��Ŀ����
ij�л�����һ�������¼ȿ������������ᣬ�ֿ��Ի�ԭ�ɴ�������ô���Ӧ�����ɷ���ʽΪC4H8O2������������˵��������ǣ�������
| A�����л���������������л�ԭ�� |
| B�����л����ܷ���������Ӧ |
| C�������л���ͷ���ʽΪC4H8O2������ɻ���ֻҪ������һ�������������������ߵ����ʵ���֮�ȣ���ȫȼ��ʱ�õ�������̼������Ϊ��ֵ�� |
| D�����л����Ǽ����ͬϵ�� |
���㣺�л���Ľṹ������
ר�⣺�л���Ӧ
�������л�����һ�������¼ȿ������������ᣬ�ֿ��Ի�ԭ�ɴ���������Ϊȩ������ô������ɷ���ʽΪC4H8O2����B������л���ΪCH3CHO���Դ������
���
�⣺�л�����һ�������¼ȿ������������ᣬ�ֿ��Ի�ԭ�ɴ���������Ϊȩ������ô������ɷ���ʽΪC4H8O2����B������л���ΪCH3CHO��
A����ȩ��-CHO���������������л�ԭ�ԣ���A��ȷ��
B����-CHO���ɷ���������Ӧ����B��ȷ��
C��������һ��������£�������CH3CHO��C4H8O2�����ʽ��ͬ��C������������ͬ����������ȫȼ�����õ�������̼������ȣ���C��ȷ��
D�������Ĺ����Ų�ͬ���ṹ��ͬ������ͬϵ���D����
��ѡD��
A����ȩ��-CHO���������������л�ԭ�ԣ���A��ȷ��
B����-CHO���ɷ���������Ӧ����B��ȷ��
C��������һ��������£�������CH3CHO��C4H8O2�����ʽ��ͬ��C������������ͬ����������ȫȼ�����õ�������̼������ȣ���C��ȷ��
D�������Ĺ����Ų�ͬ���ṹ��ͬ������ͬϵ���D����
��ѡD��
���������⿼���л���Ľṹ�����ʣ�Ϊ�߿��������ͣ�ע����սṹ�����ʵĹ�ϵ����Ϥ�ϳ���Cԭ�ӵ���Ŀ�仯��ȩ�����ʼ��ɽ����Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
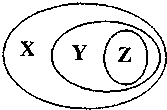 ����ѡ���У�������ͼ��ʾ���ʻ�����Ĵ�����ϵ���ǣ�������
����ѡ���У�������ͼ��ʾ���ʻ�����Ĵ�����ϵ���ǣ�������| X | Y | Z | |
| A | ������ | ���� | ������ |
| B | ����� | ��ɢϵ | ���� |
| C | Ԫ�����ڱ������� | ���� | ���� |
| D | �� | ����ͬϵ�� | ����ϩ |
| A��A | B��B | C��C | D��D |
�������ӷ���ʽ��д��ȷ���ǣ�������
A����������NaOH��Һ����ͬŨ�ȵ�����Ca��HCO3��2��Һ��Ca2++2HCO
| ||||
| B����Ba��OH��2��Һ����μ���NH4HSO4��Һ���պó�����ȫ Ba2++2OH-+NH4++H++SO42-=BaSO4��+NH3?H2O+H2O | ||||
| C����Ca��ClO��2��Һ��ͨ������SO2 Ca2++2ClO-+SO2+H2O=CaSO3��+2HClO | ||||
| D��Fe��OH��3��������Fe��OH��3+3 H+=Fe3++3 H2O |
�ò�˿����ɫ��Ӧʵ�飬ÿ���������Ҫ��������
| A��������ϴ�ӣ������������ƾ��ƻ�����ɫ��ͬ����ʹ�� |
| B����ˮϴ��2��3�κ���ʹ�� |
| C������ֽ���ɾ�����ʹ�� |
| D��������ϴ�ӣ���������ˮϴ����ʹ�� |
����˵����ȷ���ǣ�������
| A�������Ļ�������������� |
| B��1 mol H2O���2 mol���1 mol�� |
| C���������������Ե����ǿ��ȡ���ڵõ�������Ŀ�Ķ��� |
| D�������������ֱ������������������������Һ��Ӧʱ�ݳ����������� |
�����Ӵ����װ���ũҩʱ�����ü�����Һ��������Ϊ�����ǣ�������
| A��ȩ�� | B������ | C������ | D������ |
����������������������Һ������������ͭ�������ˮ����������Һ���״����ܷ�Ӧ���ǣ�������
| A������ | B����ȩ | C������ | D����ϩ�� |
��֪��Ӧ��2BrO3-+Cl2=Br2+2ClO3-����5Cl2+I2+6H2O=2HIO3+10HCl����ClO3-+5Cl-+6H+=3Cl2+3H2O�������й���������ԭ������ǿ��˳����ȷ���ǣ�������
| A����ԭ�ԣ�Cl-��I2��Br2��Cl2 |
| B����ԭ�ԣ�Cl-��I2��Cl2��Br2 |
| C�������ԣ�BrO3-��ClO3-��Cl2��IO3- |
| D�������ԣ�Cl2��BrO3-��C1O3-��IO3- |
�����漰�л�������ʻ�Ӧ�õ�˵��������ȷ���ǣ�������


| A�������Ҵ������ᶼ�ܷ���ȡ����Ӧ |
| B�������Ƶ�Cu��OH��2�ɼ�����Һ�е������� |
| C������������Ļ�������ڹ����·�Ӧ���ɵ���һ�ȼ�����Ȼ��� |
| D����֬�ڼ�Ĵ������¿ɷ���ˮ�⣬��ҵ�����ø÷�Ӧ�������� |