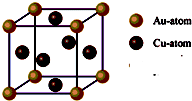
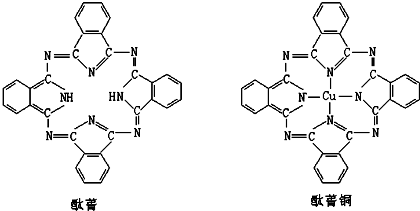
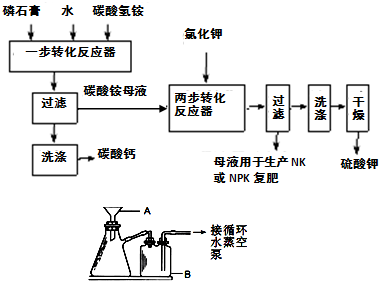
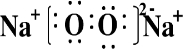��Ŀ����
��������������Ҫ�Ľ��������ǵĵ��ʼ��仯�������Ÿ��Ե����ʡ�
��1��������100mL 0.01mol∙L��1 FeCl3��Һ�����ƹ�������Ҫ�IJ�����������Ͳ����ͷ�ιܡ��ձ��⣬����Ҫ �����ƹ����г�����FeCl3���塢����ˮ�⣬����Ҫ���Լ��� ���������ƺõ���Һ�����ˮ�в����һ��ʱ�䣬�ɵõ����ɫҺ�壬�÷�Ӧ�����ӷ���ʽΪ ����Һ����е������� ������ĸ����
a������ͨ����Һ��ʱ�γɹ����ġ�ͨ·��
b�����Һ���м���AgNO3��Һ����������
c������Һ����й��ˣ��ɵõ����ɫ����
d������Һ����ȡ����ɡ����պ�������������
��2���ڸ��������������£���þ����ȼFe3O4��ĩ�����۵ľ��Ȼ���ʹ���ַ�Ӧ��
�����ַ�Ӧ���ʣ������м���������NaOH��Һ���д������ݲ���������ʣ������г�������еĹ��������� ���ѧʽ����
����֪��3Fe(s)+2O2(g)=Fe3O4(s) ��H��−1118 kJ��mol��1
2Al(s)+3/2O2(g)=Al2O3(s) ��H��−1675.7 kJ��mol��1
������Fe3O4������Ӧ��������Al2O3���Ȼ�ѧ����ʽΪ
��
��1��100mL����ƿ�������� ���� Fe3++3H2O=Fe(OH)3(����)+3H+ a��d
��2����Al��Al2O3 ��8Al(s)+3Fe3O4(s)=4Al2O3(s)+9Fe(s) ��H��−3348.8 kJ��mol��1
��������
�����������2�������ַ�Ӧ���ʣ������м���������NaOH��Һ���д������ݲ�������һ������ʣ�࣬�ʳ�����Al��Al2O3���ɸ�˹���ɿɵ� 8Al(s)+3Fe3O4(s)= 4Al2O3(s)+9Fe(s) ��H��−3348.8 kJ��mol��1
���㣺���鷴Ӧ�ȼ��㡢��˹���ɡ���Һ���ơ���������֪ʶ��

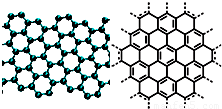
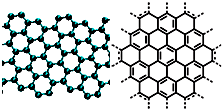 A����������ѧ����־������2009��ʮ���ѧͻ��֮һ��ʯīϩ���о���Ӧ�÷����ͻ�ƣ�ʯīϩ����ԭ�Ӽ��ĺ�ȡ�����ĵ�ѧ���ܡ���ɫ�Ļ�ѧ�ȶ��Ժ�����ѧ�ȶ��ԣ��Ʊ�ʯīϩ������ʯī���뷨����ѧ����������ȣ�ʯīϩ�����ģ�ͼ����ӽṹʾ��ͼ���ң�
A����������ѧ����־������2009��ʮ���ѧͻ��֮һ��ʯīϩ���о���Ӧ�÷����ͻ�ƣ�ʯīϩ����ԭ�Ӽ��ĺ�ȡ�����ĵ�ѧ���ܡ���ɫ�Ļ�ѧ�ȶ��Ժ�����ѧ�ȶ��ԣ��Ʊ�ʯīϩ������ʯī���뷨����ѧ����������ȣ�ʯīϩ�����ģ�ͼ����ӽṹʾ��ͼ���ң�