��Ŀ����
8����ͼ��ʾ����ʵ��װ�ã��ֱ�ش��������⣮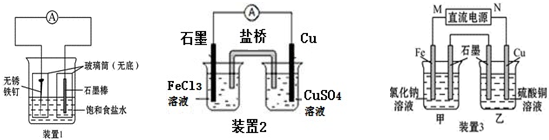
��1��װ��1Ϊ����������ʴʵ�飮һ��ʱ��������ʯī���IJ���Ͳ�ڵ����̪��Һ���ɹ۲쵽ʯī����������Һ��죬���װ����ʯī�缫�ķ�ӦʽΪO2+2H2O+4e-�T4OH-��
��2��װ��2�е�ͭ�Ǹ����������������������װ����ʯī�缫�������ķ�ӦΪFe3++e-=Fe2+��
��3��װ��3�м��ձ�ʢ��100mL 0.2mol•L-1��NaCl��Һ�����ձ�ʢ��100mL0.5mol•L-1��CuSO4��Һ����Ӧһ��ʱ���ֹͣͨ�磮����ձ��е��뼸�η�̪���۲쵽ʯī�缫�������ȱ�죮
�ټ��ձ�����Ϊ�����������ձ���ʯī�缫�ĵ缫��ӦʽΪ2H2O+2e-=H2��+2OH-��2H++2e-�TH2����
�����ձ��е�ⷴӦ�����ӷ���ʽΪ2Cu2++2H2O$\frac{\underline{\;���\;}}{\;}$2Cu+O2��+4H+��
��ֹͣ��⣬ȡ��Cu�缫��ϴ�ӡ�����������缫����0.64g�����ձ��в����������ڱ�״���µ����Ϊ224 mL��
���� ��1��������������ʴ��������������Fe2+������������ԭ��Ӧ�������õ����ӱ���ԭ����OH-��
��2���γ�ԭ��أ��ܷ���ʽΪCu+2Fe3+=Cu2++2Fe2+�������ܷ���ʽ�жϣ�
��3������ձ��е��뼸�η�̪���۲쵽ʯī�缫�������ȱ�죬˵��ʯī������OH-��ӦΪ���ص���������MΪ������NΪ���������ձ�Ϊ�������ķ�Ӧ������������������������ͭ����ϵ缫����ʽ�����⣮
��� �⣺��1��������������ʴ������������ԭ��Ӧ�������õ����ӱ���ԭ����OH-���缫����ʽΪO2+2H2O+4e-�T4OH-��
�ʴ�Ϊ��O2+2H2O+4e-�T4OH-��
��2��ͭ�ɱ�Fe3++����������Cu+2Fe3+=Cu2++2Fe2+���γ�ԭ��ط�Ӧʱ��ͭΪ����������������Ӧ��ʯīΪ������������ӦΪFe3++e-=Fe2+��
�ʴ�Ϊ������ Fe3++e-=Fe2+��
��3�����ձ�Ϊ�������ķ�Ӧ������������������������ͭ��
������ձ��е��뼸�η�̪���۲쵽ʯī�缫�������ȱ�죬˵��ʯī������OH-��ӦΪ���ص���������MΪ������NΪ��������Ϊ����������Ȼ�����Һ������������ԭ��Ӧ�����������缫����ʽΪ2H++2e-�TH2����
�ʴ�Ϊ������ 2H2O+2e-=H2��+2OH-��2H++2e-�TH2����
�����ձ�Ϊ�������ķ�Ӧ������������������������ͭ����ⷽ��ʽΪ2Cu2++2H2O$\frac{\underline{\;���\;}}{\;}$2Cu+O2��+4H+��
�ʴ�Ϊ��2Cu2++2H2O$\frac{\underline{\;���\;}}{\;}$2Cu+O2��+4H+��
��ȡ��Cu�缫��ϴ�ӡ�����������缫����0.64g��������Cu�����ʵ���Ϊ$\frac{0.64g}{64g/mol}$=0.01mol��ת�Ƶĵ��ӵ����ʵ���Ϊ0.01mol��2=0.02mol��
���ձ��У���������������������������Ϊ������
2H2O+2e-�T2OH-+H2����
2mol 22.4L
0.02mol V
V=$\frac{22.4L��0.02mol}{2mol}$=0.224L����224mL��
�ʴ�Ϊ��224��
���� ���⿼��绯ѧ֪ʶ��Ϊ��Ƶ���㣬������ѧ���ķ��������������Ŀ��飬��Ŀ�Ѷ��еȣ�����ʱע��缫���жϺ͵缫��Ӧ����д��ע�����·�и��缫ת�Ƶĵ�����Ŀ��ȣ����÷�Ӧ�ķ���ʽ���㣮

a������ֱ���Ĵ�С b�����Ӽ����Ĵ�С
c�����Ӽ��������С d��������Ŀ�Ĵ�С��
| A�� | a��b | B�� | b��c | C�� | a��d | D�� | b��d |
| A�� | Һ�� | B�� | Ư�� | C�� | ���� | D�� | �ơ� |

| A�� | I�͢� | B�� | I��IV | C�� | ��͢� | D�� | ���IV |
�ټӳɷ�Ӧ����������Ӧ����ȼ�ա��ܼӾ۷�Ӧ����ȡ����Ӧ��
| A�� | �٢ڢۢܢ� | B�� | ֻ�Т٢ڢۢ� | C�� | ֻ�Т٢ڢ� | D�� | ֻ�Т٢ۢ� |
| A�� | ���軯� | B�� | ������ | C�� | ���� | D�� | ���Ȼ�̼ |
 ʵ��������һδ֪Ũ�ȵ�ϡ���ᣬijѧ����ʵ�����н��вⶨ����Ũ�ȵ�ʵ�飻�����������գ�
ʵ��������һδ֪Ũ�ȵ�ϡ���ᣬijѧ����ʵ�����н��вⶨ����Ũ�ȵ�ʵ�飻�����������գ���1������100mL0.10mol•L-1NaOH����Һ��
����������IJ��������У��ձ�����Ͳ����������100mL����ƿ����ͷ�ιܵȣ�
�ڳ�ȡ0.4g�������ƹ���������������ƽ�������롢���ӣ���ҩ�ס��ձ���
��2��ȡ20.00mL����ϡ���������ƿ�У����μ�2��3�η�̪��ָʾ�����ü�ʽ�ζ��ܣ����������ƣ�װ���Ƶı�NaOH��Һ���еζ����ظ������ζ�����2��3�Σ���¼�������£�
| �ζ����� | ��������������mL�� | ��NaOH��Һ��� | |
| �ζ�ǰ�Ŀ̶ȣ�mL�� | �ζ���Ŀ̶ȣ�mL�� | ||
| ��һ�� | 20.00 | 0.40 | 20.50 |
| �ڶ��� | 20.00 | 4.10 | 24.00 |
| ������ | 20.00 | 1.00 | 24.00 |
�ڸ����������ݣ��ɼ�����������Ũ��ԼΪ0.10 mol/L������С������λ������
�۸���ϴ��ĵζ�����ע����ҺʱӦC������ţ�
A��������©�� B�������ڲ����� C���������ձ� D��ֱ��ע��
����ȥ��ʽ�ζ��������ݵķ���Ӧ������ͼ��������Ȼ�����ἷѹ������ʹ���첿�ֳ�����Һ��
��������ʵ���У����в���������������ȷ������ɲⶨ���ƫ�͵���AD
D���ζ��յ����ʱ���Ӷ�
A����ʽ�ζ���ʹ��ǰ��ˮϴ��δ�ô���������ϴ
B����ƿˮϴ��δ����
C������NaOH�������Na2CO3����
E����ʽ�ζ��ܼ��첿�������ݣ��ζ�����ʧ��
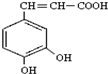 ��Ҳ��3��4-���ǻ���Ƥ�ᣬ��������������ҩ�У���Ұ���ܲ�����Ҷˮ�ա�����ľ���ĵȣ���������ֹѪ���ã��������ֹѪЧ���Ϻã����Խ�С��������ȡ���̵����Ч��
��Ҳ��3��4-���ǻ���Ƥ�ᣬ��������������ҩ�У���Ұ���ܲ�����Ҷˮ�ա�����ľ���ĵȣ���������ֹѪ���ã��������ֹѪЧ���Ϻã����Խ�С��������ȡ���̵����Ч�� ��д����������NaHCO3��Һ��Ӧ�ķ���ʽ����
��д����������NaHCO3��Һ��Ӧ�ķ���ʽ����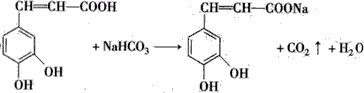 ��
��