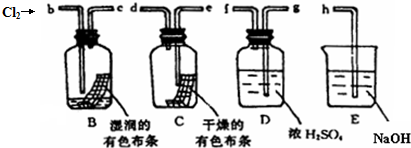
�ش��������⣺
��1�������������������װ������������������ӿڱ�ţ���Cl2��b��c��g��f��e��d ��h��
��2��ͨ��Cl2�Ӽ���ƿB��C�к�ɫ����������ɵó���ʵ������Ǹ����Cl2û��Ư���ԣ���ˮ��Ư���ԣ����з����ķ�Ӧ�Ļ�ѧ����ʽΪCl2+H2O?HCl+HClO��
��3��Ϊ��ȷ֤��HClOʹʪ��ĺ�ɫ������ɫ�����������ɷ֣����������Լ����ٸ������� ��ϡ���� ��������ˮ ����ɫ���� ��ϡ���� ��ʳ��ˮ������ѡ�âܢݣ����Լ���ţ����к���ʵ�飮
��4���ձ�E�з�����Ӧ�Ļ�ѧ����ʽΪCl2+2NaOH�TNaCl+NaClO+H2O��
���� ��1��������˳���ǣ���Ӧװ�á�ϴ��װ�á�����װ�á�����װ�á�β������װ�ã�
��2���������������ʹ�������ɫ������ɫ������ʹʪ�����ɫ������ɫ��������ˮ��Ӧ����HCl��HClO��
��3��Cl2��H2O��Ӧ����HClO����Ӧ�Ļ�ѧ����ʽΪCl2+H2O=HCl+HClO������Ư���Ե�����ΪHClO����ȷ֤���ɼ��������Ƿ���ʹ�첼����ɫ��
��4��������NaO��Ӧ�����Ȼ��ơ��������ƺ�ˮ��
��� �⣺��1��������˳���ǣ���Ӧװ�á�ϴ��װ�á�����װ�á�����װ�á�β������װ�ã����Ա���ʵ��B��C�Ǽ���װ�á�D�Ǹ���װ�á�E��β������װ�ã�����������˳����Cl2��g��f��e��d��b��c��h���ʴ�Ϊ��g��f��e��d��
��2��B��û��ˮ��C����ˮ����������ˮ�õ���ˮ��B����ɫ������ɫ��C����ɫ������ɫ��˵�������Cl2û��Ư���ԣ���ˮ��Ư���ԣ�������ˮ��Ӧ����ʽΪCl2+H2O?HCl+HClO���ʴ�Ϊ�������Cl2û��Ư���ԣ���ˮ��Ư���ԣ�Cl2+H2O?HCl+HClO��
��3��Cl2+H2O?HCl+HClO��������Ҫ���һ��ʵ����һ����ɫ�����ϵμ����ᣬ�����ޱ仯���ų��Ȼ����Ư���ԣ���֤����Ư�����õ������Ǵ����ᣬ
���Ի���ѡ����ɫ������ϡ���ᣬ�ʴ�Ϊ���ܢݣ�
��4��������NaO��Ӧ�����Ȼ��ơ��������ƺ�ˮ����Ӧ����ʽΪCl2+2NaOH�TNaCl+NaClO+H2O���ʴ�Ϊ��Cl2+2NaOH�TNaCl+NaClO+H2O��
���� ���⿼��HClO��Ư���Ե�̽������Ŀ�ѶȲ�����ע����������������ʵIJ����ԣ�ע���йط�Ӧ��ѧ����ʽ�������ӷ���ʽ����д�����շ�Ӧ��ԭ����

 ������ʱͬ����ϰ��ϵ�д�
������ʱͬ����ϰ��ϵ�д� ѧҵ����һ��һ��ϵ�д�
ѧҵ����һ��һ��ϵ�д�| A�� | Һ���������մ������ȣ������������ | |
| B�� | SO2���л�ԭ�ԣ�������Ư�� | |
| C�� | ������۵��Ӳ�ȴ����������뵼����� | |
| D�� | ������Ũ�������ǿ�����ԣ���������ˮ�� |
| A�� | �Ȼ�����Һ�м��������ˮ��Al3++4NH3•H2O�TAlO2-+4NH4++2H2O | |
| B�� | ̼��������ᷴӦ��CO32-+2H+�TCO2��ʮH2O | |
| C�� | ��̼������Һ����������������2CO32-+SO2+H2O�T2HCO3-+SO32- | |
| D�� | �Ȼ�������Һ��ͨ��������Fe2++Cl2�TFe3++2Cl- |
| A�� | 78gNa2O2���庬�е�������Ϊ4NA | |
| B�� | 22.4L��ϩ�к��еķ�����ΪNA | |
| C�� | �ö��Ե缫���NaCl��Һ��ÿ����agNaOHʱ����·��ͨ���ĵ��Ӹ���ΪaNA | |
| D�� | 1molSiO2������Si-O����Ϊ4NA |
| A�� | ������̼�Ľṹʽ��O=C=O | B�� | �����ӵĽṹʾ��ͼ��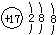 | ||
| C�� | �����ӵĵ���ʽ��Ca2+ | D�� | �廯����ӵĵ���ʽ��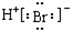 |
| A�� | ������������C2H5OH��������H2SO4�����ӻ������Һ��������ʣ� | |
| B�� | CO2���ǵ���ʣ���NH4Cl����Σ���HNO3�������ᣩ��NaOH��ǿ����ʣ� | |
| C�� | HCl�����ۻ������CH3COOH�����ᣩ��Na2O2�������������CH3I��±������ | |
| D�� | NO2�������������CH3CHO������CO������CH3COOCH3������ |