��Ŀ����
�������ӷ���ʽ�У���д��ȷ���ǣ� ��A����1mol��������Ͷ�뺬��1molAlCl3����Һ�У�6Na2O2+6H2O+4Al3+=4Al��OH��3��+3O2��+12Na+
B�����ˮ�еμ�FeCl3��Һ�Ʊ�Fe��OH��3���壺Fe3++3H2O
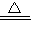 Fe��OH��3��+3H+
Fe��OH��3��+3H+C����ȩ��������Һ�ķ�Ӧ��CH3CHO+2Ag��NH3��2OH
 CH3COO-+NH4++2Ag��+3NH3+2H2O
CH3COO-+NH4++2Ag��+3NH3+2H2OD����FeCl3��Һ��ʴӡˢ��·�壺Fe3++Cu=Fe2++Cu2+
���𰸡�������A������������ˮ��Ӧ����NaOH��������������AlCl3��Ӧ����Al��OH��3���������ʵ����Ĺ�ϵ�жϣ�
B�����ɽ�������dz�����
C��Ag��NH3��2OH����Һ�е�������ӣ�
D����ɲ��غ㣮
����⣺A.1mol����������ˮ��Ӧ����2molNaOH��0.5mol��������AlCl3��Ӧ���� molAl��OH��3����Ӧ�����ӷ���ʽΪ6Na2O2+6H2O+4Al3+=4Al��OH��3��+3O2��+12Na+����A��ȷ��
molAl��OH��3����Ӧ�����ӷ���ʽΪ6Na2O2+6H2O+4Al3+=4Al��OH��3��+3O2��+12Na+����A��ȷ��
B�����ˮ�еμ�FeCl3��Һ�Ʊ�Fe��OH��3��������ӷ���ʽΪFe3++3H2O Fe��OH��3�����壩+3H+����B����
Fe��OH��3�����壩+3H+����B����
C����ȩ��������Һ�ķ�Ӧ�����ӷ���ʽΪCH3CHO+2[Ag��NH3��2]++2OH- CH3COO-+NH4++2Ag��+3NH3+2H2O����C����
CH3COO-+NH4++2Ag��+3NH3+2H2O����C����
D����Ӧ�����ӷ���ʽΪ2Fe3++Cu=2Fe2++Cu2+����D����
��ѡA��
���������⿼�����ӷ���ʽ����д����Ŀ�Ѷ��еȣ������״���ΪA��ע�������йص����ӷ���ʽ����д��
B�����ɽ�������dz�����
C��Ag��NH3��2OH����Һ�е�������ӣ�
D����ɲ��غ㣮
����⣺A.1mol����������ˮ��Ӧ����2molNaOH��0.5mol��������AlCl3��Ӧ����
 molAl��OH��3����Ӧ�����ӷ���ʽΪ6Na2O2+6H2O+4Al3+=4Al��OH��3��+3O2��+12Na+����A��ȷ��
molAl��OH��3����Ӧ�����ӷ���ʽΪ6Na2O2+6H2O+4Al3+=4Al��OH��3��+3O2��+12Na+����A��ȷ��B�����ˮ�еμ�FeCl3��Һ�Ʊ�Fe��OH��3��������ӷ���ʽΪFe3++3H2O
 Fe��OH��3�����壩+3H+����B����
Fe��OH��3�����壩+3H+����B����C����ȩ��������Һ�ķ�Ӧ�����ӷ���ʽΪCH3CHO+2[Ag��NH3��2]++2OH-
 CH3COO-+NH4++2Ag��+3NH3+2H2O����C����
CH3COO-+NH4++2Ag��+3NH3+2H2O����C����D����Ӧ�����ӷ���ʽΪ2Fe3++Cu=2Fe2++Cu2+����D����
��ѡA��
���������⿼�����ӷ���ʽ����д����Ŀ�Ѷ��еȣ������״���ΪA��ע�������йص����ӷ���ʽ����д��

��ϰ��ϵ�д�
�����Ŀ
�������ӷ���ʽ����ȷ���ǣ�������
| A��̼����狀�����������������Һ��Ӧ��NH4++OH-?NH3��+H2O | B��ƫ��������Һ��ͨ������Ķ�����̼���壺2AlO2-+CO2+2H2O?2Al��OH��3��+CO32- | C����ϡ�����������������Ӧ���Թܣ�Ag+2H++NO3-?Ag++NO2��+H2O | D�����Ȼ�����Һ��ͨ���������壺2Fe3++H2S?Fe2++S��+2H+ |
|
�������ӷ���ʽ����ȷ���� | |
A�� |
̼����狀�����������������Һ��Ӧ��NH4+��OH�� |
B�� |
�������ʵ���������������������������Һ��ϣ� H+��SO42����Ba2+��OH����BaSO4����H2O |
C�� |
��ϡ�����������������Ӧ���Թ� ��3Ag��4H+��NO3�� |
D�� |
������ͭ��Һ�м��������ˮ ��Cu2+��2NH3��H2O |
|
�������ӷ���ʽ����ȷ���� | |
A�� |
̼����狀�����������������Һ��Ӧ��NH4+��OH�� |
B�� |
�������ʵ���������������������������Һ��ϣ� H+��SO42����Ba2+��OH����BaSO4����H2O |
C�� |
��ϡ�����������������Ӧ���Թ� ��3Ag��4H+��NO3�� |
D�� |
������ͭ��Һ�м��������ˮ ��Cu2+��2NH3��H2O��Cu(OH)2����2NH4+ |
�������ӷ���ʽ����ȷ����
| A��̼����狀�����������������Һ��Ӧ��NH4����OH����NH3����H2O |
| B���������ʵ���������������������������Һ��ϣ� H+ + SO42-+Ba2+ +OH- ��BaSO4 ��+H2O |
| C����ϡ�����������������Ӧ���Թܣ�3Ag��4H����NO3����3Ag����NO����2H2O |
| D��������ͭ��Һ�м��������ˮ��Cu2+��2NH3��H2O��Cu(OH)2����2NH4+ |