��Ŀ����
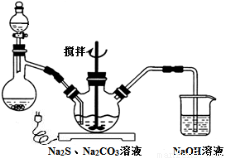
��������Ӱ�����ǵ������뽡����ij�����������п��ܺ������¿����������ӣ�Na����NH4+��Mg2����Al3����SO42����NO3-��Cl����ijͬѧ�ռ��˸õ���������������Ҫ��Ԥ�������������Һ����Ʋ����������ʵ�飺

��֪��3NO3-��8Al��5OH����2H2O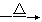 3NH3����8AlO2-
3NH3����8AlO2-
�������ϵ�ʵ�����������ͬѧ�ó��Ľ��۲���ȷ����( )
A�������п϶�����NH4+��Mg2����SO42����NO3-
B��������һ������Al3��
C�������п��ܴ���Na����Cl��
D���������п��ܴ���NaNO3��NH4Cl��MgSO4
��ϰ��ϵ�д�
 ���Ǽ���С����ϵ�д�
���Ǽ���С����ϵ�д�
�����Ŀ