��Ŀ����
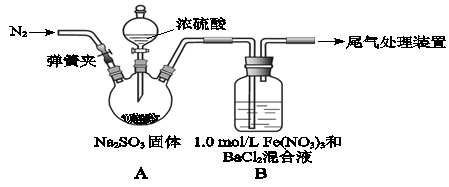
ij�о�С��̽��SO2��Fe(NO3)3��Һ�ķ�Ӧ��

��֪�� 1.0 mol/L��Fe(NO3)3��Һ��pH=1�� ����̨��֧����������ȥ ��
��ش�
��1��װ�������������ԵĴ���ָ������������ �� ��װ��A�з�����Ӧ�Ļ�ѧ����ʽ�� ��
��2���μ�Ũ����֮ǰҪ�ȴ��ɼ�ͨ��һ������N2����Ŀ���� ��
��3��װ��B�������� ��
��4��ʵ���У�װ��C�в����˰�ɫ�������ó����ijɷ��� ����һ����˵��SO2���� �ԡ�
��5������C�в�����ɫ������ԭ��
�۵�1��SO2��Fe3+��Ӧ��
�۵�2��������������SO2��NO3����Ӧ��
�۵�3�� ��
���ڹ۵�1��װ��C�з�Ӧ�����ӷ���ʽ�� ��Ϊ֤���ù۵㣬����������ṩ���Լ��У�ѡ���ʵ����Լ���һ��ȷ�Ϸ�Ӧ�����ɵ������ʣ��ٶ��ڷ�Ӧ��SO2�������㣩
A.KSCN��Һ B.���Ƶ���ˮ C.����KMnO4��Һ D.NaOH��Һ
��1��A���ټ���װ�ã�ֻ��ʯ���������֣���C������β������װ��
Cu + 2H2SO4(Ũ) ![]() CuSO4+ SO2��+ 2H2O
CuSO4+ SO2��+ 2H2O
��2����ȥ����װ���е���������ֹ����+4��
��3����ȫƿ����ֹC��Һ�嵹����A��
��4��BaSO4 ��ԭ��
��5�� �۵�3��SO2��Fe3+������������NO3������Ӧ
SO2+2Fe3++Ba2++2H2O=BaSO4��+2Fe2++4H+
C

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�