��Ŀ����
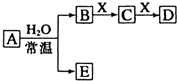 �ɶ�����Ԫ����ɵ���ѧ��������A��B��C��D��E��X������ͼת����ϵ������������ͷ�Ӧ������ȥ����
�ɶ�����Ԫ����ɵ���ѧ��������A��B��C��D��E��X������ͼת����ϵ������������ͷ�Ӧ������ȥ������1����A�dz����µ���̬���ʣ�X��Na2CO3����D��A��ˮ��Һ��Ӧ�Ļ�ѧ����ʽ
��2����EΪ������ˮ�İ�ɫ������B��X�ķ�Ӧ����ѧ��ѧ����Ҫ�Ļ�����Ӧ��д��D��ˮ��Ӧ���õ�ϡ��Һ��ͭƬ��Ӧ�����ӷ���ʽ
��3����DΪ��ɫ����������A��Ħ���������
��a�� A�ĵ���ʽ
��b����NaHCO3�ֽ������������������A������յõ���������ס���������VmL Cmol?L-1�������У�������ɫ��������ң���Һ�����ԣ���A�����ʵ���Ϊ
���㣺������ƶ�
ר�⣺
��������1��X��Na2CO3��BӦΪ�ᣬ��̼���Ʒ�Ӧ����CΪCO2��C��̼���Ʒ�Ӧ��������DΪNaHCO3����A�dz����µ���̬���ʣ���AΪCl2��
��2����EΪ������ˮ�İ�ɫ������B��X�ķ�Ӧ����ѧ��ѧ����Ҫ�Ļ�����Ӧ����EΪMg3N2��BΪNH3��CΪNO��DΪNO2��
��3����DΪ��ɫ����������A��Ħ��������ȣ���AΪNa2O2��EΪ������BΪNaOH��XΪ���Σ�CΪNaAlO2��DΪAl��OH��3��
��2����EΪ������ˮ�İ�ɫ������B��X�ķ�Ӧ����ѧ��ѧ����Ҫ�Ļ�����Ӧ����EΪMg3N2��BΪNH3��CΪNO��DΪNO2��
��3����DΪ��ɫ����������A��Ħ��������ȣ���AΪNa2O2��EΪ������BΪNaOH��XΪ���Σ�CΪNaAlO2��DΪAl��OH��3��
���
�⣺��1��X��Na2CO3��BӦΪ�ᣬ��̼���Ʒ�Ӧ����CΪCO2��C��̼���Ʒ�Ӧ��������DΪNaHCO3����A�dz����µ���̬���ʣ���AΪCl2����D��A��ˮ��Һ��Ӧ�Ļ�ѧ����ʽΪ��Cl2+NaHCO3=NaCl+CO2��+HClO���ʴ�Ϊ��Cl2+NaHCO3=NaCl+CO2��+HClO��
��2����EΪ������ˮ�İ�ɫ������B��X�ķ�Ӧ����ѧ��ѧ����Ҫ�Ļ�����Ӧ����EΪMg3N2��BΪNH3��CΪNO��DΪNO2��D��ˮ��Ӧ���õ�ϡ��Һ��ͭƬ��Ӧ�����ӷ���ʽ��3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��
�ʴ�Ϊ��3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��
��3����DΪ��ɫ����������A��Ħ��������ȣ���AΪNa2O2��EΪ������BΪNaOH��XΪ���Σ�CΪNaAlO2��DΪAl��OH��3��
��a�� AΪNa2O2������ʽΪ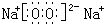 ��X��C��Ӧ����D�����ӷ���ʽΪ��3AlO2-+Al3++6H2O=4Al��OH��3�����ʴ�Ϊ��
��X��C��Ӧ����D�����ӷ���ʽΪ��3AlO2-+Al3++6H2O=4Al��OH��3�����ʴ�Ϊ��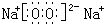 ��3AlO2-+Al3++6H2O=4Al��OH��3����
��3AlO2-+Al3++6H2O=4Al��OH��3����
��b����NaHCO3�ֽ������������������Na2O2������յõ����������Ϊ̼���ơ��������ƣ���������VmL Cmol?L-1�������У�������ɫ���������Ϊ������̼����������Һ�����ԣ���Һ������ΪNaCl�������������غ�n��Na2O2��=
n��NaCl���������������غ㣬��n��Na2O2��=
n��NaCl��=
n��HCl��=
��V��10-3L��Cmol/L=5VC��10-4mol��
��2Na2O2+4HCl=4NaCl+2H2O+O2����2Na2O2+CO2=2Na2CO3+O2����֪����������������Ϊ5VC��10-4mol��
=2.5VC��10-4mol�����ڹ�������ʣ�࣬��̼���Ƶ����ʵ���С��5VC��10-4mol����������Ӧ��Na2CO3+2HCl=2NaCl+H2O+CO2�������ɶ�����̼С��5VC��10-4mol����������������ʵ���С��2.5VC��10-4mol+5VC��10-4mol=7.5VC��10-4mol�����ҵ����ʵ�����2.5VC��10-4mol��n���ң���7.5VC��10-4mol��
�ʴ�Ϊ��5VC��10-4mol��2.5VC��10-4mol��n���ң���7.5VC��10-4mol��
��2����EΪ������ˮ�İ�ɫ������B��X�ķ�Ӧ����ѧ��ѧ����Ҫ�Ļ�����Ӧ����EΪMg3N2��BΪNH3��CΪNO��DΪNO2��D��ˮ��Ӧ���õ�ϡ��Һ��ͭƬ��Ӧ�����ӷ���ʽ��3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��
�ʴ�Ϊ��3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��
��3����DΪ��ɫ����������A��Ħ��������ȣ���AΪNa2O2��EΪ������BΪNaOH��XΪ���Σ�CΪNaAlO2��DΪAl��OH��3��
��a�� AΪNa2O2������ʽΪ
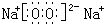 ��X��C��Ӧ����D�����ӷ���ʽΪ��3AlO2-+Al3++6H2O=4Al��OH��3�����ʴ�Ϊ��
��X��C��Ӧ����D�����ӷ���ʽΪ��3AlO2-+Al3++6H2O=4Al��OH��3�����ʴ�Ϊ��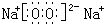 ��3AlO2-+Al3++6H2O=4Al��OH��3����
��3AlO2-+Al3++6H2O=4Al��OH��3������b����NaHCO3�ֽ������������������Na2O2������յõ����������Ϊ̼���ơ��������ƣ���������VmL Cmol?L-1�������У�������ɫ���������Ϊ������̼����������Һ�����ԣ���Һ������ΪNaCl�������������غ�n��Na2O2��=
| 1 |
| 2 |
| 1 |
| 2 |
| 1 |
| 2 |
| 1 |
| 2 |
��2Na2O2+4HCl=4NaCl+2H2O+O2����2Na2O2+CO2=2Na2CO3+O2����֪����������������Ϊ5VC��10-4mol��
| 1 |
| 2 |
�ʴ�Ϊ��5VC��10-4mol��2.5VC��10-4mol��n���ң���7.5VC��10-4mol��
���������⿼�������ƶϣ����ڲ²���֤����Ŀ����Ҫѧ����������Ԫ�ػ�����֪ʶ����3���м���Ϊ�״��㡢�ѵ㣬�����غ㷨�������Ƶ����ʵ�����ע��̼�����Ƶ����ʵ����ķ�Χȷ�����Ѷ��еȣ�

��ϰ��ϵ�д�
 Сѧ��ʱ��ѵϵ�д�
Сѧ��ʱ��ѵϵ�д�
�����Ŀ
��֪��2H2��g��+O2��g���T2H2O ��l����H=-571.6kJ?mol-1������˵����ȷ���ǣ�������
| A��2 LҺ̬ˮ�ֽ��2L������1L��������571.6 kJ���� |
| B��2 mol������1mol������Ӧ����2mol��̬ˮ�ų���������571.6 kJ |
| C������ͬ�����£�2 mol������1mol�����������ܺʹ���2molҺ̬ˮ������ |
| D��ʹ�ô����ܽ��÷�Ӧ�Ļ��ʹ��Ӧ�ų�������С��571.6 kJ |
����ʵ���У�������ȷ���ܴﵽ��Ӧʵ��Ŀ���ǣ�������
| A�����к��Ȳⶨʵ��ʱ����û�л��β�������������û���ͭ�ʽ�������� |
| B���к͵ζ�ʵ���У����ֵ��صζ��ܵĻ��������ֳ���ƿ���۾�ע�ӵζ�����Һ��ı仯 |
| C���������Ͷ����������ֺ���ɫ������õ��۵⻯����Һ������ |
| D����ȥ��������Һ��������NaCl���ɽ�װ�л��Һ�İ�Ĥ����������������ˮ�� |
Ҫ�ᴿ�������ʣ�����������Ϊ���ʣ���ʵ�������������ǣ�������
| A��CO2��SO2����ͨ��ʢ��Ʒ����Һ������ƿ���ռ� |
| B��Fe2+��Fe3+������KSCN��Һ����� |
| C��Cl2��HCl����ͨ��ʢ�б����Ȼ���ˮ��Һ��ϴ��ƿ���ռ� |
| D���Ȼ����Һ��FeCl3����������NaOH��Һ����� |
��ͼ��ijˮ��Һ��pH�� 0��14�ķ�Χ�ڣ�H2CO3��HCO3-��CO32-��������ƽ��ʱ����ɰٷ��ʣ�������������ȷ���ǣ�������
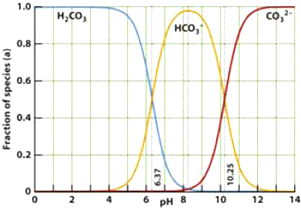

| A����ͼ����ȫ����1.0 mol/L����ζ�1.0 mol/L̼������Һ��ʵ�����ݻ�� |
| B��H2CO3��CO32-����Һ�в��ܴ������� |
| C����1.0 mol/L������뵽�����1.0 mol/L̼������Һ�У���ַ�Ӧ����Һ�ʼ��� |
| D����pHΪ6.37ʱ����Һ�У�c��H2CO3��=c��HCO3-������pHΪ10.25ʱ����Һ�У�c��CO32-��=c��HCO3-�� |
