��Ŀ����
16��ͭ���������ϵ���е���ɫ��������֪�������£�����Һ��Cu2+�ȶ���Cu+�������������·�����Ӧ2Cu+=Cu2++Cu����֪�����+1��ͭ�Ļ�������������磺Cu2O��CuI��CuCl��CuH�ȣ���1��������Cu��OH��2����Һ�е�����������Һ���������ɲ��������ɫΪש��ɫ��ijͬѧʵ��ʱȴ�к�ɫ���ʳ��֣����ֺ�ɫ���ʵĻ�ѧʽΪCuO��
��2����CuCl2��Һ����μ������KI��Һ�����ܷ����ķ�Ӧ�У�
a.2Cu2++4I-=2CuI������ɫ��+I2
b.2Cu2++2Cl-+2I-=2CuCl����ɫ��+I2
Ϊ��˳���۲쵽������Ӧ�в����İ�ɫ���������Լ��������Լ���B��
A��SO2 B���� C��NaOH��Һ D���Ҵ�
��3��һ�������£���CuSO4�м���NH5��Ӧ�����⻯��ͭ��CuH����
����֪NH5�����ӻ�����������ԭ�Ӷ��ﵽϡ��������ȶ��ṹ����д��NH5�ĵ���ʽ
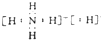 ��
����д��CuH�ڹ���ϡ������������������ӷ���ʽ2CuH+2H+=Cu2++Cu+2H2����
�۽�CuH�ܽ���������ϡ�����У�������л�ѧ����ʽ��
��CuH+��HNO3=��Cu��NO3��2+��H2��+��NO+��H2O��
��4�������£���0.20mol•L-1����ͭ��Һ�м�������������Һ������dz��ɫ������ͭ����������Һ��pH=6ʱ��c��Cu2+��=2.2��10-4������֪��KSP[Cu��OH��2]=2.2��10-20��
���� ��1�������Ƿ����к���ȩ����������������ͭ��Һ���ȷ�Ӧ����ש��ɫ����Cu2O����ɫ�����ʵIJ���Դ�ڼ����¶ȹ��ߣ�
��2����ˮ����ɫӰ���ɫ�����Ĺ۲죬����ѡ����ȡ���ѵ���ȡ������
��3����NH5������ԭ�Ӷ��ﵽϡ��������ȶ��ṹ��˵��NH5����NH4+��H-��ɵ����ӻ����
��CuH�ܽ���ϡ�����У�CuH�е�H-ʧ���ӣ�������H+�õ��ӣ�����������Ϊ��������������Һ��2Cu+=Cu2++Cu��
�۷���������ԭ��Ӧ��������NO��ˮ�ȣ�
��4��pH=6ʱ��c��OH-��=10-8mol•L?1�����Ksp[Cu��OH��2]���㣮
��� �⣺��1����������һ�����ǻ�ȩ����������������ͭ��Һ���ȷ�Ӧ����ש��ɫ����Cu2O����������¶ȹ��ߣ�������ͭ���ȷֽ�Ϊ��ɫ������ͭ��CuO����ˮ��
�ʴ�Ϊ��ש��ɫ��CuO��
��2����ˮ���غ�ɫ��Ӱ���ɫ�����Ĺ۲죬�����ñ��ѵⵥ����ȡ���������ھƾ���ˮ������������ܣ���˲�������ȡ�����ʴ�Ϊ��B��
��3����NH5������ԭ�Ӷ��ﵽϡ��������ȶ��ṹ������ԭ�������ﵽ8�����ȶ��ṹ��������ԭ�Ӵﵽ2�����ȶ��ṹ��˵��NH5����NH4+��H-��ɵ����ӻ���������ʽΪ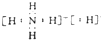 ��
��
�ʴ�Ϊ��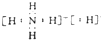 ��
��
��CuH�ܽ���ϡ�����У�CuH�е�H-ʧ����������H+�õ��ӣ�����������Ϊ������������Һ��2Cu+=Cu2++Cu�������ӷ���ʽΪ��2CuH+2H+=Cu2++Cu+2H2��
�ʴ�Ϊ��2CuH+2H+=Cu2++Cu+2H2����
��ϡ���ᱻ��ԭΪNO��ͬʱ��ˮ���ɣ�1molCuHʧȥ����2mol����NO�����ʵ���֮����3��2����ӦΪ6CuH+16HNO3=6Cu��NO3��2+3H2��+4NO��+8H2O��
�ʴ�Ϊ��6��16��6��3��4NO��8H2O��
��4��pH=6ʱ��c��OH-��=10-8mol•L?1����Ksp[Cu��OH��2]=2.2��10-20mol3•L-3��֪��c��Cu2+��=$\frac{2.2��1{0}^{-20}}{��1{0}^{-8}��^{2}}$=2.2��10-4mol•L?1���ʴ�Ϊ��2.2��10-4��
���� ���⿼����ۺϣ��漰���ʵ����ʡ�������ԭ��Ӧ��Ksp����ȣ�ע�ػ�ѧ��Ӧԭ���Ŀ��飬����ϰ���е���Ϣ��Ǩ��Ӧ������Ϊ���Ĺؼ�����Ŀ�ѶȲ���

| A�� | 0.2mol��ϩ���к���˫������ĿΪ0.2NA | |
| B�� | ��ϩ��1-��ϩ��ɵ�21g�����������ԭ�ӵĸ���Ϊ3NA | |
| C�� | ��״���£�11.2L���к��з��ӵ���ĿΪ0.5NA | |
| D�� | 1mol�ǻ���1mol����������������������Ϊ9NA |
| A�� | ��ϡ�ı���ˮ��Һ�еμ�����ϡ��ˮ���ȿ�����ɫ�����������ʧ����Ϊ���ɵ����屽�������ڱ�����Һ�� | |
| B�� | ��Ũ�Ⱦ�Ϊ0.1mol/L��MgCl2��CuCl2�����Һ����μ��백ˮ���ȳ�����ɫ��������ΪKSP[Mg��OH��2]��KSP[Cu��OH��2] | |
| C�� | ��������ʵ�����ģ�����������ޣ����ձ�����Ͳ�����β����������ֽ����Ӳֽ�塢�¶ȼ�������кͷ�Ӧ��Ӧ�ȵIJⶨ | |
| D�� | ��ʵ��������ϩ�ķ���װ�����ƵõIJ���ֱ��ͨ�����Ը��������Һ����Һ��ɫ��˵����ϩ�л�ԭ�� |
��1��±��Ԫ��λ�����ڱ���p������ļ۵����Ų�ʽΪ4s24p5
��2���ڲ�̫ϡ����Һ�У���������Զ����ӵϣ�HF��2��ʽ���ڣ�ʹ�������ӵϵ������������
��3��������±��ṩ�ĵ�һ�����������жϣ����п������ɽ��ȶ��ĵ��������ӵ�±��ԭ���ǵ�
| �� | �� | �� | �� | �� | |
| ��һ������ ��kJ/mol�� | 1681 | 1251 | 1140 | 1008 | 900 |
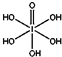 ����HIO4��ǰ��Ϊ��Ԫ�ᣬ����ΪһԪ�ᣮ��Ƚ϶�������ǿ����H5IO6��HIO4�����������������=����
����HIO4��ǰ��Ϊ��Ԫ�ᣬ����ΪһԪ�ᣮ��Ƚ϶�������ǿ����H5IO6��HIO4�����������������=������5����֪ClO2-Ϊ���ͣ�������ԭ����Χ���ĶԼ۲���ӣ�ClO2-������ԭ�ӵ��ӻ��������Ϊsp3��д��CN-�ĵȵ�����ķ���ʽN2�� CO��д��1����
��6����ͼ1Ϊ�⾧�徧���ṹ���й�˵������ȷ����AD
A������ӵ�������2�ֲ�ͬ��ȡ��2��ȡ��ͬ�ĵ������4��λ��������λ�γɲ�ṹ
B���þ�̯����֪ƽ��ÿ����������4����ԭ��
C���⾧��Ϊ��������Ŀռ�ṹ����ԭ�Ӿ���
D���⾧���еĵ�ԭ�Ӽ���ڷǼ��Լ��ͷ��»���
��7����֪CaF2���壨��ͼ2�����ܶ�Ϊ��g/cm3��NAΪ�����ӵ����������ڵ�����Ca2+�ĺ˼��Ϊa cm����CaF2����Է����������Ա�ʾΪ$\frac{\sqrt{2}}{2}$��NAa3

| A�� | ����ʽΪC3H6���л���ֻ������ͬ���칹�� | |
| B�� | ��״���£�22.4 L CCl4������Ϊ154 g | |
| C�� | ѡ���ʵ��Ĵ������ȿ��Ըı仯ѧ��Ӧ���ʣ�Ҳ���Ըı䷴Ӧ���ƽ��ת���� | |
| D�� | 2 g H2��g����O2��g������ȫȼ������H2O��l���ų�����Ϊ285.8 kJ����÷�Ӧ���Ȼ�ѧ����ʽ�ɱ�ʾΪ��2H2��g��+O2��g���T2H2O��l����H=-285.8 kJ•mol-1 |
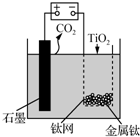 ����ͼ��ʾװ�ã�����CaF2-CaO������ʣ���ý����ƣ����øƻ�ԭTiO2�Ʊ������ѣ�����˵����ȷ���ǣ�������
����ͼ��ʾװ�ã�����CaF2-CaO������ʣ���ý����ƣ����øƻ�ԭTiO2�Ʊ������ѣ�����˵����ȷ���ǣ�������| A�� | �������У�Ca2+�������ƶ� | |
| B�� | �����ĵ缫��ӦʽΪ��C+2O2--4e-�TCO2�� | |
| C�� | ���Ʊ�������ǰ������װ����CaO���������� | |
| D�� | ����Ǧ��������װ�õĹ����Դ����+����������Pb�缫 |
| A�� | һ������������ȼ�ϣ����������ԴΣ�� | |
| B�� | ����ȼ�յĻ�ѧ����ʽ����Ϊ4NH3+5O2$\frac{\underline{\;��ȼ\;}}{\;}$4NO+6H2O | |
| C�� | ���������״��棬��Ϊ����Һ�� | |
| D�� | �ڰ�ȼ�ϵ���У��������� |
| A�� | ��ˮ | B�� | ������������ | C�� | �����¶� | D�� | �������ռ���Һ |
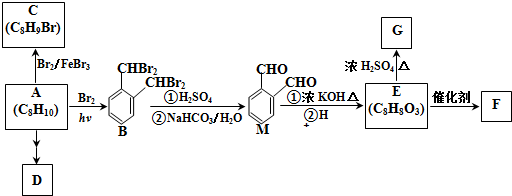

 ����A����B�ķ�Ӧ�����У���ͬʱ���ɶ��ָ��������һ����B��Ϊͬ���칹�壬���ĺ˴Ź���������5�����շ壮
����A����B�ķ�Ӧ�����У���ͬʱ���ɶ��ָ��������һ����B��Ϊͬ���칹�壬���ĺ˴Ź���������5�����շ壮 ��
��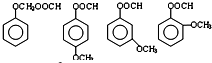 ��
��