��Ŀ����
3��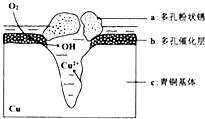 ͭ��һ����Ҫ����ɫ��������������;Խ��Խ�㷺����ش��������⣺
ͭ��һ����Ҫ����ɫ��������������;Խ��Խ�㷺����ش��������⣺��1���������ֻ������к�ͭ����ߵ���C������ĸ��
A��Cu5FeS4��������B��CuFeS2������C��Cu2S������D��Cu2��OH��2CO3
��2��2014���ҹ�����ͭ����796��֣���ȫ���ɺ�Cu2S��������Ϊ32%��ͭ��ʯұ���õ�������Ҫͭ��ʯ����Ϊ3109.4��֣�������һλС����
��3��������ͭ�γ���������������ͭ�������KOH��Һ�м���һ������CuSO4��Һ���ټ���һ�����Ļ�ԭ��--�£�N2H4�������Ȳ������¶���90�棬����һ�ֶԻ�������Ⱦ�����壬��Ӧ��ȫ���룬ϴ�ӣ���ո���õ�����������ͭ���壨Cu2O����
�ٸ��Ʊ����̵ķ�Ӧ����ʽΪ4CuSO4+N2H4+8KOH$\frac{\underline{\;90��\;}}{\;}$2Cu2O+N2��+4K2SO4+6H2O��
�ڹ�ҵ�ϳ��õĹ�Һ�����豸��AC������ĸ��
A�����Ļ�������B������������C����ʽѹ�˻�������D����Ӧ��
��4���ҹ���������ͭ�����վ�տ�����кܸߵ�������ֵ����ʷ��ֵ������������ͭ������ܵ�������ʴ����ͼ����ͭ���ڳ�ʪ�����з����绯ѧ��ʴ��ԭ��ʾ��ͼ��
�ٸ�ʴ�����У�������c���a����b����c������������Ӧ����ʽΪO2+2H2O+4e-�T4OH-��
�ڻ����е�Cl-��ɢ���ڣ�����������������������ɶ��״��Cu2��OH��3Cl�������ӷ���ʽΪ2Cu2++3OH-+Cl-=Cu2��OH��3Cl����
���� ��1����ͭ��=$\frac{����ͭ�����ԭ������}{���ʵ�ʽ��}$��
��2�����ݻ�ѧʽCu2S����ͭԪ�ص�����������������һ��������Cu2S������ͭԪ�ص�������������ijԪ�ص��������������Ǹ�Ԫ�ص�������������ʵ�Ԫ��������֮�ȣ�
��3�������������֪��������������ͭ��N2H4����������ԭ��Ӧ����Cu2O��N2��
�ڹ�ҵ�ϳ��õĹ�Һ�����豸�����Ļ��Ϳ�ʽѹ�˻���
��4���ٸ���ͼ֪�������õ����������������ӡ�Cuʧ��������ͭ���ӣ�����������ʴ����Cu��������
��Cl-��ɢ���ڣ�����������Ӧ���������Ӧ�����������ɶ��״��Cu2��OH��3Cl������������ͭ���ӡ��������������������ӣ����Ը����ӷ�ӦΪ�����ӡ�ͭ���Ӻ����������ӷ�Ӧ����Cu2��OH��3Cl������
��� �⣺��1��A��Cu5FeS4�к�ͭ��Ϊ$\frac{320}{504}$=0.63��
B��CuFeS2�к�ͭ��Ϊ$\frac{64}{184}$=0.35��
C��ͭ��Ϊ$\frac{128}{160}$=0.8��
D��Cu2��OH��2CO3�к�ͭ��Ϊ$\frac{128}{221}$=0.58��
�ʴ�Ϊ��C��
��2��Cu2S��ͭԪ�ص���������=$\frac{64��2}{160}$��100%=80%����X�ֺ�Cu2S 32%��ͭ��ʯ�к�ͭԪ������=Xt��80%��32%=796��t�����X=3109.4��֣�
�ʴ�Ϊ��3109.4��
��3�����������֪��������������ͭ��N2H4����������ԭ��Ӧ����Ӧ�Ļ�ѧ����ʽΪ4CuSO4+N2H4+8KOH$\frac{\underline{\;90��\;}}{\;}$2Cu2O+N2��+4K2SO4+6H2O��
�ʴ�Ϊ��4CuSO4+N2H4+8KOH$\frac{\underline{\;90��\;}}{\;}$2Cu2O+N2��+4K2SO4+6H2O��
�ڹ�ҵ�ϳ��õĹ�Һ�����豸�����Ļ��Ϳ�ʽѹ�˻����ʴ�Ϊ��AC��
��4���ٸ���ͼ֪�������õ����������������ӡ�Cuʧ��������ͭ���ӣ�����������ʴ����Cu����������c�Ǹ���������������ԭ��Ӧ���缫����ʽΪO2+2H2O+4e-�T4OH-��
�ʴ�Ϊ��c��O2+2H2O+4e-�T4OH-��
��Cl-��ɢ���ڣ�����������Ӧ���������Ӧ�����������ɶ��״��Cu2��OH��3Cl������������ͭ���ӡ��������������������ӣ����Ը����ӷ�ӦΪ�����ӡ�ͭ���Ӻ����������ӷ�Ӧ����Cu2��OH��3Cl���������ӷ���ʽΪ2Cu2++3OH-+Cl-=Cu2��OH��3Cl����
�ʴ�Ϊ��2Cu2++3OH-+Cl-=Cu2��OH��3Cl����
���� ���⿼��Cu���仯��������ʡ�ԭ���ԭ����Ӧ�ã��������ʵ������Լ��绯ѧ��Ӧԭ����Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬��Ŀ�Ѷ��еȣ�

 ˫��ͬ������ѵ��ϵ�д�
˫��ͬ������ѵ��ϵ�д� �Ƹ�С״Ԫͬ������������ϵ�д�
�Ƹ�С״Ԫͬ������������ϵ�д�| A�� |  | B�� |  | C�� |  | D�� |  |
| A�� | ���ͷ�Ӧ�¶� | B�� | �ӳ���Ӧʱ�� | C�� | �������ʯ | D�� | ��ˮϡ������ |
| A�� | ������������������ʵ���֮��1��2 | |
| B�� | �������ϡ���ᷴӦ�����Һ�����ܽ�1.28g Cu | |
| C�� | ��0.28mol����ͨ��ˮ�в���ȫ������ʱ����Ҫ����0.14mol���� | |
| D�� | 10.7g����ΪAl��OH��3��Fe��OH��3�Ļ���� |
| A�� | ������ˮ�д������ֵ���ƽ�� | |
| B�� | ��ͬԭ��֮���γɵĹ��ۼ���һ���Ǽ��Թ��ۼ� | |
| C�� | ������ڿ����е�Na2SiO3��NaOH��CaCl2��Ư����Һ����������ᣬ���ܲ������� | |
| D�� | �������ữ��FeCl2��Һ�������������Һ�У���Һ��ƣ����������������ݣ� |
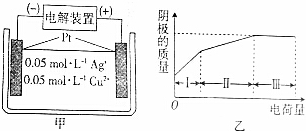
| A�� | ���������������������� | |
| B�� | ���������Һ��pH������ | |
| C�� | ��������������������ų� | |
| D�� | �������������������������2�� |
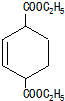 ��
��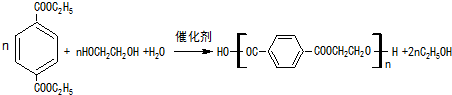 ��
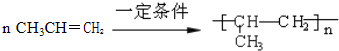
��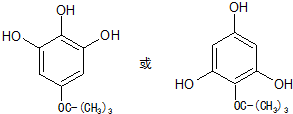 ����дһ�֣�
����дһ�֣�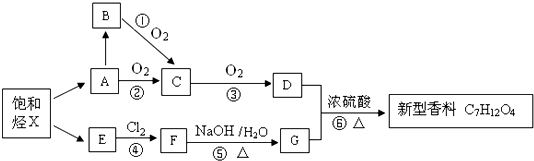
 ��
�� ��
��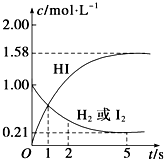 ij�¶�ʱ����ijV L���ܱ������г���3mol
ij�¶�ʱ����ijV L���ܱ������г���3mol