��Ŀ����
1�����λ�����Ⱦ��������̬�����ѳ�Ϊȫ��ʶ����2013�꣬ȫ�����ʡ�г������ص��������������������γɵ���Ҫ��Ⱦ����C ������ĸ����
A��O2 B��CO2 C��PM2.5
�ڲ��÷��մ����������з������ڽ���Ͷ��ʹ�ã������������еij���������
A���ϵ�� B�������� C������ƿ
�ڷ��մ���ǰ�����ϵ���⣬����BC������ĸ��Ӧ�������գ�
��Ư�ۿ�����������ˮ��ɱ����������ҵ������������ʯ������ȡƯ�۵Ļ�ѧ��Ӧ����ʽ��2Cl2+2Ca��OH��2�TCaCl2+Ca��ClO��2+2H2O��
���� ��PM2.5����ָ��������ֱ����2.5��m�Ŀ�����ܱ������ղ�����ѪҺ��������Σ���ܴ����γ���������Ҫ��Ⱦ�
�ڲ���ȼ�յ����������ܲ��÷��մ�����
��������ʯ���鷴Ӧ�����Ȼ��ơ�������ơ�ˮ��
��� �⣺�ٵ��������γɵ���Ҫ��Ⱦ����pM2.5���ܱ������ղ�����ѪҺ��������Σ���ܴ�ѡC��
�������ޡ�����ƿ����ȼ�գ����Բ��ܲ��÷��մ�����
�ʴ�Ϊ��BC��
��������ʯ���鷴Ӧ�����Ȼ��ơ�������ơ�ˮ���÷�ӦΪ2Cl2+2Ca��OH��2�TCaCl2+Ca��ClO��2+2H2O���ʴ�Ϊ��2Cl2+2Ca��OH��2�TCaCl2+Ca��ClO��2+2H2O��
���� ���⿼�鳣���Ļ�����Ⱦ�Ư�۵��Ʊ���ҩ�����Ҫ�ɷֺ���Ч�ȣ��ѶȲ���ע��֪ʶ�Ļ��ۣ�����ע�ػ�ѧ���������ϵ����ȷ��ѧ���������ѧ�����û�ѧ֪ʶ��������е����⣮

��ϰ��ϵ�д�
�����Ŀ
11��ijѧ����0.1032mol•L-1���������Һ�ζ�δ֪Ũ�ȵ�KOH��Һ��������ֽ�Ϊ���¼�����
A��ȡ��������ˮϴ������ʽ�ζ��ܣ����������Һ��ϴ�ζ���2��3�Σ�
B����ȡ20mL����KOHע��ྻ����ƿ�У�������2��3�μ��ȣ�
C��ȡ��������Һע����ʽ�ζ�������0���̶�����1��2cm��
D����ʢ�б���Һ����ʽ�ζ��̶ܹ��ã����ڵζ��ܼ���ʹ֮������Һ��
E������Һ������0����0������ijһ�̶ȣ���¼������
F������ƿ���ڵζ��ܵ����棬�ñ�����ζ����յ㲢��¼�ζ���Һ��Ŀ̶ȣ�
����������⣺
��1�����������������ȷ˳����ACDEBF����д��ĸ��ţ���
��2���ж���Һ����ζ��յ�ķ����ǵ���Һ�ɻ�ɫ��Ϊ��ɫ�����ڰ�����ڲ���ɫ��
��3������A���������Ŀ���ǵζ���������ˮϴ�Ӻ��ڱ���һ��ˮĤ�����ֱ��װҺ��ʹŨ�Ƚ��ͣ�
��4��������B�������֮ǰ�����ô���Һ��ϴ��ƿ����Եζ����������Ӱ����ƫ�ߣ�����ȡһ������KOH���壨������NaOH�����ƴ�����Һ���������������Һ���еζ�����Եζ����������Ӱ����ƫ�ߣ���������Ϊͬ������NaOH��KOH�к�����ǿ�����ĵ�����࣮
��5����0.1032mol•L-1������ζ�δ֪Ũ�ȵ�KOH��Һ���ظ����ε�ʵ���������±���ʾ��
�����KOH�����ʵ���Ũ����0.1124mol•L-1��
A��ȡ��������ˮϴ������ʽ�ζ��ܣ����������Һ��ϴ�ζ���2��3�Σ�
B����ȡ20mL����KOHע��ྻ����ƿ�У�������2��3�μ��ȣ�
C��ȡ��������Һע����ʽ�ζ�������0���̶�����1��2cm��
D����ʢ�б���Һ����ʽ�ζ��̶ܹ��ã����ڵζ��ܼ���ʹ֮������Һ��
E������Һ������0����0������ijһ�̶ȣ���¼������
F������ƿ���ڵζ��ܵ����棬�ñ�����ζ����յ㲢��¼�ζ���Һ��Ŀ̶ȣ�
����������⣺
��1�����������������ȷ˳����ACDEBF����д��ĸ��ţ���
��2���ж���Һ����ζ��յ�ķ����ǵ���Һ�ɻ�ɫ��Ϊ��ɫ�����ڰ�����ڲ���ɫ��
��3������A���������Ŀ���ǵζ���������ˮϴ�Ӻ��ڱ���һ��ˮĤ�����ֱ��װҺ��ʹŨ�Ƚ��ͣ�
��4��������B�������֮ǰ�����ô���Һ��ϴ��ƿ����Եζ����������Ӱ����ƫ�ߣ�����ȡһ������KOH���壨������NaOH�����ƴ�����Һ���������������Һ���еζ�����Եζ����������Ӱ����ƫ�ߣ���������Ϊͬ������NaOH��KOH�к�����ǿ�����ĵ�����࣮
��5����0.1032mol•L-1������ζ�δ֪Ũ�ȵ�KOH��Һ���ظ����ε�ʵ���������±���ʾ��
| ��� | ������������/mL | ����KOH��Һ�����/mL |
| 1 | 27.23 | 25.00 |
| 2 | 28.24 | 25.00 |
| 3 | 27.25 | 25.00 |
9��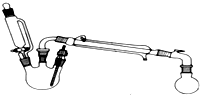 ij�о���С��̽�����������ķ�Ӧ������ʵ�����£���֪������������ʣ����³�ѹ��
ij�о���С��̽�����������ķ�Ӧ������ʵ�����£���֪������������ʣ����³�ѹ��
�ϳɷ�Ӧ����������ƿ�м����Ҵ�5mL������5mL��2СƬ���Ƭ��©����������14.3mL���Ҵ�20mL����������ͨ����ȴˮ��ʼ�������ȣ����Ƶμ��ٶȵ��������ٶȣ���Ӧ�¶Ȳ�����120�森�����ᴿ������Ӧ�ֲ��ﵹ���Һ©���У��������������͵�Na2CO3��Һ������NaCl��Һ������CaCl2��Һϴ�ӣ�����������ˮ̼��أ�����һ��ʱ�����ȥ̼��أ�����ͨ������õ�����������������
�ش��������⣺
��1����ȡ����������ѧ����ʽΪ��CH3COOH+CH3CH2OH$?_{��}^{ŨH_{2}SO_{4}}$CH3COOCH2CH3+H2O��
��2��Ũ�����봼��λ�ϣ�����������ƿ�����Ҵ���������ƿ�ڻ�������Ũ���ᣬ���ߵμӣ�
��3���������һ��ʱ��������ǼӴ�Ƭ��Ӧ�ò�ȡ����ȷ������B������ȷ�𰸱�ţ���
A���������� B����ȴ�� C�����貹�� D����������
��4�����Ƶμ�������Ҵ����Һ���ٶȵ��������ٶ�Ŀ���ǣ���֤�Ҵ�����������10�����ϣ���������ת���ʣ�
��5�������Ĵ�������������Ҫ����Щ���ʣ����ѡ����ᡢ�Ҵ���ˮ��
��6�����͵�Na2CO3��Һϴ�ӳ�ȥ���ᣬ�ܷ�NaOH��Һ�������ܣ�Ϊʲô�����û�ѧ����ʽ��ʾ�����ܣ�CH3COOCH2CH3+NaOH��CH3COONa+CH3CH2OH��
�ñ���NaCl��Һϴ�ӳ�ȥ������Na2CO3��Һ��Ϊʲô����ˮ���������������ܽ⣮
 ij�о���С��̽�����������ķ�Ӧ������ʵ�����£���֪������������ʣ����³�ѹ��
ij�о���С��̽�����������ķ�Ӧ������ʵ�����£���֪������������ʣ����³�ѹ��| �ܶ�g/mL | �۵�/�� | �е�/�� | ˮ���� | |
| �Ҵ� | 0.79 | -114 | 78 | �� |
| ���� | 1.049 | 16.2 | 117 | �� |
| �������� | 0.902 | -84 | 76.5 | ���� |
�ش��������⣺
��1����ȡ����������ѧ����ʽΪ��CH3COOH+CH3CH2OH$?_{��}^{ŨH_{2}SO_{4}}$CH3COOCH2CH3+H2O��
��2��Ũ�����봼��λ�ϣ�����������ƿ�����Ҵ���������ƿ�ڻ�������Ũ���ᣬ���ߵμӣ�
��3���������һ��ʱ��������ǼӴ�Ƭ��Ӧ�ò�ȡ����ȷ������B������ȷ�𰸱�ţ���
A���������� B����ȴ�� C�����貹�� D����������
��4�����Ƶμ�������Ҵ����Һ���ٶȵ��������ٶ�Ŀ���ǣ���֤�Ҵ�����������10�����ϣ���������ת���ʣ�
��5�������Ĵ�������������Ҫ����Щ���ʣ����ѡ����ᡢ�Ҵ���ˮ��
��6�����͵�Na2CO3��Һϴ�ӳ�ȥ���ᣬ�ܷ�NaOH��Һ�������ܣ�Ϊʲô�����û�ѧ����ʽ��ʾ�����ܣ�CH3COOCH2CH3+NaOH��CH3COONa+CH3CH2OH��
�ñ���NaCl��Һϴ�ӳ�ȥ������Na2CO3��Һ��Ϊʲô����ˮ���������������ܽ⣮
6���������Ƶ��л���ʵ���ϲ����ܴ��ڵ��ǣ�������
| A�� | 2��3-�������� | B�� | 2-��-4-�һ�-1-��ϩ | ||
| C�� | 3-��-1-��ϩ | D�� | 3��3-����-2-��ϩ |
13��Ϊ�˺������û�ѧ�ܣ�ȷ����ȫ���������������Ҫ��ֿ��ǻ�ѧ��Ӧ���ʱ䣬����ȡ��Ӧ��ʩ����ѧ��Ӧ���ʱ�ͨ����ʵ����вⶨ��Ҳ�ɽ����������㣮
��1��ʵ���ã�5g�״��������г��ȼ�����ɶ�����̼�����Һ̬ˮʱ�ͷų�113.5kJ����������д���״�ȼ�յ��Ȼ�ѧ����ʽ��2CH3OH��l��+3O2��g��=2CO2��g��+4H2O��l����H=-1452.8kJ/mol��
��2������̬��̬ԭ���γ�1mol��ѧ���ͷŵ���������м��ܣ��ӻ�ѧ���ĽǶȷ�������ѧ��Ӧ�Ĺ��̾��Ƿ�Ӧ��Ļ�ѧ�����ƻ���������Ļ�ѧ�����γɹ��̣��ڻ�ѧ��Ӧ�����У���ѧ����Ҫ�����������γɻ�ѧ���ֻ��ͷ�������
��֪��ӦN2��g��+3H2��g��?2NH3��g����H=a kJ•mol-1���Ը��ݱ������м������ݹ���a ����ֵ��-93��
��1��ʵ���ã�5g�״��������г��ȼ�����ɶ�����̼�����Һ̬ˮʱ�ͷų�113.5kJ����������д���״�ȼ�յ��Ȼ�ѧ����ʽ��2CH3OH��l��+3O2��g��=2CO2��g��+4H2O��l����H=-1452.8kJ/mol��
��2������̬��̬ԭ���γ�1mol��ѧ���ͷŵ���������м��ܣ��ӻ�ѧ���ĽǶȷ�������ѧ��Ӧ�Ĺ��̾��Ƿ�Ӧ��Ļ�ѧ�����ƻ���������Ļ�ѧ�����γɹ��̣��ڻ�ѧ��Ӧ�����У���ѧ����Ҫ�����������γɻ�ѧ���ֻ��ͷ�������
| ��ѧ�� | H-H | N-H | N��N |
| ����/kJ•mol-1 | 436 | 391 | 945 |
7����25��ʱ��H2R�������ε���Һ�У�H2R��HR-��R2-������ռ�����ʵ���������a������ҺpH�仯��ϵ��ͼ��ʾ����������������ǣ�������


| A�� | H2R�Ƕ�Ԫ���ᣬ��Kal=1��10-2 | |
| B�� | ����Һǡ�ó�����ʱ��c��Na+��=2c��R2-��+c��HR-�� | |
| C�� | NaHR����Һ��ˮ��������ڵ������� | |
| D�� | ��Na2R��NaHR��0.1mol�Ļ����Һ��pH=7.2 |
8������ϩ��������5���Ҽ���һ���м������Ƿֱ��ǣ�������
| A�� | C-H֮��ֻ��sp2�γɵĦҼ���C-C֮��ֻ��δ�μ��ӻ���2p����γɵĦм� | |
| B�� | C-C����sp2�γɵĦҼ���C-H֮����δ�μ��ӻ���2p����γɵĦм� | |
| C�� | sp2�ӻ�����γɦҼ���δ�ӻ���2p����γɦм� | |
| D�� | sp2�ӻ�����γɦм���δ�ӻ���2p����γɦҼ� |