��Ŀ����
(6��)��1���ڱ�״���£����������ʣ���4 g H2 ��33.6 L CH4 ��1 mol H2O ��3.01��1023��O2 �����к������������ǣ����ں�������д��ţ���ͬ�� ����ԭ�������ٵ��� ������������ �������С���� ��
��2����0.4 mol Al3����Al2(SO4)3��������SO42һ�����ʵ����� ��
��3����1��H2O�к���a����ԭ�ӣ����ӵ������ú�a�Ĵ���ʽ�ɱ�ʾΪ ��
��1���٣��ܣ��ڣ��ۣ� ��2��0.6mol�� ��3��9a mol-1��
��������
�����������1����4gH2��n��H2��=2mol��N��H2��=2NA��N��H��=4NA��v��H2��=44.8L���ڱ�״����33.6L CH4��n��CH4��=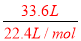 =1.5mol��N��CH4��=1.5NA��ԭ������Ϊ5��1.5NA=7.5NA������Ϊm��CH4��=1.5mol��16g/mol=24g����1molH2O�ķ�����ΪN��H2O��=1NA��ԭ������Ϊ3NA�����ԼΪ18ml������Ϊ1mol��18g/mol=18g����3.01��1023��O2�����ʵ���Ϊn��O2��=0.5mol��ԭ������Ϊ1NA�����Ϊ11.2L������Ϊ0.5mol��32g/mol=16g�����Ϸ�����֪���������������Ǣ٣���ԭ�������ٵ��Ǣܣ����������Ǣڣ������С�Ģۣ��ʴ�Ϊ�٣��ܣ��ڣ��ۣ���2��Al2��SO4��3��n��Al3+����n��SO42-��=2��3��n��Al3+��=0.4mol����n��SO42-��=0.6mol����3��1gˮ�����ʵ���Ϊ��n��H2O��=
=1.5mol��N��CH4��=1.5NA��ԭ������Ϊ5��1.5NA=7.5NA������Ϊm��CH4��=1.5mol��16g/mol=24g����1molH2O�ķ�����ΪN��H2O��=1NA��ԭ������Ϊ3NA�����ԼΪ18ml������Ϊ1mol��18g/mol=18g����3.01��1023��O2�����ʵ���Ϊn��O2��=0.5mol��ԭ������Ϊ1NA�����Ϊ11.2L������Ϊ0.5mol��32g/mol=16g�����Ϸ�����֪���������������Ǣ٣���ԭ�������ٵ��Ǣܣ����������Ǣڣ������С�Ģۣ��ʴ�Ϊ�٣��ܣ��ڣ��ۣ���2��Al2��SO4��3��n��Al3+����n��SO42-��=2��3��n��Al3+��=0.4mol����n��SO42-��=0.6mol����3��1gˮ�����ʵ���Ϊ��n��H2O��=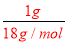 =
=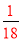 mol��
mol��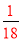 molˮ�к���Hԭ�ӵ���Ŀ=
molˮ�к���Hԭ�ӵ���Ŀ=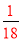 mol��2NA=a����٤������NA=
mol��2NA=a����٤������NA=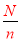 =
=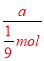 ==9amol-1��
==9amol-1��
���㣺���ʵ����ļ���

 ��ʦ����ָ���ο�ʱϵ�д�
��ʦ����ָ���ο�ʱϵ�д�| A��SO2��NO��CO2���ᵼ��������γ� | B���ȱȹ衢���ά���������մɵ������������ǽ������� | C����ȩ�ĺ�������ʳƷ��ȫ����ָ�� | D��Һ�ȹ�й©ʱ���ɽ�������ˮ���в�������ʯ�� |
��300 mL���ܱ������У��������۲�����һ������CO���壬һ�������·�����Ӧ��
Ni(s)��4CO(g)  Ni(CO)4(g)����֪�÷�Ӧƽ�ⳣ�����¶ȵĹ�ϵ���±�������˵����ȷ����
Ni(CO)4(g)����֪�÷�Ӧƽ�ⳣ�����¶ȵĹ�ϵ���±�������˵����ȷ����
�¶�/�� | 25 | 80 | 230 |
ƽ�ⳣ�� | 5��104 | 2 | 1.9��10��5 |
A����������Ni(CO)4�ķ�ӦΪ���ȷ�Ӧ
B��25 ��ʱ����ӦNi(CO)4(g)  Ni(s)��4CO(g)��ƽ�ⳣ��Ϊ2��10��5
Ni(s)��4CO(g)��ƽ�ⳣ��Ϊ2��10��5
C��80 ��ﵽƽ��ʱ�����n(CO)��0.3 mol����Ni(CO)4��ƽ��Ũ��Ϊ2 mol��L��1
D��80 ��ʱ�����ijʱ��Ni(CO)4��CO��Ũ�Ⱦ�Ϊ0.5 mol��L��1�����ʱ��(��)���ԣ���)
 xLi����R�������й�˵����ȷ���ǣ� ��
xLi����R�������й�˵����ȷ���ǣ� �� ��NO
��NO ��SO
��SO
 Inԭ�Ӻ�����49������
Inԭ�Ӻ�����49������