��Ŀ����
(13��)ʵ����Ϊ̽������Ũ����ķ�Ӧ������֤SO2�����ʣ������ͼ��ʾװ�ý���ʵ�顣

��ش��������⣺
��1��ʵ��ʱA�е���a����Ũ�����У���ʹB�е���Һ��������������ԭ����_______��
��2��B������KMnO4����ɫ����B�з�Ӧ�����ӷ���ʽ��_______________________��C�з�Ӧ�Ļ�ѧ����ʽ��_________________��
��3��Dװ�õ�������________��ʵ�����ʱ���a����ͨ�������������Ŀ����___________��
��4����̽��Aװ���з�Ӧ����Ԫ�صļ�̬�����ʵ�鷽�����£�����Ӧ���Թ��е���Һϡ�͡�ȡ������Һ������3��5��________��Һ������Һ�ʺ�ɫ����֤������Fe3+������Fe3+��ȡ������Һ������3��5��________��Һ������Һ��ɫ����֤������Fe2+������Fe2+����ʵ��֤����Ӧ�����Һ��ֻ����Fe3+����A�з�Ӧ�Ļ�ѧ����ʽ��___________________________________________��
��1��Aװ���Թ�������ѹǿ��Сʱ�������ӵ���a����Aװ�ã�a������ƽ����ѹ������
��2��5SO2��2MnO4����2H2O��5SO42����2Mn2����4H�� 2H2S��SO2��2H2O��3S��
��3����֤SO2��Ư���Ժ�������IJ��ȶ��ԣ�ʹװ���в�����SO2������ȫ��Eװ�����գ��Է�ֹ��Ⱦ���� ��4��KSCN ����KMnO4 2Fe��6H2SO4(Ũ)  Fe2(SO4)3��6H2O��3SO2��
Fe2(SO4)3��6H2O��3SO2��
��������
�����������1������װ�ýṹ��֪��Aװ���Թ�������ѹǿ��Сʱ�������ӵ���a����Aװ�ã�a������ƽ����ѹ�����ã����Կ�ʹB�е���Һ������������
��2��SO2���л�ԭ�ԣ���ʹ���Ը��������Һ��ɫ����Ӧ�����ӷ���ʽΪ5SO2��2MnO4����2H2O��5SO42����2Mn2����4H����SO2�����������ԣ�������H2S���ɵ���S����Ӧ�����ӷ���ʽΪ2H2S��SO2��2H2O��3S����
��3��SO2����Ư���ԣ���SO2��Ư���Dz��ȶ��ģ�����Dװ�õ���������֤SO2��Ư���Ժ�������IJ��ȶ��ԣ�����װ���л�������SO2���壬����ʵ�����ʱ���a����ͨ�����������Ŀ����ʹװ���в�����SO2������ȫ��Eװ�����գ��Է�ֹ��Ⱦ������
��4������������һ����KSCN��Һ���������Ӿ��л�ԭ�ԣ���ʹ���Ը��������Һ��ɫ�����Կ���������KMnO4�����������ӡ����ڼ��ȵ���������Ũ���ᷴӦ�Ļ�ѧ����ʽΪ2Fe��6H2SO4(Ũ)  Fe2(SO4)3��6H2O��3SO2����
Fe2(SO4)3��6H2O��3SO2����
���㣺��������Ũ���ᷴӦ��ʵ�������̽��


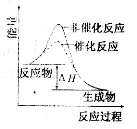
 2SO3(g)��ƽ����ϵ�м���18O2���ﵽ�µ�ƽ���18Oֻ������O2��SO3��
2SO3(g)��ƽ����ϵ�м���18O2���ﵽ�µ�ƽ���18Oֻ������O2��SO3��