��Ŀ����
��ɶ���������ƣ�Na2S2O3�����������ƻ�������������ƺ����IJⶨʵ�飮
��֪��Na2S2O3+H2SO4�TNa2SO4+SO2��++S��+H2O
�й�ʵ�����������Ӻ̶������õĽ��ܡ�����̨��װ�þ���ȥ����ͼ��ʾ��

��1������д�±�������������˳�����ϵ����������룬������ֻ��ʹ��һ�Σ����ɹ�ѡ�õ��Լ���Ʒ����Һ��NaOH��Һ��Ũ���ᡢ��ˮ����ͭ����ʯ�ң�
��2����ʵ���г�ȡm1g�������Ʒ����ʵ�飮
����װ������������Ӧ���Ƚ��е�ʵ������� ��
����ʵ��ǰ��װ��C�������ֱ�Ϊm2g��m3g��װ��B�������ֱ�Ϊm4g��m5g������Ʒ��Na2SO3������������Ϊa���ļ���ʽΪ ��
��֪��Na2S2O3+H2SO4�TNa2SO4+SO2��++S��+H2O
�й�ʵ�����������Ӻ̶������õĽ��ܡ�����̨��װ�þ���ȥ����ͼ��ʾ��

��1������д�±�������������˳�����ϵ����������룬������ֻ��ʹ��һ�Σ����ɹ�ѡ�õ��Լ���Ʒ����Һ��NaOH��Һ��Ũ���ᡢ��ˮ����ͭ����ʯ�ң�
| ���� | �����Լ� | ���� |
| A | ||
| E |
����װ������������Ӧ���Ƚ��е�ʵ�������
����ʵ��ǰ��װ��C�������ֱ�Ϊm2g��m3g��װ��B�������ֱ�Ϊm4g��m5g������Ʒ��Na2SO3������������Ϊa���ļ���ʽΪ
���㣺̽�����ʵ���ɻ�������ʵĺ���
ר�⣺ʵ��̽�������ݴ�����
��������1��Aװ��Ӧ����������ƣ�Na2S2O3�����������ƻ�����Aװ���з������·�Ӧ��Na2S2O3+H2SO4�TNa2SO4+SO2��+S��+H2O��Na2SO3+H2SO4�TNa2SO4+SO2��+H2O����˱�ʵ����ͨ���ⶨ�����������������Ӳⶨ�������Ƶĺ�����Ҫȷ�ⶨ��������Ҫ�Զ���������и������ȫ���գ��������ҩƷ�������������Ӧѡ��Ũ���������ѡ��Cװ�ã����ն���������ü�ʯ�ң�ѡ��Bװ�ã�Ϊʹ����������ȫ�����գ�Ӧ��������װ���еĶ��������������װ���У�ѡ��Dװ�ã�Ϊ����ʯ�����տ����е�ˮ�ֺͶ�����̼��Ӧ��װ��ʯ�ҵ�װ�ú���ʢ�м�ʯ�ҵĸ���ܣ�ѡ��Eװ�ã�����˳��Ϊ�Ȳ������壬��������壬���������壻
��2����װ�����������������Բ���װҩƷ����ʵ�飻
��3�����ն���������Bװ���У���Ӧѡ������m1g��m4g��m5g�����ݷ�Ӧ����ʽ���м��㣮
��2����װ�����������������Բ���װҩƷ����ʵ�飻
��3�����ն���������Bװ���У���Ӧѡ������m1g��m4g��m5g�����ݷ�Ӧ����ʽ���м��㣮
���
�⣺��1��Aװ��Ӧ����������ƣ�Na2S2O3�����������ƻ�����Aװ���з������·�Ӧ��Na2S2O3+H2SO4�TNa2SO4+SO2��+S��+H2O��Na2SO3+H2SO4�TNa2SO4+SO2��+H2O����˱�ʵ����ͨ���ⶨ�����������������Ӳⶨ�������Ƶĺ�����Ҫȷ�ⶨ��������Ҫ�Զ��������Ƚ��и���ͺ���ȫ���գ��������ҩƷ�������������Ӧѡ��Ũ���������ѡ��Cװ�ã����ն���������ü�ʯ�ң�ѡ��Bװ�ã�Ϊʹ����������ȫ�����գ�Ӧ��������װ��A��C�еĶ��������������װ��B�У�ѡ��Dװ���ҷ���A��ǰ�棻Ϊ����ʯ�����տ����е�ˮ�ֺͶ�����̼��Ӧ��װ��ʯ�ҵ�װ�ú���ʢ�м�ʯ�ҵĸ���ܣ�ѡ��Eװ�ã�����B�ĺ��棬
�ʴ�Ϊ��
��2������װ�����������������Բ���װҩƷ����ʵ�飬�ʴ�Ϊ�����װ�õ������ԣ�
��Na2SO3������������Ϊa������Na2SO3����Ϊm1ag��Na2SO3�����ʵ���Ϊ��
mol��Na2S2O3�����ʵ���Ϊ
mol��
��Ӧ�����������������Ϊ����m5-m4��g�����ʵ���Ϊ����
��mol���������غ㣬�õ�ʽΪ
mol+
mol=��
��mol��
�ʴ�Ϊ��
=
+
��
�ʴ�Ϊ��
| ���� | �����Լ� | ���� |
| D | ����������Һ | ��ȥ�����е��������壬ʹ���ɵĶ������������ֱ�B���� |
| A | ��Ʒ | ���������������� |
| C | Ũ���� | ��ȥ�����е�ˮ���� |
| B | ��ʯ�� | �������ɵĶ����������� |
| E | ��ʯ�� | ��ֹ�������������弰ˮ��������B |
��Na2SO3������������Ϊa������Na2SO3����Ϊm1ag��Na2SO3�����ʵ���Ϊ��
| m1a |
| 126 |
| m1(1-a) |
| 158 |
��Ӧ�����������������Ϊ����m5-m4��g�����ʵ���Ϊ����
| m5-m4 |
| 64 |
| m1a |
| 126 |
| m1(1-a) |
| 158 |
| m5-m4 |
| 64 |
�ʴ�Ϊ��
| m5-m4 |
| 64 |
| m1a |
| 126 |
| m1(1-a) |
| 158 |
���������⿼���˻������ij�ɷֺ����ļ��㣮Ҫ����ʵ��Ŀ�ĺ�����ҩƷ����������ȷѡ��ʵ������ҩƷ���������غ㷨�dz��õ�һ�ֽ��ⷽ����Ҫ������ã�

��ϰ��ϵ�д�
�����Ŀ
��ѧ���ѧ����������ᡢ����������أ������й�˵���в���ȷ���ǣ�������
| A���ޡ��顢˿��ë��ȫȼ�ն�ֻ����CO2��H2O |
| B�����ع��͡�Σ����������Ҫ�������������ά����E��֬������֬�Ѿ����ƻ� |
| C��ʯ���ѽ⡢ú����������ˮ��þ�ȹ����ж�������ѧ�仯 |
| D�����Ƹ����ܵ���ĥ��̥���ɼ��ٿ���ο����PM2.5���IJ��� |
����NH4+��Mg2+��Fe2+��Al3+����Һ�м�������Na2O2���Ȳ�������ټ������ϡ���ᣬ��Һ�д������ٵ������ǣ�������
| A��NH4+ |
| B��NH4+��Fe2+ |
| C��Al3+��Mg2+ |
| D��Al3+ |
�ۺ��� �ĵ����ǣ�������
�ĵ����ǣ�������
 �ĵ����ǣ�������
�ĵ����ǣ�������A�� |
B�� |
C��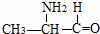 |
D�� |
 �绯ѧ�ڹ�ҵ�������й㷺Ӧ�ã�
�绯ѧ�ڹ�ҵ�������й㷺Ӧ�ã�


