��Ŀ����
4�����������Ȼ�淋Ļ����Һ��������Ӧ2H++NH4++3Cl-$\frac{\underline{\;���\;}}{\;}$3H2��+NCl3������VL���壮��NA��������٤��������ֵ������˵����ȷ���ǣ�������| A�� | 0.1mol•L-1��NH4Cl��Һ�к�NH4+����ĿС��0.01NA | |
| B�� | �÷�Ӧ�ͷ�H2�ķ�����Ϊ$\frac{V}{22.4}$NA | |
| C�� | ��Ӧ��ÿ����2molH+ת�Ƶĵ�����Ϊ6NA | |
| D�� | 120.5gNCl3���������ۼ���Ϊ4NA |
���� A����Һ�������ȷ��
B������������״̬����ȷ��
C����Ӧ2H++NH4++3Cl-$\frac{\underline{\;���\;}}{\;}$3H2��+NCl3ת��6mol���ӣ�����2mol�����ӣ�
D��NCl3�к�3mol���ۼ���
��� �⣺A����Һ�������ȷ������Һ�е�笠����ӵĸ��������㣬��A����
B������������״̬����ȷ�������ɵ������ķ��Ӹ������ܸ���22.4L/mol�����㣬��B����
C����Ӧ2H++NH4++3Cl-$\frac{\underline{\;���\;}}{\;}$3H2��+NCl3ת��6mol���ӣ�����2mol�����ӣ��ʵ�����2mol������ʱת��6NA�����ӣ���C��ȷ��
D��120.5gNCl3�����ʵ���Ϊ1mol����NCl3�к�3mol���ۼ�����1molNCl3�к�3NA�����ۼ�����D����
��ѡC��
���� ���⿼���˰���٤���������йؼ��㣬�ѶȲ���Ӧע�ʽ�����ú����ʵĽṹ�ص㣮

��ϰ��ϵ�д�
�����Ŀ
1����NAΪ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A�� | �����£�1LpH=13��NaOH��Һ�У���ˮ�����OH-��ĿΪ0.1NA | |
| B�� | 0.1mol${\;}_{38}^{90}$Srԭ���к�������Ϊ3.8NA | |
| C�� | ������O2��1molNa��ַ�Ӧ��ת�Ƶĵ�����ΪNA | |
| D�� | 50mL12mol•L-1������������MnO2���ȣ�ת�Ƶ�����Ϊ0.3NA |
1��Li4Ti5O12��LiFePO4��������ӵ�صĵ缫���ϣ���������������Ҫ�ɷ�ΪFeTiO3������������MgO��SiO2�����ʣ����Ʊ��������������£�

�ش��������⣺
��1���������ʵ���У����Ľ����ʽ������ͼ��ʾ����ͼ��֪�������Ľ�����Ϊ70%ʱ�������õ�ʵ������Ϊ100�桢2h��90�桢5h��

��2���������������Ҫ��TiOCl42-��ʽ���ڣ�д����Ӧ��Ӧ�����ӷ���ʽFeTiO3+4H++4Cl-=Fe2++TiOCl42-+2H2O��
��3��TiO2•xH2O������˫��ˮ����ˮ��Ӧ40min����ʵ�������±���ʾ��
����40��ʱTiO2•xH2Oת������ߵ�ԭ���¶ȸ߷�Ӧ���ʼӿ죬���¶ȹ��߹�������ֽ⡢��ˮ�ӷ���Ӱ��ת���ʣ�
��4��Li2Ti5O15��Ti�Ļ��ϼ�Ϊ+4�����й���������ĿΪ4��
��5��������Һ�ڡ���c��Mg2+��=0.02mol•L-1������˫��ˮ�����ᣨ����Һ�������1������ʹFe3+ǡ�ó�����ȫ����Һ��c��Fe3+��=1.0��10-5����ʱ�Ƿ���Mg3��PO4��2�������ɣ�c��PO43-��=$\frac{1.3��1{0}^{22}}{1��1{0}^{-5}}$=1.0��10-17mol/L��Qc[Mg3��PO4��2]=��$\frac{0.02mol/L}{2}$��3����1.0��10-17mol/L��2=1.69��10-40��Ksp=1.0��10-24�����������ɣ���ʽ���㣩��FePO4��Mg3��PO4��2��Ksp�ֱ�Ϊ1.3��10-22��1.0��10-24
��6��д�����������բڡ�����FePO4�Ʊ�LiFePO4�Ļ�ѧ����ʽ2FePO4+Li2CO3+H2C2O4$\frac{\underline{\;����\;}}{\;}$2LiFePO4+H2O+3CO2����

�ش��������⣺
��1���������ʵ���У����Ľ����ʽ������ͼ��ʾ����ͼ��֪�������Ľ�����Ϊ70%ʱ�������õ�ʵ������Ϊ100�桢2h��90�桢5h��

��2���������������Ҫ��TiOCl42-��ʽ���ڣ�д����Ӧ��Ӧ�����ӷ���ʽFeTiO3+4H++4Cl-=Fe2++TiOCl42-+2H2O��
��3��TiO2•xH2O������˫��ˮ����ˮ��Ӧ40min����ʵ�������±���ʾ��
| �¶�/�� | 30 | 35 | 40 | 45 | 50 |
| TiO2•xH2Oת����/% | 92 | 95 | 97 | 93 | 88 |
��4��Li2Ti5O15��Ti�Ļ��ϼ�Ϊ+4�����й���������ĿΪ4��
��5��������Һ�ڡ���c��Mg2+��=0.02mol•L-1������˫��ˮ�����ᣨ����Һ�������1������ʹFe3+ǡ�ó�����ȫ����Һ��c��Fe3+��=1.0��10-5����ʱ�Ƿ���Mg3��PO4��2�������ɣ�c��PO43-��=$\frac{1.3��1{0}^{22}}{1��1{0}^{-5}}$=1.0��10-17mol/L��Qc[Mg3��PO4��2]=��$\frac{0.02mol/L}{2}$��3����1.0��10-17mol/L��2=1.69��10-40��Ksp=1.0��10-24�����������ɣ���ʽ���㣩��FePO4��Mg3��PO4��2��Ksp�ֱ�Ϊ1.3��10-22��1.0��10-24
��6��д�����������բڡ�����FePO4�Ʊ�LiFePO4�Ļ�ѧ����ʽ2FePO4+Li2CO3+H2C2O4$\frac{\underline{\;����\;}}{\;}$2LiFePO4+H2O+3CO2����
5����ѹ���ʻ�������������ԭ��Ϊ��Ni��s��+4CO��g��?Ni��CO��4��g����230��ʱ���÷�Ӧ��ƽ�ⳣ��K=2��10-5����֪��Ni��CO��4�ķе�Ϊ42.2�棬�������ʲ����뷴Ӧ��
��һ�Σ���������CO��Ӧת������̬Ni��CO��4��
�ڶ��Σ�����һ�η�Ӧ���������������������230���Ƶøߴ�����
�����ж���ȷ���ǣ�������
��һ�Σ���������CO��Ӧת������̬Ni��CO��4��
�ڶ��Σ�����һ�η�Ӧ���������������������230���Ƶøߴ�����
�����ж���ȷ���ǣ�������
| A�� | ����c��CO����ƽ���������ƶ�����Ӧ��ƽ�ⳣ������ | |
| B�� | ��һ�Σ���30���50������֮��ѡ��Ӧ�¶ȣ�ѡ50�� | |
| C�� | �ڶ��Σ�Ni��CO��4�ֽ��ʽϵ� | |
| D�� | �÷�Ӧ�ﵽƽ��ʱ��v����[Ni��CO��4]=4v������CO�� |
4������˵����ȷ���ǣ�������
| A�� | ú�ĸ����ʯ�͵ķ����������仯 | |
| B�� | 1-�ȱ����2-�ȱ������ȥ������ͬ | |
| C�� | �����顢�����顢������ķе������� | |
| D�� | ��ȥ���б��ӣ�������ˮ����� |
10�����з�Ӧ�У���ԭ���Ͽ�����Ƴ�ԭ��ص��ǣ�������
| A�� | Ba��OH��2•8H2O��NH4Cl�ķ�Ӧ | B�� | NaOH��Һ������ķ�Ӧ | ||
| C�� | �����������ķ�Ӧ | D�� | ʯ��ʯ�ķֽⷴӦ |
 �о����֣���CO2��ѹ�ϳɼ״���Ӧ��CO2+3H2=CH3OH+H2O���У�Co�����︺�ص�Mn�������������Ӵ������и��ԣ���ʾ�����õ�Ӧ��ǰ�����ش��������⣺
�о����֣���CO2��ѹ�ϳɼ״���Ӧ��CO2+3H2=CH3OH+H2O���У�Co�����︺�ص�Mn�������������Ӵ������и��ԣ���ʾ�����õ�Ӧ��ǰ�����ش��������⣺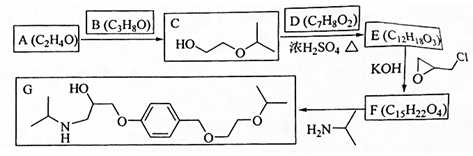
 ��
��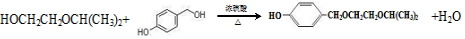 ��
�� ��
�� ��
��

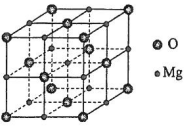
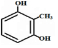
 �����в�ͬ��ѧ��������ԭ�ӹ���4�֣�����ԭ����Ŀ���Ϊ13��
�����в�ͬ��ѧ��������ԭ�ӹ���4�֣�����ԭ����Ŀ���Ϊ13�� ��
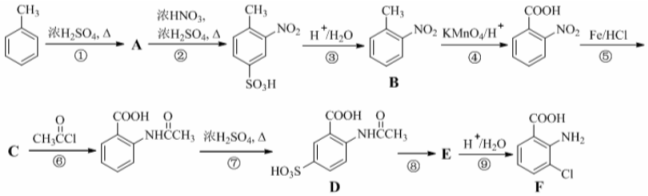
�� ���ò���Ӧ����ҪĿ���DZ���������
���ò���Ӧ����ҪĿ���DZ���������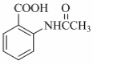 Ϊ��Ҫԭ�ϣ������ٲ����Ʊ����ļ��ۺ�������̣�
Ϊ��Ҫԭ�ϣ������ٲ����Ʊ����ļ��ۺ�������̣�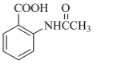 ��$��_{��Ӧ����}^{��Ӧ�Լ�}$Ŀ�껯����
��$��_{��Ӧ����}^{��Ӧ�Լ�}$Ŀ�껯����