��Ŀ����
��������һ����Ҫ�����ȼ�ϣ�����ͨ��CH3OH���Ӽ���ˮ�Ƶã�2CH3OH(g)  CH3O
CH3O CH3(g) �� H2O(g) ��H����23.5kJ��mol��1����t1�棬�����ܱ������н�������ƽ�⣬��ϵ�и����Ũ����ʱ��仯��ͼ��ʾ��
CH3(g) �� H2O(g) ��H����23.5kJ��mol��1����t1�棬�����ܱ������н�������ƽ�⣬��ϵ�и����Ũ����ʱ��仯��ͼ��ʾ��

(1)�������·�Ӧƽ�ⳣ������ʽK��______________����t1 ��ʱ����Ӧ��ƽ�ⳣ��Ϊ____________���ﵽƽ��ʱn(CH3OCH3)��n(CH3OH)��n(H2O)=_____________________��
(2)��ͬ�����£����ı���ʼŨ�ȣ�ijʱ�̸����Ũ������Ϊ��c(CH3OH)��0.4 mol��L��1��c(H2O)��0.6 mol��L��1��c(CH3OCH3)��2.4mol��L��1����ʱ�����淴Ӧ���ʵĴ�С��v��________v��(�>������<������)����Ӧ��__________��Ӧ�������(��������桱)����
���� ֪���淴Ӧ��M(g)��N(g)
֪���淴Ӧ��M(g)��N(g)  P(g)��Q(g) ��H��0����ش��������⣺
P(g)��Q(g) ��H��0����ش��������⣺
(1)��ij�¶��£���Ӧ�����ʼŨ�ȷֱ�Ϊc(M)��1 mol��L��1��c(N)��2.4 mol��L��1���ﵽƽ���M��ת����Ϊ60%����ʱN��ת����Ϊ____________��
(2)����Ӧ�¶����ߣ�M��ת����__________(���������С�����䡱)��
(3)����Ӧ�¶Ȳ��䣬��Ӧ�����ʼŨ�ȷֱ�Ϊc(M)��4 mol��L��1��c(N)��a mol��L��1���ﵽƽ���c(P)��2 mol��L��1��a��____________��


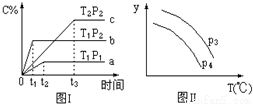
 xC(g)����ͼ����ʾ�ķ�Ӧ���ߣ����ж϶�ͼ���˵������ȷ����(T��ʾ�¶ȣ�P��ʾѹǿ��C%��ʾC���������)( )
xC(g)����ͼ����ʾ�ķ�Ӧ���ߣ����ж϶�ͼ���˵������ȷ����(T��ʾ�¶ȣ�P��ʾѹǿ��C%��ʾC���������)( )