��Ŀ����
��Ҫ�ⶨij�Ȼ��������Ȼ������������Ԫ�ص���������,�����²������ʵ��:
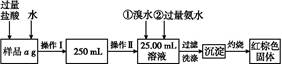
��.�������������,�ش���������:
��1�����������õ��IJ����������ձ�����������,�������������������������������������ƣ���
��2����д��������ˮ���������ӷ�Ӧ����ʽ: ����
��3�������������,��ȴ������,����ƽ����������Ϊb1 g,�ٴμ��Ȳ���ȴ�����³���������Ϊb2 g,��b1-b2="0.3" g,���������Ӧ���еIJ������� ��
��������������W1 g,����������Ⱥ������������W2 g,����Ʒ����Ԫ�ص���������������������������
��.��ͬѧ���,�����Բ������·������ⶨ:
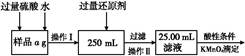
��1���ܽ���Ʒ����������,������������,Ϊʲô? ����
��2��ѡ��Ļ�ԭ���Ƿ�������������������ǡ���,ԭ����:�� ����
��3�����ζ��õ�c mol/L KMnO4��Һb mL,����Ʒ����Ԫ�ص���������������������

��.�������������,�ش���������:
��1�����������õ��IJ����������ձ�����������,�������������������������������������ƣ���
��2����д��������ˮ���������ӷ�Ӧ����ʽ: ����
��3�������������,��ȴ������,����ƽ����������Ϊb1 g,�ٴμ��Ȳ���ȴ�����³���������Ϊb2 g,��b1-b2="0.3" g,���������Ӧ���еIJ������� ��
��������������W1 g,����������Ⱥ������������W2 g,����Ʒ����Ԫ�ص���������������������������
��.��ͬѧ���,�����Բ������·������ⶨ:

��1���ܽ���Ʒ����������,������������,Ϊʲô? ����
��2��ѡ��Ļ�ԭ���Ƿ�������������������ǡ���,ԭ����:�� ����
��3�����ζ��õ�c mol/L KMnO4��Һb mL,����Ʒ����Ԫ�ص���������������������
��.��1��250 mL����ƿ��2�֣�����ͷ�ιܣ�2�֣�
��2��2Fe2++Br2 2Fe3++2Br-��2�֣�
2Fe3++2Br-��2�֣�
��3���ٴμ��Ȳ���ȴ�����³���,ֱ������������С��0.1 g��2�֣���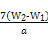 ��100%��1�֣�
��100%��1�֣�
��.��1������������Ժ���KMnO4�ĵζ��и��ţ�2�֣�
��2����1�֣��������������ԭ��,����������������Fe2+,�һ�����������ᷴӦ����Fe2+,������Ԫ�صIJⶨ��2�֣�
��3��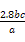 ��100%��1�֣�
��100%��1�֣�
��2��2Fe2++Br2
 2Fe3++2Br-��2�֣�
2Fe3++2Br-��2�֣���3���ٴμ��Ȳ���ȴ�����³���,ֱ������������С��0.1 g��2�֣���
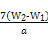 ��100%��1�֣�
��100%��1�֣���.��1������������Ժ���KMnO4�ĵζ��и��ţ�2�֣�
��2����1�֣��������������ԭ��,����������������Fe2+,�һ�����������ᷴӦ����Fe2+,������Ԫ�صIJⶨ��2�֣�
��3��
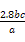 ��100%��1�֣�
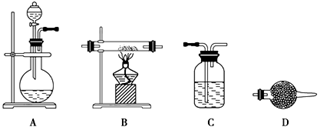
��100%��1�֣���.��1����������ͼ,������������һ�����ʵ���Ũ�ȵ���Һ,�������IJ�������Ϊ250 mL����ƿ�ͽ�ͷ�ιܡ���2��������ˮ��Fe2+��������3��b1-b2="0.3" g,˵������,������ٴμ��Ȳ���ȴ�����³���,ֱ������������С��0.1 g,������������Ϊ��W2-W1��g,��ԭ��Ʒ����Ԫ�ص�����Ϊ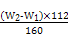 ��
��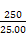 g=7��W2-W1�� g,����������Ϊ
g=7��W2-W1�� g,����������Ϊ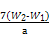 ��100%��
��100%��
��.�÷�����ԭ�����û�ԭ����Fe3+��ԭΪFe2+,Ȼ����KMnO4��Һ�ζ�,��������,����������ᱻKMnO4����,ʹ��Ԫ�ص���������ƫ��;������������ԭ��,����ʹ��Ԫ�ص���������ƫ��5F ��Mn
��Mn ,��ԭ��Ʒ��n��Fe��=n��Fe2+��=5��bc��10-3��
,��ԭ��Ʒ��n��Fe��=n��Fe2+��=5��bc��10-3��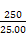 mol="0.05bc" mol,��m��Fe��="0.05bc��56" g="2.8bc" g,����������Ϊ
mol="0.05bc" mol,��m��Fe��="0.05bc��56" g="2.8bc" g,����������Ϊ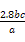 ��100%��
��100%��
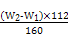 ��
��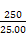 g=7��W2-W1�� g,����������Ϊ
g=7��W2-W1�� g,����������Ϊ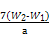 ��100%��
��100%����.�÷�����ԭ�����û�ԭ����Fe3+��ԭΪFe2+,Ȼ����KMnO4��Һ�ζ�,��������,����������ᱻKMnO4����,ʹ��Ԫ�ص���������ƫ��;������������ԭ��,����ʹ��Ԫ�ص���������ƫ��5F
 ��Mn
��Mn ,��ԭ��Ʒ��n��Fe��=n��Fe2+��=5��bc��10-3��
,��ԭ��Ʒ��n��Fe��=n��Fe2+��=5��bc��10-3��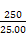 mol="0.05bc" mol,��m��Fe��="0.05bc��56" g="2.8bc" g,����������Ϊ
mol="0.05bc" mol,��m��Fe��="0.05bc��56" g="2.8bc" g,����������Ϊ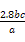 ��100%��
��100%��
��ϰ��ϵ�д�
 �ƸԾ���Ȥζ����ϵ�д�
�ƸԾ���Ȥζ����ϵ�д� ����С����ҵ��ϵ�д�
����С����ҵ��ϵ�д�
�����Ŀ
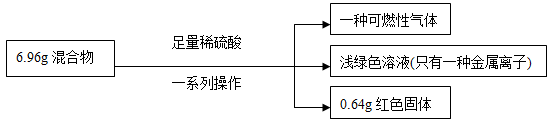
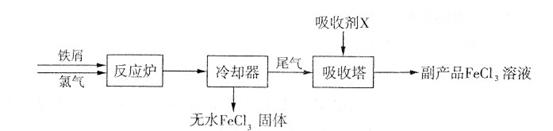
 +A����A�Ļ�ѧʽΪ ��
+A����A�Ļ�ѧʽΪ ��