��Ŀ����
����̼ѭ������������ĸ߶����ӣ���֪ú������ȿ�����ˮ������Ӧ������CO��H2Ϊ���ĺϳ������ϳ����й㷺Ӧ�á��Իش��������⣺
��1����¯������CO�������Ҫ��;֮һ���������ӦΪ��
FeO(s)��CO(g) ![]() Fe(s)��CO2(g) ��H ��0
Fe(s)��CO2(g) ��H ��0
��֪��1100��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��K=0.263��
���¶����ߣ���ѧƽ���ƶ���ﵽ�µ�ƽ�⣬��ʱƽ�ⳣ��Kֵ__________�����������С�����䡱����
��1100��ʱ��ø�¯�У�c(CO2)=0.025mol��L-1��c(CO)=0.1 mol��L-1��������������£��÷�Ӧ��_______���У�����ҡ������ж�������
��
��2��һ����̼��������Ӧ���ɼ״�������ΪCu2O/ZnO����
��֪��2CO(g) + O2(g)��2CO2(g) ��H ���C566 kJ��mol�D1
2H2O(g) ��2H2(g) + O2(g) ��H ��+484 kJ��mol�D1
CH3OH(g) + 1/2O2(g)��CO2(g) + 2H2(g) ��H ���C192.9 kJ��mol�D1
��д��CO��H2��һ�������·�Ӧ����CH3OH(g)���Ȼ�ѧ����ʽ
��
��3��Ŀǰ��ҵ��Ҳ����CO2������ȼ�ϼ״����йط�ӦΪ��CO2(g)+3H2(g)![]() CH3OH(g)+H2O(g) ��H ��-49.0 kJ��mol-1���������Ϊ1 L���ܱ� �����У�����1mol CO2��3mol H2����Ӧ�����в��CO2��CH3OH(g)��Ũ����ʱ��ı仯��ͼ��ʾ��
CH3OH(g)+H2O(g) ��H ��-49.0 kJ��mol-1���������Ϊ1 L���ܱ� �����У�����1mol CO2��3mol H2����Ӧ�����в��CO2��CH3OH(g)��Ũ����ʱ��ı仯��ͼ��ʾ��
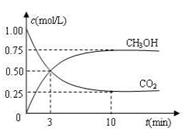
�ٴӷ�Ӧ��ʼ��ƽ�⣬������ƽ����Ӧ����v(H2)��_______ _��
�ڷ�Ӧ�ﵽƽ������д�ʩ��ʹ![]() �������______������ţ���
�������______������ţ���
A�������¶� B���ٳ���H2 C���ٳ���CO2
D����H2O(g)����ϵ�з��� E������He(g)
��1���� ����2�֣���
�� �ң�1�֣�����Ϊc(CO2)/c(CO)=0.25��0.263��2�֡�ֻҪ����������ɣ�
��2��CO(g) +2 H2(g) �� CH3OH(g) ��H���C90.1kJ��mol�D1 ��4�֡�û������״̬����ȷ�����֣�
��3����0.225mol/(L??min) ��3�֡���λ�����֣���B D��2�֡�ֻ��1��������ȷ��1�֣�2�������ϴ�ÿ���1������1�֣������ָ��֡���
����:
��

 ��2013?����һģ������̼ѭ������������ĸ߶����ӣ�����ν��ʹ�����CO2�ĺ�������Ч�ؿ�������CO2��������ȫ������ձ����ӣ����ԡ���̼���á�����Ϊ��ѧ���о�����Ҫ����
��2013?����һģ������̼ѭ������������ĸ߶����ӣ�����ν��ʹ�����CO2�ĺ�������Ч�ؿ�������CO2��������ȫ������ձ����ӣ����ԡ���̼���á�����Ϊ��ѧ���о�����Ҫ���� ����̼ѭ������������ĸ߶����ӣ���֪ú������ȿ�����ˮ������Ӧ������CO��H2Ϊ���ĺϳ������ϳ����й㷺Ӧ�ã��Իش��������⣺
����̼ѭ������������ĸ߶����ӣ���֪ú������ȿ�����ˮ������Ӧ������CO��H2Ϊ���ĺϳ������ϳ����й㷺Ӧ�ã��Իش��������⣺ CO2��g����H2��g�����õ������������ݣ�
CO2��g����H2��g�����õ������������ݣ�
 Fe(s)��CO2(g) ��H ��0
Fe(s)��CO2(g) ��H ��0 CH3OH(g)+H2O(g) ��H ��-49.0 kJ��mol-1���������Ϊ1 L���ܱ� �����У�����1mol CO2��3mo
CH3OH(g)+H2O(g) ��H ��-49.0 kJ��mol-1���������Ϊ1 L���ܱ� �����У�����1mol CO2��3mo l H2����Ӧ�����в��CO2��CH3OH(g)��Ũ����ʱ��ı仯��ͼ��ʾ��
l H2����Ӧ�����в��CO2��CH3OH(g)��Ũ����ʱ��ı仯��ͼ��ʾ��
 �������______������ţ���
�������______������ţ���