��Ŀ����
��10�֣�����һ������ˮ��Һ��ֻ���ܺ������������е������֣�K+��Al3+��Fe3+��Mg2+��Ba2+��NH4+��Cl-��SO42-����ȡ����100mL��Һ��������ʵ�飺
�ٵ�һ����Һ������������Һ�г�������
�ڵڶ��ݼ������NaOH��Һ���Ⱥ�ֻ�ռ�������0.02mol���������ɣ�ͬʱ�õ���Һ�ס�
���ڵڶ������õļ���Һ��ͨ�������CO2�����ɰ�ɫ���������������ˡ�ϴ�ӡ����գ�����Ϊ1.02g��
�ܵ�������Һ��������BaCl2��Һ�õ���ɫ��������������������ϴ�ӡ����������Ϊ11.65g��
��������ʵ��ش�
��1��һ�������ڵ������� ��
��2����ȷ��һ�����ڵ������Ӽ���Ũ�ȣ��ɲ�������
���ӷ��� ��Ũ�� ��
���ӷ��� ��Ũ�� ��
��3����ȷ��K+�Ƿ���� ����ǡ��������ǣ� ��
��10�֣�ÿ��2�֣�
��1��Fe3+��Mg2+��Ba2+��2��SO42- 0.5mol?L?1
�� 3���� ��Һ�п϶����ڵ������ǡ�NH4+��Al3+��SO42-��������NH4+��Al3+�����ʵ�������0.02mol��SO42-�����ʵ�����0.05mol�����ݵ���غ㣬K+һ�����ڡ�
��������
����������ڶ��ݼ������NaOH��Һ���Ⱥ�ֻ�ռ�������0.02mol��������Ϊ��������ԭ��Һһ������NH4+��NH4+���ʵ���Ϊ0.02mol���������ɣ���ԭ��Һһ������Fe3+��Mg2+���ڵڶ������õļ���Һ��ͨ�������CO2�����ɰ�ɫ�������ó���ΪAl(OH)3�����������ˡ�ϴ�ӡ����գ��õ�Al2O3������Ϊ1.02g�����ʵ���Ϊ0.01mol��������Ԫ���غ㣬ԭ��Һ��Al3+���ʵ���Ϊ0.02mol����������Һ��������BaCl2��Һ�õ���ɫ������ΪBaSO4����������Ϊ11.65g������ԭ��Һ����SO42?�����ʵ���Ϊ��11.65g��233g/mol=0.05mol����ԭ��Һһ������Ba2+��n��NH4+��+3n��Al3+��=0.02mol+3��0.02mol=0.08mol��2n��SO42?��=0.1mol�����ݵ���غ㣬ԭ��Һһ������K+����Ϊ��ȷ��ԭ��Һ�Ƿ���Cl?������K+����С���ʵ���Ϊ0.02mol��
��1����������������һ�������ڵ������ǣ�Fe3+��Mg2+��Ba2+��
��2���϶����ڵ�������Ϊ�� SO42?��SO42?���ʵ���Ϊ0.05mol����c��SO42?��=0.05mol��0.1L=0.5mol?L?1��
��3�����ݵ���غ㣬��Һ�п϶����ڵ������ǡ�NH4+��Al3+��SO42-��������NH4+��Al3+�����ʵ�������0.02mol��SO42-�����ʵ�����0.05mol�����ݵ���غ㣬K+һ�����ڡ�
���㣺���⿼�����ӵļ��鼰�ƶϡ���ѧ���㡣

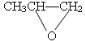 ��CO2��Ӧ���ɿɽ���ۺ���
��CO2��Ӧ���ɿɽ���ۺ���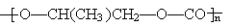 ���÷�Ӧ������ɫ��ѧ��ԭ��
���÷�Ӧ������ɫ��ѧ��ԭ�� �������ʵ���Ϊ0.9 mol
�������ʵ���Ϊ0.9 mol Cu2S��3SO2��2FeO(¯��)��
Cu2S��3SO2��2FeO(¯��)�� �������й����ʵ��ƶϲ���ȷ����
�������й����ʵ��ƶϲ���ȷ����