��Ŀ����
����˵����ȷ����( )
A��H2(g)��ȼ���Ȧ�H����285.8 kJ/mol��
��2H2O(g)===2H2(g)��O2(g) ��H����571.6 kJ/ mol
B����Ӧ��������������������������ʱ���÷�ӦΪ���ȷ�Ӧ
C����ʯī�Ƚ��ʯ�ȶ���֪��C(���ʯ��s)��C(ʯī��s) ��H��0
D����֪��298 Kʱ��N2(g)��3H2(g)  2NH3(g) ��H ����92.0 kJ/mol������ͬ�¶��£����ܱ�������ͨ��0.5 mol N2��1.5 mol H2���ﵽƽ��ʱ�ų�46.0 kJ������
2NH3(g) ��H ����92.0 kJ/mol������ͬ�¶��£����ܱ�������ͨ��0.5 mol N2��1.5 mol H2���ﵽƽ��ʱ�ų�46.0 kJ������
��ϰ��ϵ�д�
�����Ŀ


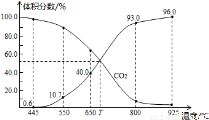
 ��ֵ______(���������С�����䡱)��
��ֵ______(���������С�����䡱)�� A��ͼ�ױ�ʾ�����ܸı仯ѧ��Ӧ���ʱ�
A��ͼ�ױ�ʾ�����ܸı仯ѧ��Ӧ���ʱ� C(g)�ġ�H >0���� a=2
C(g)�ġ�H >0���� a=2