��Ŀ����
������Һ�и�����Ũ�ȹ�ϵ����ȷ���ǣ�������
| A��0.1mol?L-1 HCOOH��Һ�У�c��HCOO-��+c��OH-��=c��H+�� |
| B��0.1mol?L-1 NH4Cl��Һ�У�c��NH4+��+c��NH3?H2O��=c��Cl-�� |
| C��0.1mol?L-1NaHCO3��Һ�У�c��Na+��+c��H+��+c��H2CO3��=c��HCO3-��+c��CO32-��+c��OH-�� |
| D��������������ʵ���Ũ�ȵ�CH3COONa������CH3COOH��Ϻ����Һ�У�c��CH3COO-��-c��CH3COOH��=2c��H+��-2c��OH-�� |
���㣺����Ũ�ȴ�С�ıȽ�
ר�⣺�����ˮ��ר��
������A��������Һ�������غ��жϣ�
B������Ԫ���غ㣬��Ԫ�غ���Ԫ�ص�������ȣ�
C��������Һ�ĵ���غ��жϣ�
D�����ݵ���غ�������غ�������ʽ�жϣ�
B������Ԫ���غ㣬��Ԫ�غ���Ԫ�ص�������ȣ�
C��������Һ�ĵ���غ��жϣ�
D�����ݵ���غ�������غ�������ʽ�жϣ�
���
�⣺A��HCOOH��Һ�и��������غ㣬��֪c��HCOO-��+c��OH-��=c��H+������A��ȷ��
B��NH4Cl��Һ�У���Ԫ�غ���Ԫ�ص�������ȣ�����Ԫ�ش����ڰ�ˮ��笠������У�������c��NH4+��+c��NH3?H2O��=c��Cl-������B��ȷ��
C��NaHCO3��Һ����ѭ����غ㣬��Һ�д���c��Na+��+c��H+��=c��HCO3-��+2c��CO32-��+c��OH-������C����
D�����ݵ���غ���c��CH3COO-��+c��OH-��=c��Na+��+c��H+�������������غ���c��Na+��=
��c��CH3COO-��+c��CH3COOH��������ʽ��ȥc��Na+���ɵ�c��CH3COO-��-c��CH3COOH��=2c��H+��-2c��OH-������D��ȷ��
��ѡC��
B��NH4Cl��Һ�У���Ԫ�غ���Ԫ�ص�������ȣ�����Ԫ�ش����ڰ�ˮ��笠������У�������c��NH4+��+c��NH3?H2O��=c��Cl-������B��ȷ��
C��NaHCO3��Һ����ѭ����غ㣬��Һ�д���c��Na+��+c��H+��=c��HCO3-��+2c��CO32-��+c��OH-������C����
D�����ݵ���غ���c��CH3COO-��+c��OH-��=c��Na+��+c��H+�������������غ���c��Na+��=
| 1 |
| 2 |
��ѡC��
���������⿼������Ũ�ȵĴ�С�Ƚϣ���Ŀ�Ѷ��еȣ�����ע�������Һ����غ�������غ��Լ������غ㣬Ϊ��������Ŀ���õ��ķ�����

��ϰ��ϵ�д�
�����Ŀ
��ʢ���Ȼ�����Һ���ձ���ͬʱ�������ۺ�ͭ�ۣ���Ӧ���������н�������ܳ��ֵ��ǣ�������
| A���ձ�����ͭ���� |
| B���ձ���������ͭ |
| C���ձ�������ͭ���� |
| D���ձ�������ͭ���� |
�г������۵ġ�84������Һ������Ʒ��ʶ���������������ٱ�ƷΪ��ɫ��Һ���ʼ��� ��ʹ��ʱ��ˮϡ�� �ۿɶԲ;ߡ����������������Ư��dzɫ�·���������Ч�ɷݿ����ǣ�������
| A��Cl2 |
| B��SO2 |
| C��NaClO |
| D��KMnO4 |
��������10mL pH=3�Ĵ�����Һ�м���ˮϡ�ͺ�����˵����ȷ���ǣ�������
| A����Һ�е������ӵ���Ŀ���� | ||
B����Һ��
| ||
| C������ĵ���̶�����c��H+�������� | ||
| D����ȫ�кʹ��������10mLpH=11��NaOH��Һ |
������Դ���ķ����ǣ�������
| A������ | B������ |
| C���ѷ� | D��������� |
�����й�ԭ�ӽṹ��Ԫ������˵����ȷ���ǣ�������
| A��ԭ�Ӻ�������Ų�ʽΪ1s2��Ԫ����ԭ�Ӻ�������Ų�ʽΪ1s22s2��Ԫ�ػ�ѧ�������� |
B����̬̼ԭ�ӵļ۵����Ų�ͼ��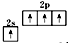 |
C����̬ͭԭ�ӵļ۵����Ų�ͼ��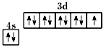 |
| D��Fe3+�����������Ų�ʽΪ��3s23p63d5 |
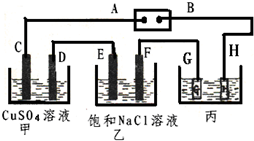 ��ͼ��ʾ��װ�ã�C��D��E��F���Ƕ��Ե缫������Դ��ͨ�������е����̪��Һ����F�������Ժ�ɫ���Իش��������⣺
��ͼ��ʾ��װ�ã�C��D��E��F���Ƕ��Ե缫������Դ��ͨ�������е����̪��Һ����F�������Ժ�ɫ���Իش��������⣺