��Ŀ����
2��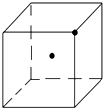 ˮ������֮Դ���������ǵ�����������أ��ڻ�ѧʵ��Ϳ�ѧ�о��У�ˮҲ��һ�ֳ��õ��Լ���
ˮ������֮Դ���������ǵ�����������أ��ڻ�ѧʵ��Ϳ�ѧ�о��У�ˮҲ��һ�ֳ��õ��Լ�����1����H2O�����е�������ȵ�5�˵ķ��Ӻ����ӵĻ�ѧʽ�ֱ���CH4��NH4+��
��2��ˮҲ��һ�����õ��ܼ���������������ˮ��ԭ���ǣ��ٰ�������Ϊ���Է��ӣ������ڼ����ܼ�ˮ�Тڰ���������ˮ���Ӽ����γ�����۰�������ˮ��Ӧ��
��3�����ľ����������ơ��⡢���ʯ���ɱ����Ȼ��ƾ����е�һ�־������ƣ���ͼ�DZ�������һ���֣�����С�ڵ��ʾˮ���ӣ��뽫ʣ���ˮ������С�ڵ㲹��������
���� ��1��H2O�����е�����Ϊ10��
��2���������Ӱ�����ʵ��ܽ��ԣ�
��3�����ľ�����������ʯ�������ƣ�������������ṹ��
��� �⣺��1��H2O�����е�����Ϊ10����H2O�����е�������ȵ�5�˵ķ��Ӻ����ӵĻ�ѧʽ�ֱ���CH4��NH4+���ʴ�Ϊ��CH4��NH4+��
��2��ˮҲ��һ�����õ��ܼ���������������ˮ��ԭ���Ǣٰ�������Ϊ���Է��ӣ������ڼ����ܼ�ˮ�Тڰ���������ˮ���Ӽ����γ�����۰�������ˮ��Ӧ��
�ʴ�Ϊ���ٰ�������Ϊ���Է��ӣ������ڼ����ܼ�ˮ�Тڰ���������ˮ���Ӽ����γ�����۰�������ˮ��Ӧ��
��3�����ľ�����������ʯ�������ƣ�������������ṹ�����������С�ڵ��ʾˮ���ӣ�ʣ���ˮ������С�ڵ㲹������Ϊ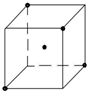 ��
��
�ʴ�Ϊ��
���� ���⿼����10�����������������ṹ�ȣ�������������ʵ�����Ӱ�졢����ṹ��Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�
�����Ŀ
13����ѧ֪ʶ��������������������Ҫ��Ӧ�ã�����˵���в���ȷ���ǣ�������
| A�� | ���Ʋ;߲������������ʱ�������ԡ����Ի��̵�ʳ�� | |
| B�� | ���ͷ��к��е�̼�����ƣ���ʹ���Ƴ��ĸ�����ɶ�� | |
| C�� | Na2O2�ȿ������������O2����Դ���ֿ�Ư��֯���ˡ���ë�� | |
| D�� | ������ˮ������Al��OH��3���壬������������ˮ�������� |
10����NA��ʾ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A�� | ��״���£�11.2LCCl4�к��еķ�����ĿΪ0.5NA | |
| B�� | �����£�1L0.1mol/LCH3COOH��Һ�к��е�H+��ĿΪ0.1NA | |
| C�� | 25�棬PH=13��NaOH��Һ�к���OH-����ĿΪ0.1NA | |
| D�� | 1L 1mol/L��NH4Cl��Һ�У�����NH4+����ĿС��NA |
17�����и��������У����ֵΪ2��1���ǣ�������
| A�� | ��ˮ�루NH4��2SO4��Һ��Ϻ�pH=7����Һ�У�[NH4+]��[SO42-] | |
| B�� | ��ͬ�¶��£�0.2 mol•L-1������Һ��0.1 mol•L-1������Һ�У�[H+]֮�� | |
| C�� | Na2CO3��Һ�У�[Na+]��[CO32-] | |
| D�� | pH=12��Ba��OH��2��Һ��pH=12��KOH��Һ�У�c[Ba��OH��2]��c��KOH�� |
14����ͬѧ�о��ӿ�ʯ��ʼ�����������Ĺ������̣�
��1����6.62g����ʯ��ƷͶ����������������ȫ�ܽ����ˣ������в�����Ԫ�أ�����Һ�мӹ�����NaOH��Һ����ַ�Ӧ�����ˡ�ϴ�ӡ����յ�4.80g Fe2O3���������ʯ����������������
��2�����Ը�����ʯΪԭ����������������������Ԫ����ʧ4%������ÿ����1.00������������96%����������Ҫ��������ʯ���ٶ֣���������λС����
��3��ȡij������ĩ28.12g������ֻ��Fe��C�������������г�ַ�Ӧ���õ�CO2����224mL����״����������˸�����ĩ������̼�����ʵ���֮�ȣ�
��4����ȡ���ݲ�ͬ����������������ĩ���ֱ�ӵ�100mL��ͬŨ�ȵ�H2SO4��Һ�У���ַ�Ӧ��õ�ʵ���������±���ʾ��
����������Һ�����ʵ���Ũ��
��5������ʵ����м�������mg������ĩ�����㷴Ӧ������ʣ��Ĺ�������Ϊ���٣����ú�m�Ĵ���ʽ��ʾ��
��1����6.62g����ʯ��ƷͶ����������������ȫ�ܽ����ˣ������в�����Ԫ�أ�����Һ�мӹ�����NaOH��Һ����ַ�Ӧ�����ˡ�ϴ�ӡ����յ�4.80g Fe2O3���������ʯ����������������
��2�����Ը�����ʯΪԭ����������������������Ԫ����ʧ4%������ÿ����1.00������������96%����������Ҫ��������ʯ���ٶ֣���������λС����
��3��ȡij������ĩ28.12g������ֻ��Fe��C�������������г�ַ�Ӧ���õ�CO2����224mL����״����������˸�����ĩ������̼�����ʵ���֮�ȣ�
��4����ȡ���ݲ�ͬ����������������ĩ���ֱ�ӵ�100mL��ͬŨ�ȵ�H2SO4��Һ�У���ַ�Ӧ��õ�ʵ���������±���ʾ��
| ʵ����� | �� | �� | �� |
| ���������ĩ������/g | 2.812 | 5.624 | 8.436 |
| ������������/L����״���� | 1.120 | 2.240 | 2.800 |
��5������ʵ����м�������mg������ĩ�����㷴Ӧ������ʣ��Ĺ�������Ϊ���٣����ú�m�Ĵ���ʽ��ʾ��
12������˵��������ǣ�������
| A�� | ����������ʢ������ͭ��Һ�������ױ���ʴ | |
| B�� | ���ô���ʩ�����Խ�����β���е�CO��NOת��Ϊ������ | |
| C�� | ��ֹ���귢������Ҫ��ʩ֮һ��ʹ�������Դ | |
| D�� | ����ͭ�ʺ�ɫ������Ϊ�����մɵĺ�ɫ���� |
