��Ŀ����
3��̼��һ������Ԫ�أ�̼Ԫ�ص�ѭ���Ե�������������������CO2������������;���ش��������⣺��1����֪�¶�ΪTʱ��
CH4��g��+2H2O=CO2��g��+4H2��g����H=+165KJ•mol
CO��g��+H2O��g��=CO2��g��+H2��g����H=-41KJ•mol
���õ���CO��H2�ں��ʴ����������¿��Ժϳ�CH4�ķ�Ӧ���¶�ΪTʱ���÷�Ӧ���Ȼ�ѧ����ʽΪCO��g��+3H2��g��=CH4��g��+H2O��g����H=-206kJ•mol?1��
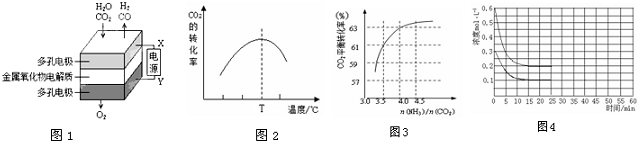
��2���ڹ�̬��������������У����µ��H2O��CO2��������Ʊ�H2��CO��һ���µ���Դ���÷�ʽ������ԭ����ͼ1��ʾ�����ʣ�X�ǵ�Դ�ģ�����������������д������H2�ĵ缫��Ӧʽ��H2O+2e��=H2+O2-��
����CO2��NH3Ϊԭ�Ϻϳ������ǹ̶�������CO2�ijɹ������������غϳ����е���Ҫ��Ӧ�����Է����У��ɱ�ʾ���£�
��Ӧ��2NH3��g��+CO2��g��?NH2CO2NH4��s������H1
��Ӧ��NH2CO2NH4��s��?CO��NH2��2��s��+H2O��l������H2
��Ӧ��2NH3��g��+CO2��g��?CO��NH2��2��s��+H2O��g������H3
��֪��H2O��g��?H2O��l������H4
��ش��������⣺
��1����Ӧ��ġ�H1=��H3+��H4-��H2����������H��ʾ����
��2����Ӧ��ġ�S��0 �����������=����������
��3��ij�о�С��Ϊ̽����Ӧ��Ӱ��CO2ת���ʵ����أ�����������һ��ʱ��ͼ2ΪCO2ת�������¶ȱ仯Ӱ������ߣ�ͼ3�ǰ���ͬ��̼��[n��NH3��/n��CO2��]Ͷ��ʱCO2ƽ��ת���ʵı仯���ߣ�
���¶ȸ���T���CO2ת���ʱ仯������ͼ2��ʾ����ԭ�����¶ȸ���T��ʱ����Ϊ��Ӧ��Ϊ�Է���Ӧ����S��0����Ӧ��H3��0���Ƿ��ȷ�Ӧ���¶�����ƽ�����淽����У�CO2��ƽ��ת���ʽ��ͣ�
��4����ij�㶨�¶��£���NH3��CO2���ʵ���֮�Ȱ�2��1����һ���Ϊ10L���ܱ������У���������������䣬�������������Բ�����ֻ������Ӧ����15min�ﵽƽ�⣬������Ũ�ȵı仯������ͼ4��ʾ��
�������������£���Ӧ���ƽ�ⳣ����ֵΪ250��
��������ƽ����¶Ⱥ�������䣬25minʱ����������г���2mol NH3��1mol CO2����40minʱ���´ﵽƽ�⣬����ͼ4�л���25��50min��CO2��Ũ�ȱ仯�������ߣ�
���� ��1����֪����CH4��g��+2H2O=CO2��g��+4H2��g����H=+165KJ•mol
��CO��g��+H2O��g��=CO2��g��+H2��g����H=-41KJ•mol
�ɸ�˹���ɢ�-�ٿɵ�CO��g��+3H2��g��=CH4��g��+H2O��g�����ݴ˼��㣻
��2��������������������Ӧ������������ԭ��Ӧ��������X�������ĵ缫�����������жϣ���������������ԭ��Ӧ�����ݷ�Ӧ���ϻ��ϼ۵ı仯������
��1����2NH3��g��+CO2��g��?NH2CO2NH4��s������H1
��NH2CO2NH4��s��?CO��NH2��2��s��+H2O��l������H2
��2NH3��g��+CO2��g��?CO��NH2��2��s��+H2O��g������H3
��H2O��g��?H2O��l������H4
�ɸ�˹���ɢ�=��+��-�ڿɵã�
��2����Ӧ��NH2COONH4��s��?CO��NH2��2��s��+H2O��g���������������ķ�Ӧ���ر��S��0��
��3���¶ȸ���T��ʱ����Ϊ��Ӧ��Ϊ�Է���Ӧ����S��0����Ӧ��H3��0���Ƿ��ȷ�Ӧ���¶�����ƽ�����淽����У�CO2��ƽ��ת���ʽ��ͣ�
��4������ͼ���֪NH3��CO2����ʼŨ�ȷֱ�Ϊ0.6mol/L��0.3mol/L��ƽ��ʱNH3��CO2��Ũ�ȷֱ�Ϊ0.2mol/L��0.1mol/L�����뻯ѧƽ�ⳣ��K���㣻
�ڸ��ݵ�Чƽ��֪ʶ����ͼ��25minʱ���������г���2mol������1mol������̼����40minʱ���´ﵽƽ�⣬�������ǵ�Чƽ�⣬������̼��ƽ��Ũ�ȱ��ֲ��䣮
��� �⣺��1����֪����CH4��g��+2H2O=CO2��g��+4H2��g����H=+165KJ•mol
��CO��g��+H2O��g��=CO2��g��+H2��g����H=-41KJ•mol
�ɸ�˹���ɢ�-�ٿɵ�CO��g��+3H2��g��=CH4��g��+H2O��g����H=-206kJ•mol?1��
�ʴ�Ϊ��CO��g��+3H2��g��=CH4��g��+H2O��g����H=-206kJ•mol?1��
��2������ͼʾ֪����X�����ĵ缫����CO�����H2O-CO2������壬������̼�õ���������һ����̼��������ԭ��Ӧ����������������ԭ��Ӧ������X�ǵ�Դ�ĸ�����
��������������ԭ��Ӧ�����H2O-CO2��������Ʊ�H2��CO��������ˮ�е���ԭ�ӵõ���������������H2O+2e-�TH2��+O2-��
�ʴ�Ϊ��������H2O+2e��=H2��+O2-��
��1������2NH3��g��+CO2��g��?NH2CO2NH4��s������H1
��NH2CO2NH4��s��?CO��NH2��2��s��+H2O��l������H2
��2NH3��g��+CO2��g��?CO��NH2��2��s��+H2O��g������H3
��H2O��g��?H2O��l������H4
�ɸ�˹���ɿ�֪��=��+��-�ڣ����H1=��H3+��H4-��H2��
�ʴ�Ϊ����H3+��H4-��H2��
��2����Ӧ��NH2COONH4��s��?CO��NH2��2��s��+H2O��g���������������ķ�Ӧ���ر��S��0��
�ʴ�Ϊ������
��3���¶ȸ���T��ʱ����Ϊ��Ӧ��Ϊ�Է���Ӧ����S��0����Ӧ��H3��0���Ƿ��ȷ�Ӧ���¶�����ƽ�����淽����У�CO2��ƽ��ת���ʽ��ͣ�
�ʴ�Ϊ���¶ȸ���T��ʱ����Ϊ��Ӧ��Ϊ�Է���Ӧ����S��0����Ӧ��H3��0���Ƿ��ȷ�Ӧ���¶�����ƽ�����淽����У�CO2��ƽ��ת���ʽ��ͣ�
��4���ٷ�Ӧ2NH3��g��+CO2��g��?NH2CO2NH4��s������ͼ���֪NH3��CO2����ʼŨ�ȷֱ�Ϊ0.6mol/L��0.3mol/L��ƽ��ʱNH3��CO2��Ũ�ȷֱ�Ϊ0.2mol/L��0.1mol/L������Ũ����Ϊ���䣬K=$\frac{1}{{c}^{2}��N{H}_{3}��•c��C{O}_{2}��}$=$\frac{1}{0��{2}^{2}��0.1}$=250��
�ʴ�Ϊ��250��
��25minʱ���������г���2mol������1mol������̼����40minʱ���´ﵽƽ�⣬�������ǵ�Чƽ�⣬������̼��ƽ��Ũ�ȱ��ֲ��䣬�������£� ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼���Ϊ�ۺϣ��漰��˹���ɵļ��㡢���ص�ԭ������ѧƽ���֪ʶ�㣬��Ŀ�Ѷ��еȣ������ۺ��Խ�ǿ�����ؿ���ѧ����������������

| A�� | 10��1 | B�� | 100��1 | C�� | 1��100 | D�� | ��ȷ�� |
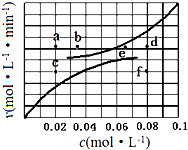 ��1.0L�ܱ������з���0.10molA��g������һ���¶Ƚ������·�Ӧ��
��1.0L�ܱ������з���0.10molA��g������һ���¶Ƚ������·�Ӧ��A��g��?B��g��+C��g����H=+285.1kJ•mol-1
��Ӧʱ�䣨t����������������ѹǿ��P����A�����ʵ���Ũ�ȵ����ݼ��±���
| ʱ��t/h | 0 | 1 | 2 | 4 | 8 | 16 |
| ��ѹǿp/100kPa | 4.91 | 5.58 | 6.32 | 7.31 | 8.54 | 8.54 |
| c��A��/��mol•L-1�� | 0.10 | 0.086 | 0.071 | 0.051 | a | a |
��1��������Ӧ�ܹ��Է����е������Ǹ��£�
��2������ѹǿP����ʼѹǿP0��ʾ��Ӧ��A��ת���ʦ���A���ı���ʽΪ��$\frac{P}{{P}_{0}}$-1����100%�����ݱ������ݼ���ƽ��ʱA��ת����Ϊ73.9%��
��3���������ƽ���ѹ����ѹ=��ѹ�����ʵ����ķ���������ƽ��Ũ�ȱ�ʾ�÷�Ӧ��ƽ�ⳣ���ı���ʽKP=$\frac{{��}^{2}��A��}{[1+����A��]��[1-����A��]}P$���ú�����A����P��ʽ�ӱ�ʾ����
��4������ѹǿP����ʼѹǿP0��ʾ��Ӧ��ϵ�������ʵ���n���ͷ�Ӧ��A�����ʵ���n��A����
n��=0.1��$\frac{P}{{P}_{0}}$mol��n��A��=0.1����2-$\frac{P}{{P}_{0}}$��mol�������ϱ�����a=0.026mol/L��
��5��������ʵ�����ݵõ���Ӧ�����뷴Ӧ������������ʵ���Ũ�ȵĹ�ϵ����ͼ��ʾ�������ߵ�ijһ�¶�ʱ��Ӧ���´ﵽƽ�⣬��Ӧ�ĵ�ֱ�Ϊa��d��
��1��2SO2��g��+O2��g��?2SO3��g������H1=-197kJ/mol��K1=a
��2��2NO��g��+O2��g��?2NO2��g������H2=-144kJ/mol��K2=b
��3��NO2��g��+SO2��g��?SO3��g��+NO��g������H3=m kJ/mol��K3=c
����˵����ȷ���ǣ�������
| A�� | m=-26.5��2c=a-b | B�� | m=-53��c2=$\frac{a}{b}$ | C�� | m=-26.5��c2=$\frac{a}{b}$ | D�� | m=-53��2c=a-b |
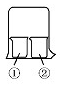 ����ͼ��ʾװ�ý�������ʵ�飬ʵ������Ԥ�������һ�µ��ǣ�������
����ͼ��ʾװ�ý�������ʵ�飬ʵ������Ԥ�������һ�µ��ǣ������� | ���е����� | ���е����� | Ԥ��ٵ����� | |
| A | ����KI��Һ | Ũ���� | �����Ա仯 |
| B | ��̪��Һ | Ũ���� | �����Ա仯 |
| C | AlCl3��Һ | Ũ��ˮ | �а�ɫ���� |
| D | ʪ���ֽ�� | ������ˮ | ��ֽ����ɫ |
| A�� | ����KI��ҺŨ���������Ա仯 | B�� | ��̪��ҺŨ���������Ա仯 | ||
| C�� | AlCl3��ҺŨ��ˮ�а�ɫ���� | D�� | ʪ���ֽ��������ˮ��ֽ����ɫ |
 ��1����֪��ͼ��ʾ�Ŀ��淴Ӧ��
��1����֪��ͼ��ʾ�Ŀ��淴Ӧ��